Biomedical and Fungicidal Application of Copper Surfactants Derived From Pure Fatty Acid
Arun Kumar Sharma1*, Rashmi Sharma2 and Antima Gangwal2
1Department of Chemistry, Government P.G. College Jhalawar, India
2Department of Chemistry, S.P.C. Govt. College Ajmer, India
Submission: March 22, 2018 Published: April 03, 2018
*Corresponding author: Arun Kumar Sharma, Department of Chemistry, Government P.G. College Jhalawar-326001, Rajasthan, India, Email: sharmaarun423@gmail.com
How to cite this article: Arun K S, Rashmi S, Antima G. Biomedical and Fungicidal Application of Copper Surfactants Derived From Pure Fatty Acid. Organic & Medicinal Chem IJ. 2018; 6(1): 555680. DOI: 10.19080/OMCIJ.2018.06.555680
Abstract
The anti-fungal activities of the pure ligands, pure soaps and their four corresponding complexes have been evaluated by testing these against Alternaría alternata at different concentration by Agar plate technique. All complexes show higher activity than pure soaps and ligands suggesting that complexes are more powerful anti-fungal agents further N, S, O etc containing compounds like benzothiazole are able to enhance the performance of copper soaps. These results show that the complexes of ligands (-Cl and -CH3 substituted benzothiazole) are much more toxic than the ligands themselves. Their efficiency increases with their concentration.
Keywords: Surfactants; Caprylate; Caprate; 2-amino; 6-chloro benzothiazole; 6-methyl benzothiazole; Benzene; Methanol
Introduction
Recently, the deeper understanding of the role of metal ion in bio-system has led to the awareness that metal complexing is useful in the treatment of bacterial, fungicidal and viral diseases. Most of the antimicrobial agents are obviously organic compounds, which behave as good chelating agent. Their pathogenic activity is enhanced on complexing with various transition, inner transition and toxic metals. The biological effect of these derivatives depends on the nature and structure of ligands and their metal complexes and also on the presence of particular element [1,2].
Nitrogenous ligands have been found to be effective against many metal enzymes, bacteria and number of fungi. Large number of compounds containing nitrogen and sulpher atoms in the heterocyclic ring shows different types of activities. Some examples are given here possessing benzothizaole or similar type of structure containing nitrogen and sulpher atoms in the aromatic cyclic ring. Recently, Wood has developed metabolic antagonist theory of drug action and Fields led to the synthesis of several co-ordination compounds, which have been used for antibacterial and anti-tubercular activity [3,4].
Synthesized and evaluated biological activities of some new 2-arylamino-4-fluoroaryl thiazoles, were found to possess fungicidal, herbicidal and anti-arthritic activities. Thus on the basis of the foregoing studies, it becomes very clear that benzothiazole and their substituted derivatives have been found to be very active against variety of bacteria, fungi, herbs and insects [5,6]. It has also been established by several workers that chelation of the benzothiazole and their substituted derivatives enhances the biological and fungicidal activity [7,8]. The ligands used during the present investigation viz. 2-amino-6-substituted (-Cl and -CH3) benzothiazole have been very well reported to possess biocidal activity. Our continuing interest in the search for better fungicides and bactericides has led us to synthesise some new complexes derived from Cu-caprylate and caprate soaps with the above mentioned ligands and screen them for their fungicidal activities [9,10].
Experimental
The general laboratory techniques followed in the course of this investigation are as suggested by Booth and Hawks worth [11].
Sterilisation of Glasswares
Glasswares used in our present study were of Pyrex brand. The glassware viz. test tubes, conical flasks, pipettes (micro and macro), glass rods and petridishes were thoroughly washed after rinsing with chromic acid each time before sampling. The petriplates and other glassware were then sterilised in hot air oven at 1600C for 24 hours before use.
Culture Media Used
The culture medium used for the growth of the organism of the present study was P.D.A. The following media were used in the present study [12] (Table 1).

200gm of potatoes were cleaned, cut into pieces and boiled in about 1000ml of tap water for 2 hours. Then the contents were strained using muslin cloth. To this extract 20gm of dextrose, 20gm of agar were added and made upto 1000ml in graduated flask, before sterilising the medium.
Preparation of Sample Solutions
The four complexes were prepared by Copper (II) caprylate and caprate soaps with 2-amino-6-chloro benzothiazole and 2-amino-6-methyl benzothiazole. They were designated as CCpl ,ACB, CCpr ACB, CCpl .AMB and CCpr AMB. All chemicals used were of reagent grade. The calculated amount of the complex was weighed in a standard flask and the solution containing different concentration of complex in methanol-benzene mixtures of varying compositions were prepared.
Test Organism
The test organism used in the present study was Alternaria alternata which was isolated from its natural habitat [plants, (debris)]. Purified, characterised and identified [13].
Fungicidal Testing
1 ml of the diluted solution [complex dissolve in 40% methanol-benzene solvent mixture and 80% methanol- benzene solvent mixture] was aseptically transferred into sterile petriplates. Into these plates, 20 ml of P.D.A was poured and was mixed with complex solution by rotating the petriplates in clockwise and anti clock wise direction 3-4 times and was allowed to solidify. After the solidification of the above medium, single hypha / spore of Alternaria alternata were aseptically transferred in the centre of the petriplates. The plats were incubated at 28 + 10C for 7 days. After the period of incubation the plats were observed for the growth of fungus in different concentration of the complex solution used in the present study The data were statistically analysed according to the following formula [14]:
% Inhibition= (C-T)/C*100
C = diameter of fungal colony in control plate after 7 days;
T = diameter of fungal colony in test plate after 7 days
Results and Discussion
The antifungal activities of the pure ligands, pure soaps and their corresponding complexes have been evaluated by testing against Alternaria-alternata at different concentration by Agar plate technique [15]. A perusal of results reveals that all complexes show higher activity than pure soaps and ligands suggesting that complexes are more powerful anti-fungal agents and benzothiazole and other N, S, O etc. containing compounds are able to enhance the performance of copper soaps. These results show that the Cu (II) soap-complexes of ligands [-Cl and -CH3 substituted benzothiazole] are much more toxic than the ligands themselves. The enhanced activity of newly synthesized complexes as compared to those of the ligand can possibly be explained on the basis of chelate formation, presence of donor atoms, basicity as well as the structural compatibility with molecular nature of the toxic moiety. Enhanced biological activity of complexes is in accordance with the chelation theory [16].
In final conclusion, that the appearance of enhanced activity may be due to synergistic mechanism i.e. the free ligands are less active but on complexation show more activity in combination with copper (II) soaps. The studies suggest that the copper (II) ions in soaps may be responsible for enhancement of the activity against fungi [17]. The evaluation of anti-fungal studies further revealed that fungi toxicity of the complexes also depends on the nature of metal ions. Their efficiency increases with their concentration [18,19]. Thus it is evident that concentration plays a vital role in increasing the degree of inhibition. So fungicidal screening data revealed that at lower concentration the inhibition of growth is less as compared to higher concentration (Figures 1-3).
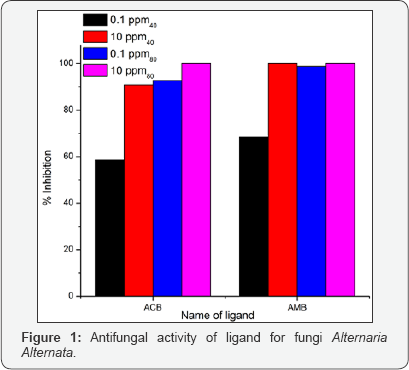
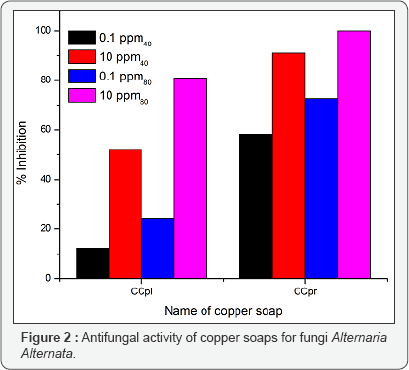
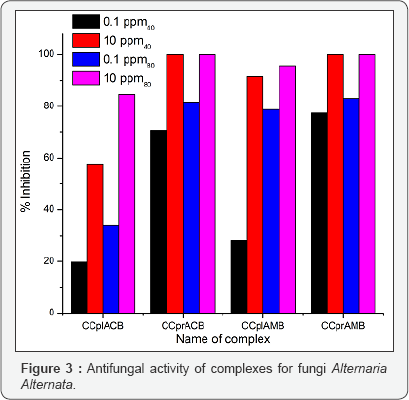
CCplAMB, CCprAMB complexes are more active than CCplACB, CC ACB complexes. Their activities also enhanced with thepr increase in chain length of the soap segment in the complex [CCpr ACB, CCpr AMB complexes are more effective in all cases]. In all complexes toxicity increases with the domination of methanol in the solvent mixture of methanol and benzene [40% methanol- benzene and 80% methanolbenzene], suggesting that complexes are more effectively playing there role to check the growth of fungi in the domination of polar solvent in the ternary system: complex + methanol + benzene. Some recent studies about antimicrobial activities of newly synthesised 1, 4-benzothiazines possessing thiazolyl/imidazolyl moieties also support our studies [20].
Conclusion
CCplAMB, CCprAMB complexes are more active than CCplACB, CC ACB. Their activities also enhanced with the increase inCCpr Their actives also enhanced with the in chain length of complex. In all complexes toxicity increase with the domination of methanol in the solvent mixture of benzene and methanol suggesting that complexes are more effectively playing their role to check the growth of fungi in the presence of polar solvent.
All the above studies made by us leads to the conclusion that copper soaps having many fungicidal, herbicidal and other biological activities possessing a tendency to make complexes with donor atom containing ligands. Their performance enhanced by the presence of the other biologically active segment in the complex. Their micellar features and other physical properties analyzed and reported in the above studies will play a significant role in their applications in various fields of industries, wood preservation and agriculture etc.
Acknowledgment
The authors pay their sincere gratitude to Principal, S. P. C. Govt. College, Ajmer, S.D. Govt. College Beawar, Rajasthan (India) for providing necessary research facilities to accomplish this study.
References
- Rashmi S, Arun KS (2017) Natural Edible Oils: Comparative Health Aspects Of Sesame, Coconut, Mustard (Rape Seed) and Groundnut (Peanut) A Biomedical Approach. Biomed J Sci & Tech Res 1(5): 000441.
- Tank P, Sharma AK, Sharma R (2017) Thermal Behaviour and Kinetics of Copper (II) Soaps and Complexes Derived from Mustard and Soyabean Oil. J Anal Pharm Res 4(3): 1-5.
- Sharma S, Sharma R, Sharma AK (2017) Synthesis, Characterization, and thermal degradation of Cu (II) Surfactants for sustainable green chem. Asian J Green Chem 2(2): 130-141.
- Sharma S, Sharma R, Heda LC, Sharma AK (2017) Kinetic parameters and Photo Degradation studies of Copper Soap derived from Soybean Oil using ZnO as a Photo catalyst in Solid and Solution Phase. J Inst Chemists 89(4): 119-136.
- Tank P, Sharma R, Sharma AK (2017) A Pharmaceutical approach & Antifungal activities of Copper Soaps with their N & S donor complexes derived from Mustard and Soyabean oils. Glob J Pharmaceu Sci 3(4): 555619.
- M Saxena, R Sharma, AK Sharma (2017) Micellar Features of Cu (II) Surfactants derived from Edible Oils. LAP Lambert Academic Publishing Germany.
- Kumar A, Rajput CS (2009) Synthesis and anti-inflammatory activity of newer quinazoline-4-one derivatives. Eur J Med Chem 44(1): 8390.
- Sharma AK, Saxena M, Sharma R (2017) Synthesis, spectroscopic and fungicidal studies of Cu (II) soaps derived from groundnut and sesame oils and their urea complexes. Bulletin of Pure and Applied Sciences. 36(2): 26-37.
- Mahajan K, Swami M, Singh RV (2009) Microwave synthesis, spectral studies, antimicrobial approach, and coordination behavior of antimony (III) and bismuth (III) compounds with benzothiazoline. Russ J Coord Chem 35(3): 179-185.
- Khan S, Sharma R, Sharma AK (2018) Acoustic studies and other Acoustic Parameters of Cu(II) Soap derived from nonedible Neem oil (Azadirecta indica), in Non-aqueous media at 298.15. Acta Ac united Ac 104(2): 277-283.
- Sharma AK, Saxena M, Sharma R (2018) Synthesis, Spectroscopic and Biocidal activities of environmentally safe Agrochemicals. J Biochem tech 7(3): 1139-1147.
- Garg BS, Kumar DN (2003) Spectral studies of complexes of nickel (II) with tetradentate schiff bases having N2O2 donor groups. Spectrochim Acta 59A: 229-234.
- Joram A, Sharma R, Sharma AK (2018) Thermal degradation of complexes derived from Cu(II) groundnut soap (Arachishypogaea) and Cu(II) sesame soap (Sesamum indicum). Z phys chem.
- Sharma AK, Saxena M, Sharma R (2018) Synthesis, Spectroscopic and Biocidal activities of environmentally safe Agrochemicals. J Biochem Tech 7(3): 1139-1147.
- Mishra AP, Mishra RK, Shrivastava SP (2009) Structural and antimicrobial studies of coordination compounds of VO(II), Co(II), Ni(II) and Cu(II) with some Schiff bases involving 2-amino-4- chlorophenol. J Serb Chem Soc 74(5): 523-535.
- Bhutra R, Sharma R, Sharma AK (2018) Synthesis, Characterization and fungicidal activities of Cu(II) surfactants derived from groundnut and mustard oils treated at high temperatures. J Inst Chemists 90(3): 66-80.
- Sharma AK, Sharma R, Saxena M (2018) Biomedical and antifungal application of Cu(II) soaps and its urea complexes derived from various oils. Open access J Trans Med res 2(2): 40-43.
- Khan S, Sharma R, Sharma AK (2017) Antifungal Activities of Copper Surfactants derived from Neem (AzadirectaIndica) and Karanj (Pongamiapinnata) Oils: A Pharmaceutical Application. Glob J Pharmaceu Sci 3(4): 55561
- Sharma AK, Sharma S, Sharma R (2017) Thermal degradation of Cu(II) metallic Soaps and their Characterizations. A Pharmaceutical Application Chro Phar Sci 1(5): 312-319.
- Sharma AK, Saxena M, Singh N (2017) Synthesis, spectroscopic and biocidal activity of Cu(II) sesame complex. LAP Lambert Academic Publishing Germany.






























