Limit of Blank (LOB), Limit of Detection (LOD), and Limit of Quantification (LOQ)
Mohamad Taleuzzaman*
Assistant Professor, Pharmaceutical Chemistry, Glocal School of Pharmacy, Glocal University, India
Submission: August 13, 2018; Published: August 30, 2018
*Corresponding author: Mohamad Taleuzzaman, Assistant Professor, Pharmaceutical Chemistry, Glocal School of Pharmacy, Glocal University, Mirzapur Pole, 247121Saharanpur, U.P, India, Email: zzaman007@gmail.com
How to cite this article: Mohamad T. Limit of Blank (LOB), Limit of Detection (LOD), and Limit of Quantification (LOQ). Organic & Medicinal Chem IJ. 2018; 7(5): 555722. DOI: 10.19080/OMCIJ.2018.07.555722
Keywords:LOB, LOD, LOQ and Signal to Noise
Abbrevations:ICH: International Conference on Harmonisation; USFDA: United states food and drug administration; AOAC: Association of Analytical Communities; USP: United States Pharmacopoeia; IUPAC: International Union of Pure and Applied Chemistry
Short Communication
For any analytical method development and validation, it is require developing arobust, accurate, precise, and linear method which is the heart for the development activities for drug product and drug substances. It is vital importance to initiate through a systematic process that the analytical method under question is acceptable for its intended purpose. Determining the limit of what and require for the quantitative determination is according of regulatory authorities globally. Limit of detection (LOD) and limit of quantitation (LOQ) can be found in the International Conference on Harmonization’s (ICH) Q2 Validation of Analytical Procedures. LOD and LOQ are parameters employ to explain the smallest concentration of an analyte that can be reliably measured by an analytical procedure. The LOQ is lowest concentration that quantitatively measured suitably with accuracy and precision while the LOD is the concentration that can be detected. The Most typical practice for determining the LOD /LOQ is to determine ratio of signal to noise. If the ratio is 3:1 it is LOD and if it is 10:1 than it is LOQ.
Introduction
In 1968 Currie published the hypothesis testing approach to detection decision and limits in chemistry this approach has gradually been accepted as detection limit theory. Analytical method development and validation strategy are crucial in the discovery and development of drugs and pharmaceuticals. Every year, millions of analyses of so many kinds are happened around the world, and millions of decisions are made, based on these analyses have the medicaments, the amount of drug reported in their container? Based on these results yet several questions are rises. Can we safely consume this water or these foods or medicaments? Analytical methods are used to aid in the process of drug synthesis, screen potential drug candidates, support formulation studies, monitor the stability of bulk pharmaceuticals and formulated products, and test final products for release. The quality of analytical data is a key factor in the success of a drug and formulation development program [1]. During the post approval commercial production stage of bulk drugs and pharmaceutical products, the official or in-house test methods that have resulted from the analytical method development and validation process cycle become indispensable for reliable monitoring of the integrity, purity, quality, strength and potency of the manufactured products. There is often a need to transfer methodology from one laboratory to another and/or to include it in official compendia. Limit of detection (LOD) and limit of quantitation (LOQ) parameters are related but have distinct definitions and should not be confused. The purpose is to define the lowest concentration of analyte that can be detected with no assurance about the bias or imprecision of the result by an assay, the concentration at which quantitation as defined by bias and precision goals is workable, and finally the concentration at which the analyte can be quantitated with a linear response [2]. Comparison of regulatory authorities such as United States Pharmacopoeia (USP), [3] Foods and Drugs Administration (FDA), [4]. International Union of Pure and Applied Chemistry (IUPAC), [5] International Conference on Harmonisation (ICH) [6] and Association of Analytical Communities (AOAC) [7,8] for limit of detection and limit of quantitation are produced.
Signal to Noise Ratio (S/N) is a dimensionless measure of the comparatively strength of an analytical signal (S) to the median strength of the background instrumental noise (N) for a particular sample and is closely related to the detection level. The ratio is useful for determining the effect of the noise on the relative error of a measurement. The S/N ratio can be measured a variety of ways, but one convenient way to approximate the S/N ratio is too divide the arithmetic mean of a series of replicates by the standard deviation of the replicate results [9].
Note: For chromatographic analyses, the S/N ratio can be calculated directly with a ruler by taking the raw chromatogram or strip chart output and measuring the distance from the signal peak to the midline between the maximum and minimum noise at baseline, off-peak. This is the signal (S). The noise (N) is the distance between the maximum and minimum baseline response, off-peak
Linear Regression
Linear calibration curve, it is presume that the instrument response y is linearly related to the standard concentration x for a limited range of concentration. In Pharmaceutical industry the role of method development and validation is very important but in other hand it is very problematic to that one method is hundred per cent correct. May be a method that is valid in one situation could well be invalid in another. An accurate and precise validation of analytical procedure is the process of govern the correctness of a given methodology for providing useful analytical data. For analytical method development the limit of detection (LOD) and the limit of quantitation (LOQ) are important parameters that need to be determined during method development and validation for any analytical procedure in either spectroscopy or chromatography [10-15].
Limit of Blank (LOB)
EP17 defines LOB as the highest observable analyte concentration assumes to be found when reproduce of a sample containing no analyte are examined. Note that while the samples examined to define LOB are lacking of analyte, a blank (zero) sample can produce an analytical signal that robustness otherwise be consistent with a low concentration of analyte. LOB is determined by measuring duplicates of a blank sample and calculating the mean result and the standard deviation (SD).
Suppose a Gaussian distribution of the raw analytical signals from blank samples, the LOB represents 95% of the observed values. (Note: typical modern clinical analysers don’t routinely display the actual analytical signal but automatically convert it to a concentration value. The raw analytical signal is preferable for establishing LOB as analysers may report all signal values below a certain fixed limit as “zero concentration”).The remaining 5% of blank values represent a response that could actually be produced by a sample containing a very low concentration of analyte. Statistically, this false positivity is known as a Type I (or α) error (Figure 1). Conversely, while a sample that actually contains analyte is expected to exceed the LOB, it must also be identify that a proportion of very low concentration samples will build responses less than the LOB, representing Type II (or β) error (Figure 1). Thus, EP17 admit that the overlap of the analytical responses of blank and low concentration is a statistical reality and uses LOB as a reasonable starting point for estimating the LOD.
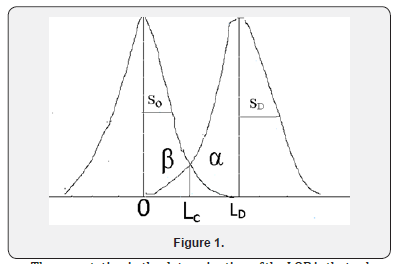
The expectation in the determination of the LOB is that values truly the 95th percentile of the spreed of values on truly blank or negative samples deviate significantly from blank or negative measurements. When a sample produces an observed value that exceeds this limit, it may be declared to contain an amount of analyte the at exceeds zero [NCCLS document EP17-A].
Determination of LOB -A α value of 5% corresponds to using the 95th percentile of the distribution of blank or negative values as the limit for declaring a measured value significantly higher than the blank or negative
LOB= μB+1.645σB
Given a Gaussian distribution of blank or negative values, this limit corresponds to where μB and σB are the mean and standard deviation of the blank or negative measurements, respectively [15-18].
Limit of Detection (LOD)
In spite of reagent package lodge may state that an assay has a dynamic range that enlarge from zero concentration to some upper limit, typically an assay is simply not capable of accurately measuring analyte concentrations down to zero. Adequate analyte concentration must be present to yield an analytical signal that can actually be well known from “analytical noise,” the signal produced in the absence of analyte. LOD is the lowest analyte concentration likely to be reliably prominent from the LOB and at which detection is operable. It is therefore greater than LOB. A standard and typical resemble to estimate LOD consists of measuring replicates, usually n=20, of a zero calibrator or blank sample, determining the mean value and SD, and calculating LOD as the mean +2 SD. Variations of this approach use the meanplus 3, 4, or even 10 SDs to provide a more conservative LOD. The assumption is that if analyte is present, it will produce a signal greater than the analytical noise in the absence of analyte. This is a simple and quick method. The weakness is that there is no objective evidence to prove that. LOD Experiment Design A minimum of 10 individual samples with concentrations ranging from the LOB to approximately 4xLOB shall be tested at least 10 times by at least two technicians in a minimum of two days or runs.
A pooled SDS estimate can be derived from repeated measurements with the set of samples. (a minimum of 10 measurements of at least 10 samples) Measurements shall be carried out by different technicians in different days or runs to be able to capture the true variability of the analytical method [19-21].
The SDS is the pooled estimate after checking the statistical assumption of homogeneity of variance
An estimate LOD is then obtained as:
LD= Kασ0+ KβσD
SDS is the estimated standard deviation of the sample distribution at a low level.
Limit of Quantitation (LOQ), or Lower Limit of Quantitation (LLOQ)
The level above which quantitative results may be obtained with a specified degree of confidence. The LOQ is mathematically defined as equal to 10 times the standard deviation of the results for a series of replicates used to determine a justifiable limit of detection. Limits of quantitation are matrix, method, and analyte specific.
LC=K σ0
LOQ =10 SD/S
SD= Standard deviation of the response based on either the standard deviation of the blank, the residual standard deviation of the regression line or the standard deviation of y= Intercepts of regression lines and S = Slope of the calibration curve.
An appropriate number of samples should be analysed at the limit to validate the level. In practice it is almost never necessary to determine the actually LOQ (Figures 1 & 2) [22-25].
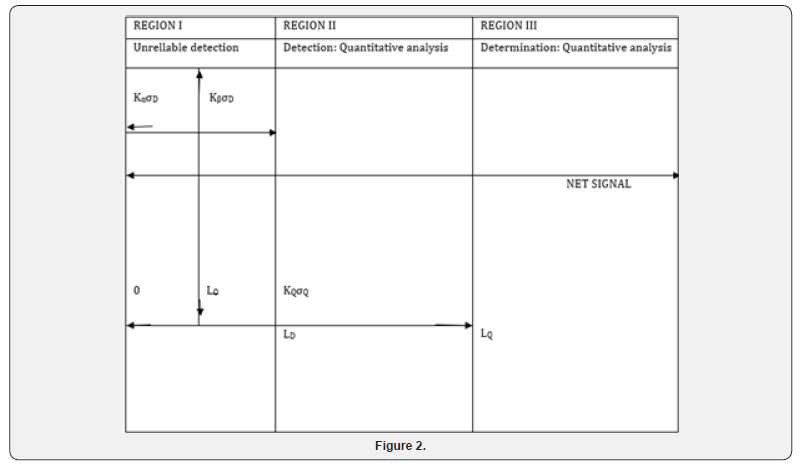
Guidelines
Definition:
a) ICH: Lowest amount of analyte in a sample, which can be quantitatively determined with suitable precision and accuracy.
b) USFDA: The lowest amount of analyte that can be quantitatively determined with suitable precision and accuracy also called LLOQ (Lower limit of quantification).
c) AOAC: The limit of quantitation is the lowest amount of analyte in a sample, which can be quantitatively determined with precision and accuracy appropriate to analyte and matrix considered.
d) USP: Lowest amount of analyte in a sample, which can be quantitatively determined with suitable precision and accuracy.
e) IUPAC: Not defined.
Method:
a) ICH: By visual evaluation Based on S/N ratio Applicable to procedure, which exhibits base line noise? Low conc. of analyte is compared with blank. Based on S.D. of response and slope LOQ=10σ/s s – Slope of calibration curve σ – S.D. of response can be obtained by Standard deviation of blank response Residual standard deviation of the regression line Standard deviation of the y-intercept of the regression line S y/x, i.e. standard error of estimate.
b) US FDA: Preparation of standard curve and lowest conc. on the calibration curve should be accepted as LLOQ if it satisfies following condition. Response at LLOQ=5×Response by blank analyte peak should be identifiable discrete and reproducible with precision of 20% and accuracy of 80%- 120%.
c) AOAC: Not specified.
d) USP: By visual evaluation Based on S/N ratio Applicable to procedure, which exhibits base line noise? Low concentration of analyte is compared with blank. Based on S.D. of response and slope LOQ=10σ/ss – Slope of calibration curve σ – S.D. of response can be obtained by Standard deviation of blank response Residual standard deviation of the regression line Standard deviation of they-intercept of the regression line S y/x, i.e. standard error of estimate.
e) IUPAC: Not recommended; only recommends expressing uncertainty of measurement as function of concentration.
Recommendation:
a) ICH: Limit should be validated by analysis of suitable no. of samples known to be near or prepared at quantitation limit.
b) US FDA
c) AOAC: Not specified.
d) USP: Not specified.
e) IUPAC: Not specified.
Expression/Calculation:
a) ICH: Limits of quantitation and method used for determining should be presented. Expressed as analyte concentration.
b) US FDA: Not specified .
c) AOAC: Mean value of the matrix blank reading plus 10standard deviations of the mean, expressed in analyte concentration.
d) USP: Expressed as analyte concentration (% or ppb).
e) IUPAC: Not specified.
Acceptance Criteria:
a) ICH: Not specified.
b) US FDA: Not specified.
c) AOAC: Not specified.
d) USP: Not specified.
e) IUPAC: Not specified.
Conclusion
It is important to fully characterise the analytical performance in order to understand their capability and limitations, and to ensure that they are “fit for purpose.” The terms LOB, LOD, and LOQ describe the smallest concentration of a measure and that can be reliably measured by an analytical procedure. To establish these parameters a large number of sample replicates to increase the robustness and the statistical confidence of the estimate. In addition, a manufacturer establishing the LOB, LOD, or LOQ should perform studies using more than one analyser and one lot of reagents to encompass the variability that users can expect to encounter in the field. LOB and LOD are important to describe between the presence and absence of an analyte and LOQ to reliably measure lowest levels of concentration.
References
- Hirsch AF (1989) Good laboratory practice regulations. New York: Marcel Dekker, USA.
- (1998) EURACHEM Guide: The fitness for purpose of analytical methods. A Laboratory Guide to method validation and related topics, Teddington: LGC.
- (2012) Title 21 of the US Code of federal regulations: 21 CFR 58, Good laboratory practice for nonclinical laboratory studies. Washington: U.S. Government Printing Office, USA.
- (2001) ICH Q7A: Good Manufacturing practice guide for active pharmaceutical ingredients. Rockville: ICH.
- (2009) USP 32- NF27 Validation of compendia methods. Rockville: USP.
- (2005) ISO 9000: Quality management systems - Fundamentals and vocabulary. Geneva: ISO.
- (1995) ISO, Guide to the expression of uncertainty in measurement. Geneva: ISO.
- Validation of analytical procedures: Text and methodology Q2 (R1). Rockville: ICH; 2005 Limit of Detection and Limit of Quantification Determination in Gas Chromatography.
- Thompson M, Ellison RL, Wood R (2002) Harmonized guidelines for single laboratory validation of methods of analysis. Pure Appl Chem 74(5): 835-855.
- US FDA (2000) Guidance for Industry: Analytical Procedures and Methods validation: Chemistry, Manufacturing, and Controls and Documentation, Rockville: FDA.
- Lopez Garcia P, Buffoni E, Pereira Gomez F, Vilchez Quero JL (2011) Analytical method validation. Wide spectra of quality control.
- (1994) Validation of Chromatographic Methods. Rockville: Center for Drug Evaluation and Research.
- (2005) Validation and Peer Review of US EPA Chemical Methods of Analysis, EPA.
- Currie LA (1995) Nomenclature and evaluation of analytical methods, including quantification and detection capabilities. Pure Appl Chem 67(10): 1699-1723.
- Foley JP, Dorsey JG (1984) Clarification of the limit of detection in chromatography. Chromatographia 8(9): 503-511.
- International Organization for Standardization (1997) Capability of detection Part 1: Terms and definitions. Geneve: ISO.
- Long GL, Winefordner JD (1983) Limit of detection, a closer look at the IUPAC definition. Anal Chem 55(7): 712A-724A.
- Currie LA (1968) Limits for qualitative detection and quantitative determination. Anal Chem 40(3): 586-593.
- Chang KH (2011) Limit of detection and its establishment in analytical chemistry. Health Env J 2(1): 38-43.
- Needleman SB, Romberg RW (1990) Limits of linearity and detection for some drugs of abuse. J Anal Tox 14(1): 34-38.
- Putheti RR, Okigbo RN, Patil SC, Advanapu MS, Leburu R (2008) Method Development and Validations: Characterization of Critical Elements in the Development of Pharmaceuticals. Int J Health Res 1(1): 5-14.
- (2007) European Pharmacopoeia European Directorate for the Quality of Medicines. Strasbourg.
- (2000) Analytical Procedures and Methods Validation: Chemistry, Manufacturing, and Controls, Federal Register 65: 776-777.
- (1996) International Conference on Harmonization (ICH) of Technical Requirements for the Registration of Pharmaceuticals for Human Use, Validation of analytical procedures: Text and Methodology. ICH-Q2B, Geneva.
- Currie LA (1997) Detection: International update, and some emerging di-lemmas involving Calibration, the blank and multiple detection decisions. Chem Intell Lab Sys 37: 151-181.






























