Synthesis of Substituted Quinazolines Containing Pharmacophoric Groups
Harutyunyan Artur A1,2*, Ghukasyan Gohar T1 and Danagulyan Gevorg G1,2
1 Russian Armenian (Slavonic) University, Armenia, Europa
2 Scientific and technological center of organic and pharmaceutical chemistry, Armenia
Submission: September 28, 2018 Published: October 01, 2018
*Corresponding author: Harutyunyan Artur A, Scientific and technological center of organic and pharmaceutical chemistry National Academy of Sciences of the Republic of Armenia, Armenia, Email: harutyunyan.arthur@yahoo.com
How to cite this article: Harutyunyan A A, Ghukasyan G T, Danagulyan G G. Synthesis of Substituted Quinazolines Containing Pharmacophoric Groups. Organic & Medicinal Chem IJ. 2018; 8(1): 555729. DOI: 10.19080/OMCIJ.2018.08.555729
Introduction
Continuing research on the synthesis of biologically active quinazoline derivatives [1,2] in this report we have described the synthesis of previously unknown substituted 2-methylquin-zolins and 2-[2-aryl(hetaryl)vinyl]quinazolines, which contain pharmacophore groups at different positions of the ring. Synthesis was carried out by the interaction of 2-methyl-4H-3,1-benzoxzin-4-ones 1a, b with aromatic and heterocyclic amines, according to the Scheme 1.
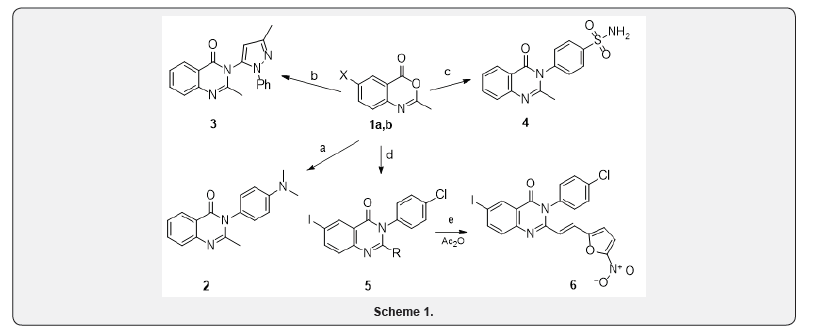
1a, b: X = H (a), I (b). a) 4-N,N-dimethylaminoaniline, b) 5-amino-3-methyl-1-phenyl-1H-pyrazole, c) 4-aminobenzenesulfonamide, d) 4-chloroaniline, e) 5-nitrofuran-2-carbaldehyde.
Fragments of biologically active compounds are introduced into the target compounds:
4-N,N-dimethylaminoaniline, 3-methyl-1-phenylpyrazole, as well as fragments of antibacterial preparations of 4-aminobenzenesulfonamide and 5-nitrofuran. In the preparation of quinazolines 2-5, the best results are obtained when the reaction is carried out under the conditions of co-heating of benzoxazines 1a, b with the corresponding amines, and quinazoline 3 in polyphosphoric acid. Heating of 2-methylquinazoline 5 with 5-nitrofurancarbaldehyde in acetic anhydride gave the substituted (5-nitrofuryl)ethenylquinazoline 6.
References
- Harutyunyan AA, Ghukasyan GT, Panosyan HA, Danagulyan GG (2018) Chem J Armenia 71(1-2): 249-253.
- Harutyunyan AA, Ghukasyan GT, Danagulyan GG (2018) International Conference 100 Years of Development of Chemistry: From Synthesis of Polyethylene to Stereodivergence. Dedicated to the 100th anniversary of the Department of Organic Chemistry of Perm State University. Perm pp. 103.






























