Basis and Applications of Silicon Reinforced Adhesives
Natalia Chacon Camacho1, Jose Roberto Vega Baudrit1, 2* and Yendry Corrales Urena2
1Escuela de Quimica, Universidad National, Costa Rica
2Laboratorio National de Nanotecnologia LANOTEC CENAT, Centro National de Alta Tecnologia, Costa Rica
Submission: December 23, 2017 Published: January 17, 2018
*Corresponding author: Jose Roberto Vega Baudrit, Escuela de Qui'mica, Universidad Nacional, Heredia, Costa Rica, USA, Email: jvegab@gmail.com
How to cite this article: Natalia C C, Jose R V B, Yendry C U. Basis and Applications of Silicon Reinforced Adhesives. Organic & Medicinal Chem IJ. 2018; 5(1): 555654.DOI10.19080/omcij.2018.05.555654
Abstract
In recent years, the adhesive industry has been improving the properties of its products for the better mechanical, functional, electrical properties, among others. This is really important to expand its applications market to increasingly specialized fields such as medical, electronics and engineering. Nanoparticles have become a highly functional additive that greatly improves the parameters mentioned above. The knowledge of its physicochemical properties as well as its interaction with polymer matrices is of great importance in reinforcing the adhesives with these particles. Specifically, silicon has been widely used for the synthesis of silicon oxide and silicon carbide nanoparticles, which have had a wide range of adhesive applications lately. The objective of this review is to lay the foundations of the chemistry of the main types of adhesives, the use of silicon nanoparticles, as well as their synthesis methods and their advantages in the reinforcement of adhesives and to expose some of the most significant advances of the improvement of adhesives with silicon in various industrial applications.
Keywords: Adhesives; Silica nanoparticles; Polyurethanes; Epoxies; Acrylics; Thermoresistence; Mechanical improvement; Conductivity; Peel strength; Electronics; Engineering; Medicine
Abbreviations: TDI: Toluene Diisocyanate; MDI: Methane Diisocyanate; PDMA: Poly Dimethyl Acrylamide; PSAs: Pressure Sensitive Adhesives; MPS: Methacryloxy Propyl Trimethoxysilane; GMA: Glycidyl Methacrylate; Vac: Vinyl Acetate; CMP: Chemo Mechanical Polishing
Introduction
Today, there are hundreds of commercial structural adhesives throughout the world that often differ only in name or in the formulation of certain additives for their improvement. The adhesives may be defined as a material which, when applied to certain surfaces, may adhere and resist separation. This binding should be capable of producing a high modulus, high strength, binding bonds and be able to transmit structural stress but without loss of structural integrity within the design limits. The set of physic-chemical interactions generated between the adhesive and the adherent surface is called adhesion [1,2]. However, chemical bonds are not required, if not more Van der Waals forces and hydrogen bonds since the long chains of the polymers that form the adhesive have multiple points of contact with the surfaces to which they are attached. Adhesives have a small volume compared to glass, metals, wood, paper and plastics [3,4].
There are many types of classification for adhesives, being perhaps the most important the physical and chemical classification. Physically, adhesives can be classified as liquid adhesives, film adhesives and two-part liquid adhesives. Chemically, they are classified by the type of polymeric material used for their manufacture. The five basic chemical compositions for the synthesis of adhesives are polyurethane, acrylic (reactive, anaerobic or toughened), cyanoacrylate, epoxy and silicone [1]. In recent years, the use of additives in adhesives of different types has been studied to improve its mechanical properties and functionality. The use of nanotechnology has demonstrated the incorporation of metal nanoparticles as nanofillers to adhesives such as silicon, aluminum, titanium and zinc oxides, carbide compounds, boron nitride particles, silicon carbide particles and mixtures of compounds such as epoxies with silicone. It has been found that the organic characteristics of the base polymers give the adhesives characteristics of flexibility and process ability, but that adding inorganic components improves the properties of thermo resistance and mechanical strength [5-7].
Also, the use of nanoparticles has advantages over the use of the classic micro-reinforcements, as they allow thin-layer bonding lines and therefore reduce the risk of embrittlement within the body of the adhesive material, improving the adhesive tensile strength. In the case of silica (SiO2) this is the most abundant compound on earth and there are various types of artificial silica on the market that differ in their synthetic methods [5]. Examples are precipitated and pyrogenic silica. With the latter, it has been found that by adding them to polyurethane adhesives the adhesion properties are improved. The pyrogenic silica present siloxane groups, responsible for the highly inert character of the silica and silanol groups on its surface, which give it hydrophilic properties. The pyrogenic silica as an additive of an adhesive acts as a rheological agent, controlling the viscosity, thixotropic, and acting as emulsifying agent and sedimentation control. An example of this is the addition of silica in percentages less than 2% In rubber-bonded polyurethane adhesives, where the viscosity increases, which also increases the resistance to thermal aging [8-10].
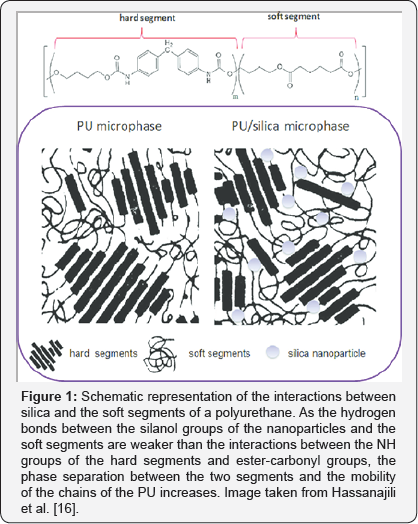
Vega et al. [10] used hydrophilic nanosilica treated with silanes to decrease its silanol group content and treated polyurethane adhesives containing 10 wt% fumed silica with different degree of silanization to verify that addition of silica increases the degree of phase separation between the hard and soft segments of the PU. As can be seen in Figure 1, this was due the interaction of the hydrogen groups between the silanol groups on the silica surface and the soft segments of the polymer, because the interactions between silanol and carbonyl groups are weaker than those between NH and ester-carbonyl groups, increasing the mobility of the chains and improving molecular ordering. With this, the thermal stability of the polymer was improved and the decomposition kinetics was decreased. Other studies have also found that by adding fillers of fumed pyrogenic nanosilicate (Non-porous synthetic silica obtained by hydrolysis of SiCl4 in a flame of oxygen and hydrogen) of varying degrees of hydrophilicity to solvent-based polyurethane adhesives, phase segregation increases with the degree of hydrophilicity of the silica through the formation of the hydrogen bonds described above.
In addition, silica favours phase separation by increasing the specific surface area of the silica and hence the content of silanol groups, thus improving the tensile strength, immediate adhesion to PVC, thermal, rheological, mechanical and adhesion properties of the polyurethanes, at the same time that the elongation at break of the polyurethane decreased by increasing the silanol content of the nanosilica [9,11-15]. Recent studies have also tested the effectiveness of silica nanoparticles on the surface of epoxy composite adhesives, where filler has been able to significantly increase elastic modulus, fatigue yield, longitudinal compression strength and longitudinal and transversal tensile strengths [1618]. In the present work, we intend to present a review of the last ten years in the use of silicon nanoparticles as reinforcement of the main types of adhesives such as polyurethanes, epoxies and acrylics, as well as their most recent applications in different industries as in the industry of construction, batteries, conductive adhesives and biomedical applications.
Polyurethane, epoxy and acrylic Adhesives chemistry
Although there are many chemical forms of adhesive bases, without considering the additives, the most used are epoxy, polyurethanes and acrylics. Being the first ones that have a larger scope of current applications. Each of these types are characterized by having different components, methods of synthesis, curing, and physical properties. Also, hybrids of these types of adhesives, such as epoxy / acrylic or epoxy / polyurethane, have been used to obtain or improve mainly mechanical, thermal and optical properties [19,20].
Polyurethanes
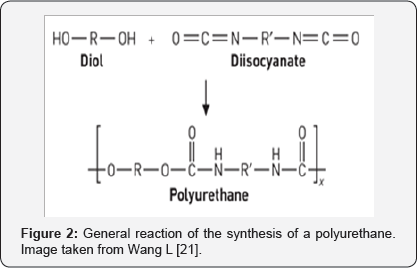
They are polymers which include a significant number of urethane groups independently of the composition of the rest of the molecule. These urethanes in turn are formed by the reaction of an isocyanate with an active hydrogen compound bound to a nitrogen atom. In addition to these urethane groups, they may contain in their chains, aromatic and aliphatic hydrocarbons, esters, ethers, amides and urea groups. The most important reaction of the isocyanates to form adhesives is with alcohols, wherein the isocyanates react with di- or polyfunctional hydroxyl compounds. Generally, they are usually polyesters (more rigid) or polyether's (more flexible) with terminal hydroxyls. This reaction occurs at room temperature without the need for catalysts and the reactivity is higher for primary alcohols, followed by secondary, tertiary and aromatic alcohols. Figure 2 shows a schematic reaction of the synthesis of polyurethanes.
One thing that is not desirable in the preparation of adhesives is the reaction of isocyanates with water, since it forms carbamic acid, an unstable compound. The isocyanates for the synthesis of adhesives may be aliphatic or aromatic and some of the most common are toluene diisocyanate (TDI), 4, 4-diphenylmethane diisocyanate (MDI), and polymeric MDI. In the case of polyols, polyether polyols based upon polyoxypropylene polyols are often the polyols of choice for polyurethane sealants, and polyesters produced from polyalkylene phthalate or adipates are preferred because they produce high strength and modulus. Polyurethanes can be bonded to almost all materials including glass, plastic, metals and stones. In addition, they have very good impact resistance and the best resistance to low temperatures of all the adhesives [9,19-22].
Epoxy's

Are the most common adhesive, formed by the addition of two dissolutions? The fist dissolution is made of a polymer containing an oxidant group that forms a cross linking (curing) between chains when is treated with a nucleophile (resin), and the other dissolution is formed of a poli aminated compound that starts the reaction because of it nucleophilic behaviour (hardener or curative).The hardener can also be an acid, phenol, alcohol or thiol [23]. This reaction is shown in Figure 3. They have a great versatility in their formulation due to the great variety of resins and hardeners available. They can be found from little viscous liquids to films or pastes. They have the best chemical resistance, produce few volatiles during curing and have small shrinkage values. These adhesives can be especially well bonded to metals, ceramics and most polymers including many thermoplastics [19]. Both epoxy and polyurethanes can have either singlepart or two-part formulations. The difference lies in the curing conditions of the adhesive. The single component adhesives have a curing process at a fixed temperature condition, whereas in the two component adhesives the curing is carried out in two stages with different ambient conditions [19].
Acrylic's
Acrylic adhesives have several classifications including anaerobic acrylics, cyanoacrylates and reactive acrylics. The acrylic structural adhesives are based on acrylate monomers or more commonly methacrylate to prevent acrylate malodors. They usually consist of two components. The first is a peroxide compound which acts as an oxidizing agent, while the second is an amine or a salt of a metal which acts as a reducing agent. It reacts with the peroxide compound upon mixing to initiate the free radical polymerization of the methacrylate monomers. Benzoyl peroxide and tertiary aromatic amines were the first compounds used and the most common ones. However, in recent years many variations have been proposed in the composition of acrylic adhesives such as the use of chlorosulfonated polyethylene in a mixture with methacrylate monomers and the use of amine aldehyde (butyraldehyde-aniline) as activator to start the curing process. Some of the most important variations in structural acrylic adhesives include elastomer-based modified methyl methacrylate adhesives, non-methyl methacrylate adhesives, and polymer blends as modifiers in methyl methacrylate based adhesives [19,24-26].
Silicon Nanoparticles
Nanoparticles are microscopic objects with at least one dimension less than 100 nm and with a large surface area that offers distinctive size-dependent properties. Due to its broad scope of applications, publications on the use of nanoparticles have increased from approximately 50 per year for 2004 to almost 300 per year by 2012 [27]. Silicon nanoparticles have been well studied for their easy preparation, well-defined dimensions and geometries and their functional properties. For this reason, they present a great variety of applications including catalysts support, pigments, chemo-mechanical polishing (CMP), adhesives and coatings, hybrid composite materials, humidity sensors, electronic and thermal insulators, polymer reinforcement, memory devices and biomedical diagnosis [2830].
Silicon nanoparticles can exhibit different geometries and this can greatly influence the interaction with the polymer because of its different physicochemical properties. For this reason, the shape of the particles is an extremely important aspect to consider according to each application. Among the most important geometries are the spherical nano particles, short and long nanorods, nanofibers or whiskers, nanosheets and nanowires. These can vary in size, with particles ranging from just over 10 nm to some 1000 nm in their wide range of applications. According to their porosity, they can be classified into non-porous and mesoporous nanoparticles [31-34]. The most used in polyurethanes adhesives reinforced with silicon nanoparticles is the fumed silica which is usually spherical and manufactured by high-temperature hydrolysis of silicon tetrachloride in a flame to produce silanol and siloxane groups on the silica surface.
The silanol groups are dominant on the hydrophilic fumed silica surface and may be chemically modified by reaction with silanes to produce the hydrophobic fumed silica, which has a similar particle size but a reduced specific surface area and a reduced silanol group density, compared to the hydrophilic fumed silica. The improvement in the properties is due to the creation of hydrogen bonds between the silanol groups on the nano silica surface and the urethane and/or ester carbonyl groups of the soft segments in the polyurethane, resulting in phase separation [35]. On the other hand, for epoxies and acrylics, perhaps the most widely used method of synthesis of silicon nanoparticles for applications in adhesives is the sol-gel process or in situ process, where colloidal precursors are generally used, generally alkoxides giving rise to a diphasic (gel) consisting of a solid and a liquid phase, resulting from small particles to continuous polymer networks.
The remaining liquid is then removed by different methods such as drying and centrifugation [36]. The most common precursor in the synthesis of silicon oxide nanoparticles istetraethyl orthosilicate (TEOS) [37,38]. However, there are also other methods among like solvothermal synthesis, plasma synthesis, chemical precipitation, chemical vapour deposition, microemulsion and carbonation under pressure [27,39]. Nowadays you can find in the industry different types of nanoparticles with different geometric and size characteristics. The nanoparticles manufacturing industry is expanding and some nanoparticles are available in large (tonne) quantities. While prices are often high, they have come in dread and vary greatly with the type of nanoparticles as well as with the purity of the material. For example, in the case of silicon nanoparticles, they are supplied dispersed to a master batch in epoxy at a price of $20 per kilogram [40].
Reinforced adhesives with NPS
For the reinforcement of adhesives, nanoparticles of silicon oxide (SiO2) or nanoparticles of silicon carbide (SiC) are usually used [41]. The interfacial interactions between nanoparticles and polymer-matrix are very important in the local stress transfer and determining the quality and properties of the nanocomposites. Also, particle shape is important in determining the properties of the nanoparticles-modified materials like stiffness, flow characteristics, tensile strength and others. The challenge in the composite fabrication is to provide a sufficiently high tensile strength. The aggregation and dispersion of the particles are also important and may also affect the properties [40,42].
Methods of Incorporation
To incorporate the nanoparticles into the adhesives, four methods are mainly used: dispersion, mixing, sonication and alignment. The biggest problem that exists at the time of modifying the adhesives with nanoparticles is the agglomeration of these, reducing their performance. The agglomerates are almost impossible to eliminate once formed. The dispersion of nanoparticles is also difficult due to its large surface area and its incompatibility with the polymer matrix, and it is often necessary to treat the surface prior to dispersion. Because of these problems, it is common to use combinations of methods to create these reinforced adhesives. For example, sonication- mixing or dispersion-sonication. Thus, agglomerates are avoided, but it is important to note that the combination of these techniques could also damage the particles if the appropriate parameters are not applied [40].
What Properties Improve Silicon Nanoparticles on Adhesives?
Basically, there are six parameters that can be improved or modified with the addition of nanoparticles to polymer matrices. First and perhaps the most important: the mechanical properties of the new material. By adding some rigid material of any size, such as silicon nanoparticles, the polymer module increases, and the increase being proportional to the size of the particles. Another parameter is the functional properties, especially the increase of the glass transition temperature. Studies conclude that this increase is due to the interaction between the particles and the polymer, which locally changes the properties of the polymer network. An example is the research by Bugnicourt et al. [43]. They reported that Tg increased from 160 °C up to 180 °C for an amine-cured epoxy modified by silica nanoparticles. They also noted that the poorer the dispersion, the lower the impact of the addition of silica on the magnitude of the Tg [40,43].
The electrical properties are the third important parameter that nanoparticles vary in the adhesives. These tend to increase conductivity and many layered silicates in epoxy resin improves the surface discharge endurance of the polymer considerably, increasing the time before electrical breakdown occurs [44]. The resistance to the fracture is also benefited with the addition of nanoparticles, being this greater among the smaller the particles. In the case of silicon nanoparticles, studies have reported a double or higher increase in fracture toughness (Kc) and fracture energy (Gc) of epoxy resins with silica percentages of 10%. Fatigue performance is also enhanced by the addition of nanoparticles. Finally, the peel and lap shear performance is also influenced by nanoparticles. It has been seen that with additions of 50% silica the peel force is increased by 40% [45-47].
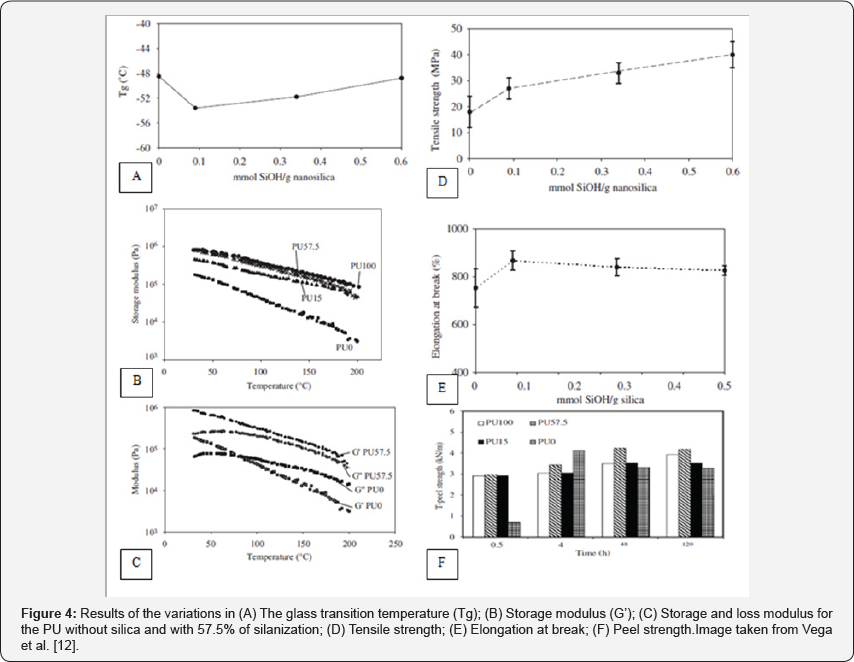
Figure 4 shows an example of silicon nano-reinforced polyurethanes taken from a study by Vega et al. [12]. This figure 4 shows some of the results discussed above as the variation in Tg, the storage and loss modulus, the tensile strength, elongation at break and peel strength for adhesives with four different percentages of nanosilica. Also, studies have revealed that by adding nano silicon particles to polyurethanes with the same NCO / OH ratio, the glass transition temperature, the crystallization temperature and the melting temperature are decreased, while the crystallization notch is significantly increased. These results are given because when there is no addition of silica, the phase separation increases with increasing NCO / OH ratio due to repulsion between the polar hard segments and the nonpolar polyol chains. However, with the addition of hydrophilic silica, the degree of separation is further enhanced by the interaction between the silanol groups and the polymer by hydrogen bridges [48].
Applications of Silicon Reinforced Adhesives
Adhesive technology has been of great importance in the construction industry, paints, electronics, footwear and textiles, automobiles, food, among others. The improvement of adhesives with silicon loads has allowed improving many products in the last 10 years. Some of the most recent applications are discussed below.
Conductive Materials and Electronic Field
Conductive adhesives are widely used in electronic packaging, one of the most important parts of the modern electronics industry. It has been wanted to replace the alloys welds with heat-sealable adhesives because of their good electrical conductivity, low cost and advantages for the environment. A group of researchers from the Institute of Nuclear and New Energy Technology in Beijing used nanoparticles of 50 nm silicon oxide as an additive to a silane-modified copper conductive adhesive consisting of an epoxy matrix with DDM and MPDA as curing agents. Although copper conductive adhesives have good electrical conductivity and high adhesion strength, copper tends to form a non-conducting oxide surface layer.
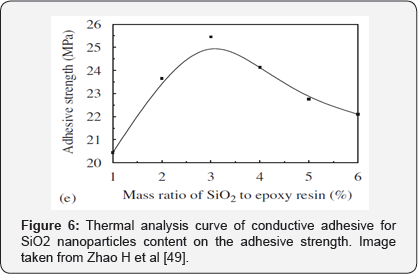
These researchers modified the copper powder with silanol to avoid this oxidation and improve the conductivity. As can be seen in Figure 5, with the addition of silane to the copper powder the agglomeration in the epoxy matrix was decreased and there was a better dispersion thanks to the bonds between the hydrolysable groups of the silanol and the polar groups of the copper. Also, the R groups of the silanol can form strong bonds with the epoxy matrix. Addition of silicon nanoparticles strengthened the samples to a pressure of 25 MPa with a SiO2/ epoxy ratio of 3: 100, as can be seen in Figure 6. However, with a higher content of nanoparticles the strength of the material began to decrease. It was concluded that the adhesion strength of the conductive adhesive could be increased by 30% and it was verified that when the material is impacted, the nanoparticles can disperse the stress and prevent the crack diffusion [49].

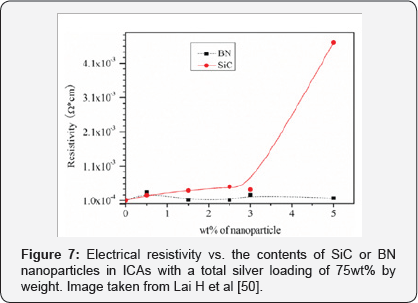
MOther more recent research has used boron nitride and silicon carbide nanoparticles to compare their effectiveness in the electrical resistivity and thermal conductivity of isotropic conductive adhesives (ICAs) also composed of an epoxy matrix and silver flakes as the conductive filler. In this case, the nanoparticles were 100 nm and added at different concentrations. The results showed that the content of boron nitride nanoparticles had no effect on electrical resistivity but silicon carbide nanoparticles did increase this parameter in concentrations between 3 and 5%, as can be seen in Figure 7. In addition, it was verified that the thermal conductivity of the adhesives with nanoparticles of SiC is greater than that of the nanoparticles of BN and that this increases with the increase of the concentration of nanoparticles, if it is below a concentration of 3% [50].
Engineering Field and Construction
MAnother wide application of the adhesives relies on the engineering area as adhesion joints to replace the seals and mechanical seals, reducing prices, reducing stress, low weight, less processing and greater environmental resistance and fatigue. The main problem presented by the adhesive adhesives are their poor mechanical properties since they are generally weaker than the pieces that must be joined, which invade hard and resistant materials because of their industrial application [51,52]. For example, the resistance of hybrid acrylic/silicon adhesives with substrates such as wood, aluminum and propylene glycol has been analyzed and it was concluded that the shear strength of the joints was improved with the nanoparticles, which was attributed to the change in the failure mode of a failure at the interfaces in the acrylic adhesive only to a slip failure in the hybrid adhesive [47].
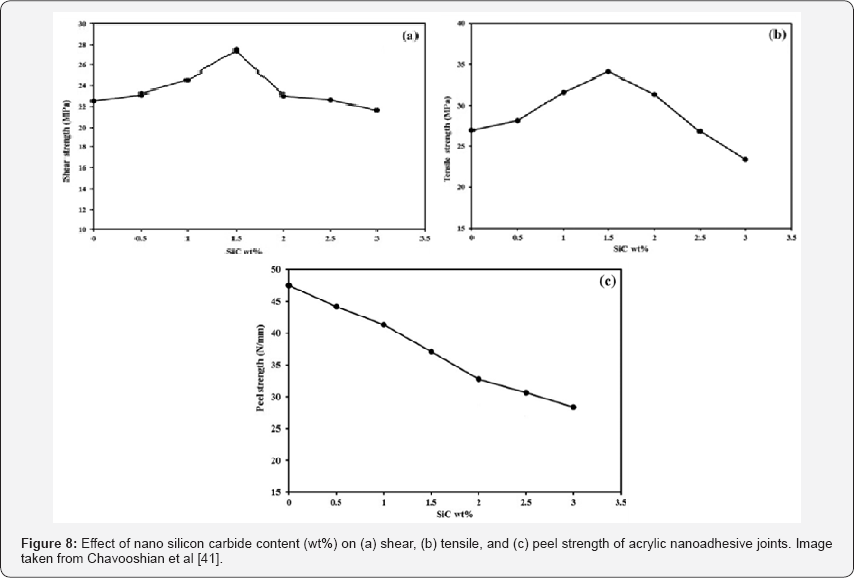
MAnother study by Chavooshian et al. [41] used silicon carbide nanoparticles of 20 to 30 nm diameter in two-part structural acrylic adhesives to improve the adhesion strength of steel-glass / epoxy composite joints. They observed that in a range of 0 -1.5 wt% they were incremental in 22 and 26% the shear strength and the tensile strength, respectively. This is due to the efficient stress transfer between nanoparticles and polymer matrix, where the local stress can be more easily transferred onto the joined particles, which leads to a larger local plastic deformation of the matrix. The result is a higher joint strength when the particles are in intimate contact with the polymer matrix. At concentrations, greater than 1.5 wt% the shear and tensile strength started to decrease because the nano silicon carbide particles fully fill the gaps and porosities and all the contact points are established, further addition of nanoparticles cannot interact effectively within the acrylic adhesive and consequently Poor matrix infiltration occurs. Also with high amounts of nanoparticles, the adhesive viscosity increases enough to cause a non-uniform distribution of the particles within the adhesive matrix [41].
MThe other important result was the decrease of the peel strength with the addition of nanoparticles caused by a reduction in the mobility of the polymer chains and an increase in the Tg. The increase of Tg is due to a strong interfacial bonding between the organic polymer matrix and silicon carbide nanoparticles. The silicon carbide nanoparticles that strongly interact with the polymeric network can restrict the segmental motion of the polymeric chains effectively. Figure 8 shows de principals results of this study. Also, experiments have been conducted with the addition of silicon nanoparticles to epoxy adhesives to be used in titanium bonding for aerospace applications. A group of researchers of Netherlands and Canada developed an adhesive based on 10 per cent silicate nanoparticles dispersed in an ultra- high temperature-resistant epoxy. Titanium joints made with this adhesive were subjected to cryogenic (-196°C) and elevated temperatures (+300°C) for 100h.
Despite exposure to these extreme conditions, the joint retained 95 per cent of its ambient temperature strength. A significant increase in the shear strength of the joints was observed. Titanium is widely used in aerospace applications such as solid rocket booster cases, guidance control pressure vessels and a wide variety of applications requiring lightweight and reliability. This is desired due to the exceptional strength and high resistance to elevated temperatures and corrosion [53,54]. Likewise, silicon nanoparticles have been used as filler in acrylic adhesives to improve the adhesion of steel with a glass / epoxy composite. The results showed a remarkable increase in the shear and tensile strength of the composite joints. With the increase of silica content from 0 to 1.5%, the shear and butt-joint tensile strength increased from 22.6 to 29.2 MPa (29%) and 27 to 35 MPa (30%), respectively.
By increasing the silica content, the glass transition temperature (Tg) values of the nanocomposites also increased noticeably due to the strong interfacial interactions between the organic polymer matrix and silica nanoparticles. Also, the addition of silica nanoparticles effectively improves the interfacial wettability and chemical compatibility of acrylic adhesive with steel and glass / epoxy substrates, increasing shear and tensile strength of joints [52]. Another application that has been optimized with the use of silicon nanoparticles are the protective varnishes since the adhesion strength and the heat tolerance of the varnish depends on the adhesive properties. Epoxy adhesives were used with nanocomposites of epoxy/ montmorillonite and mixed with 1200 mesh silicon carbide nanoparticles by different weight-nanoparticle ratios and the nanoparticles were found to be evenly dispersed in the adhesive and no agglomeration was seen.
The conductive activity of the varnish was thanks to the nanoparticles. In addition, because of its small size and large surface area and dispersion, the bond strength to the matrix increases significantly by improving stress transfer and improves the macroscopic properties of the material [55]. Polyurethanes have also been modified with silicon and alumina nanoparticles to improve abrasion, scratch, UV and solvent resistance, thermal stability, and other mechanical properties in protective varnishes of wood products based on UV cured nanocomposites [56]. UV- waterborne technology presents a faster polymerization process without VOCs emission. Also, the use of water as unique solvent reduces the viscosity of coating formulations and allows it use and spray applications in more ecological and safe conditions.
Likewise, UV-cured coatings have very good chemical and heat resistance and waterborne coatings give better adhesion particularly on wood.
However, two of the disadvantages are the need for predrying to eliminate water and poor wetting. It has been seen that for complete UV curing strict temperature control and high humidity are required [57,58].
In this regard, Sow et al. [59] reinforced UV cured polyurethane-acrylate composites with silicon nanoparticles of 12nm modified with methacryloxypropryltrimethoxysilane (MEMO) groups [59]. It was observed that, although TEM demonstrated that nanoparticles dispersion in UV-cured coatings was not efficient, as aggregates larger than nanometre size was present, there was a clear improvement of scratch resistance and aggregates did not affect the adhesion between UV-waterborne coatings and Sugar maple samples. Also, the efficiency of the modification of the surface of the nanoparticles was demonstrated. With this the amount and size of the aggregates were decreased and the reactive groups of the acrylates were increased. It was generally observed that the implementation of silica improves mechanical, optical and thermal properties to a greater extent than reinforcement with alumina [59].
Medical Field
In medical industrial adhesives are also widely used. In dentistry, different percentages of SiO2-BaO fillers were used which were silanized with silica from organosilane-methacry loxypropyltrimethoxysilane as coupling agent. The grains had a size of d50 / d99 = 180/500 nm and irregular shape. The results showed that the incorporation of nanoparticles fillers in a particular self-etching monomer mixture has the potential to promote the strengthening of the adhesive resin and the adhesive interface, with improvements to the degree of conversion and polymerization efficacy. As drawbacks, fillers may substantially increase the viscosity of the resin and expedite water sorption into the adhesive layer and the elution of solubilized components. This is important in dental industry because these methacrylic adhesives are exposed to very high stresses and to the absorption of fluids. So far, these adhesives fail is a short or medium term.
Therefore, the addition of silicon fillers can help to prolong their useful life by improving their mechanical properties and thus their healing properties [60]. Also in the medical field, one of the most promising advances in recent years is the study of the addition of silicon nano particles to hydrogels to join them. Generally, the union of polymers with polymers is difficult and requires of chemical methods, heating, and changes of pH, ultraviolet radiation or electric fields. Many medical applications and emerging technologies such as the area of surgery, tissue engineering and micro fluidics would benefit greatly from this innovation [61]. In this regard used solutions of silicon nanoparticles of different sizes that adhered to poly (dimethyl acrylamide) (PDMA) hydrogels.
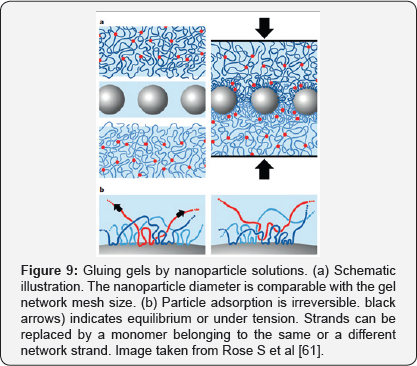
It was observed that the larger the particle size in solution, the greater the adhesion force. The particles are ashier to the surface of the hydrogel, whereby the surface of the particles must exhibit affinity for the polymer chains. Thus, in the adhesive layer, the particles act as connectors between the gel pieces, while the gel chains act as bridges between the particles. Nanoparticles retard failure and ensure good adhesion because the energy dissipated by the desorption effort is much greater than the energy gain that is obtained from the absorption of the monomers to the surface of the nanoparticles. In addition, when a piece of the modified hydrogel was immersed in water, it was subjected to a fivefold volume increase without failure. This is extremely useful since in medicine it is worked in fluids, so that with these modified adhesives biological and synthetic hydrogels and biological tissues can be joined without significantly affecting the rigidity or permeability of the whole. In the case of biological tissues, a calf liver test was also performed, where one piece was dispersed on the 60|iL surface of the nanoparticles solution and pressed for a few seconds against another untreated piece.
It was observed that both pieces adhered strongly and could be manipulated with ease [61]. The basis of this method can be seen schematically in Figure 9, where network chains are adsorbed on nanoparticles and anchor particles to gel pieces. Particles act as connectors between gel surfaces and adsorbed chains also form bridges between particles. Also, Particle adsorption is irreversible and, in equilibrium, strands can be replaced by a monomer belonging to the same or a different network strand, allowing for large deformations and energy dissipation under stress. One type of adhesive that has application in the medical, electronic, automotive, construction, among others, are pressure sensitive adhesives (PSAs), generally acrylic because of their resistance to oxidation and transparency when exposed to the sun. These are semi-solid materials with viscoelastic properties and adhesion on solid substrates after a small pressure is applied for a very short period.
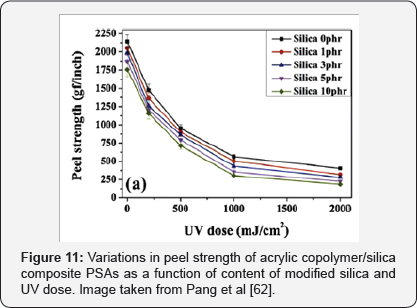
As these adhesives generally have no cross linking, but their polymer chains are connected by reversible hydrogen bonds, which interact with the carboxyl groups, have a low thermal stability. Pang et al. [62] Modified the surface of fumed silica powder of a size of approximately 5 to 15 nm withy- Methacryloxypropyl trimethoxysilane (MPS) so that they had vinyl groups on their surface and thus, with the presence of double carbon bonds, UV curing with acrylic copolymers were possible. Also, Glycidyl methacrylate (GMA) with a reactive vinyl group was employed to modify the acrylic copolymer to have UV curable sites. The results showed that the modified nanoparticles were much better dispersed in the polymer matrix than the unmodified ones.
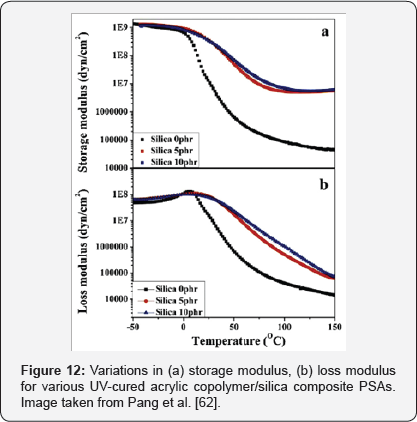
In addition, the thermal stability, both in terms of degradation temperature and residue weight, gradually improved with the increase in the modified silica content due to the greater bond strength between the particles and the PSA matrix. Also, the glass transition temperature and the storage modulus were increased due to the strong and extensive interfacial bonds between the organic polymer matrix and the nanoparticles. Something that decreased was the peeling strength of the composite with the increase of the amount of silica and the UV dose due to the great restriction in the mobility of the polymer chains due to the cross-linking [62]. Because of the characteristics of this modified material, the authors propose that it would be of great use in future applications of thin wafer manufacture [62].
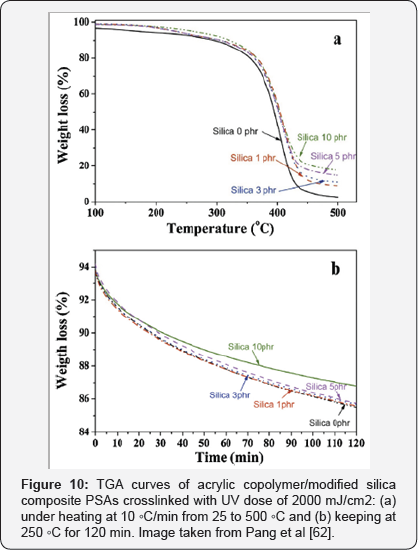
In Figure 10 the TGA curves of acrylic copolymer / silica composite PSAs cross linked can be observed. Also, Figure 11 shows the peel strength of acrylic copolymer / silica composite PSAs with variation of silica content and UV dose. Likewise, in Figure 12 the DMA thermo grams of various UV-cured acrylic copolymer / silica composite PSAs can be observed. The glass- transition temperature (Tg) region is generally identified as a strong decrease in G '.
Improvement of Natural Adhesives
The case for the improvement of natural adhesives with silicon nanoparticles is not out of the question. Natural sources of adhesives have been studied in recent years, including starch. It is cheap and renewable from many plants. Applications that have been used include binders, sizing materials, glues and pastes. As has generally been found to lack the ability to glue wood, Wang et al. Used 20 nm diameter silicon nanoparticles into vinyl acetate (VAc) grafted waxy corn starch to produce SiO2/ starch-based wood adhesive. They examined the bond strength and water resistance of the adhesive and observed a improved bonding strength and water resistance of the adhesive. The shear strength of adhesives in both dry and wet state increased with increasing silica nanoparticles content. The starch-based wood adhesive with 10% silica nanoparticles (w / w) was found to have the best shear strengths of 5.12 MPa in the dry state and 2.98 MPa in the wet state [62].
Future Perspectives and Conclusion
The adhesive industry is increasing day by day, penetrating new fields more functionalized. With this, there is a requirement in the improvement of many of the properties of these materials. Nano particles have become highly useful fillers in this type of material by not only improving their mechanical properties and the modulus of the polymer, but also providing improved electrical and functional properties, toughness, peel strength and rupture, among others. One of the main advantages of silicon, besides being extremely abundant in the earth, is the great number of methods of synthesis of nanoparticles, with many geometrical and functional possibilities and with the capacity of modifying its surface to adapt to the conditions of the polymer to be reinforced. One application that is gaining a lot of boom is the replacement of synthetic polymers by natural polymers that serve as adhesives like starch or even compounds present in the geckos' legs.
Although there is not much research on these new materials functionalized with silicon nanoparticles, it is a very promising improvement to improve the properties of these compounds with great potential of biomaterials. Also, combining silicon nanoparticles with other nanoparticles to form synergies would be an excellent option to reduce volumetric fractions. Likewise, the use of nanoparticles in composites is promising as these are small enough to flow between the fibres. Increased application of nanoparticles in structural adhesive reinforcements, increased availability and reduced costs are a great opportunity to continue research in this branch of nanotechnology to solve many of the problems that even Remain in the performance of many adhesives. These investigations and the application of new methods and improvement of new properties would help to prolong the useful life and efficiency of these products, as well as the penetration in new areas of the industry.
References
- Dillard DA (2010) Advances in structural adhesive bonding. CRC Press, USA.
- Kinloch AJ (1987) Adhesion and Adhesives : Science and Technology. Springer Netherlands.
- Gonzalez Sandi AY (2008) Utilizacion del name (Dioscorea alata) y del tiquisque (Xanthosoma sagittifolium) de rechazo para la obtencion de adhesivos naturales en base acuosa. Universidad Nacional.
- Skeist I (1990) Handbook of Adhesives. Springer, USA.
- May M, Wang HM, Akid R (2010) Effects of the addition of inorganic nanoparticles on the adhesive strength of a hybrid sol-gel epoxy system. Int J Adhes Adhes 30: 505-512.
- Cui H wang, Li D sheng, Fan Q (2013) Using nano hexagonal boron nitride particles and nano cubic silicon carbide particles to improve the thermal conductivity of electrically conductive adhesives. Electron Mater Lett 9(1): 1-5.
- He Y, Wang J A, Pei C L (2010) Novel epoxy-silicone thermolytic transparent packaging adhesives chemical modified by ZnO nanowires for HB LEDs. J Nanoparticle Res 12: 3019-3024.
- Vega Baudrit J, Sibaja Ballestero M, Martin Martinez JM, Patricia Vazquez, et al. (2007) Sintesis y caracterizacion de nanosi'lices funcionalizadas injertadas con 3-aminopropil trietoxisilano para ser utilizadas en adhesivos de poliuretano. Rev Iberoam Polimeros 8: 138162.
- Vega Baudrit J (2005) Modificacion de las propiedades de adhesivos de poliuretano por adicion de nanosi'lices con diferente polaridad superficial. Universidad de Alicante, Spain.
- Vega Baudrit JR, Martin Martinez JM, Camacho Elizondo M (2013) Influencia de las nanoparticulas de silice en polimeros termoplasticos. Mundo Nano 6: 40-51.
- Vega Baudrit J, Sibaja Ballestero M, Vazquez P, Nunez S,Martin Martinez JM, et al. (2008) Kinetics of isothermal degradation studies in adhesives by thermo gravimetric data: effect of hydrophilic nanosilica fillers on the thermal properties of thermoplastic polyurethane-silica nanocomposites. Recent Pat. Nanotechnol 2: 220-226.
- Jose Vega Baudrita, Virtudes Navarro Banonb, PatriciaVazquezc, Jose Miguel Martin Martinez, (2006) Addition of nanosilicas with different silanol content to thermoplastic polyurethane adhesives. Int J Adhes. Adhes 26: 378-387.
- Navarro Banon V, Vega Baudrit J, Vazquez P (2005) Interactions in nanosilica-polyurethane composites evidenced by plate-plate rheology and DMTA. Macromol Symp 221: 1-10.
- JoseVega-Baudrit, Maria Sibaja-Ballestero, Patricia Vazquez, Rosa Torregrosa-Macia, Jose Miguel Martin-Martinez, et al. (2007) Properties of thermoplastic polyurethane adhesives containing nanosilicas with different specific surface area and silanol content. Int J Adhes Adhes 27: 469-479.
- Una CR, Rica C (2009) Study of the Relationship between Nanoparticles of Silica and Thermoplastic Polymer ( TPU ) in Nanocomposites.
- Hassanajili S, Khademi M, Keshavarz P (2014) Influence ofvarious types of silica nanoparticles on permeation properties of polyurethane/silica mixed matrix membranes. J Memb Sci 453: 369-383.
- Uddin MF, Sun CT (2008) Strength of unidirectional glass/epoxy composite with silica nanoparticle-enhanced matrix. Compos Sci Technol 68: 1637-1643.
- Sprenger S (2013) Epoxy resin composites with surface-modified silicon dioxide nanoparticles: A review. J Appl Polym Sci 130: 14211428.
- Dillard DA (2010) Advances in structural adhesive bonding, CRC Press, USA.
- Hartshorn SR (1986) Structural Adhesives : Chemistry and Technology [Internet] Springer US. 21. Wang L (2017) Polyurethane Foam From furniture cushions to insulation, versatile polymer has found a world of applications p. 1-3.
- D M Segura, A D Nursea, A McCourta, R Phelpsa, A Seguraa, et al. (2005) Chapter 3 Chemistry of polyurethane adhesives and sealants. Handb Adhes Sealants 1: 101-162.
- Ege SN (1997) Qui'mica organica : estructura y reactividad Revertee.
- H Iovu, I Calinescu, M Radoiu, D Martin, E Mateescu, et al. Curing of the Epoxy Resins Under Microwave and / or Electron Beam Treatment p. 1-6.
- Bachmann A (1996) Advances in acrylic-adhesive technology. Adhes Sealants In: 36.
- Gosiewski D, Loven WE, Leeser DL, et al. (2002) Structural Adhesive. USA.
- Guo D, Xie G, Luo J (2014) Mechanical properties of nanoparticles: basics and applications. J Phys D Appl Phys 47: 13001.
- Oda S, Ferry D (2006) Silicon Nanoelectronics (1st edn). Group Taylor & Francis Group, LLC. Florida, USA.
- Chen KI, Li BR, Chen YT (2011) Silicon nanowire field-effect transistor- based biosensors for biomedical diagnosis and cellular recording investigation. Nano Today 6: 131-154.
- Luo Z, Hong RY, Xie HD (2012) One-step synthesis of functional silica nanoparticles for reinforcement of polyurethane coatings. Powder Technol 218: 23-30.
- Okamoto H, Sugiyama Y, Nakano H (2011) Synthesis and Modification of Silicon Nanosheets and Other Silicon Nanomaterials. Chem A Eur J 17: 9864-9887.
- Hartwig A, Luhring A, Trautmann J Spheroidal (2009) Nanoparticles in Epoxide-Based Adhesives. Macromol Mater Eng 294: 363-379.
- Xinglu Huang, Xu Teng, Dong Chen, Fangqiong Tang, Junqi He, et al.(2010) The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials 31: 438-448.
- Manuscript A, Threshold T. NIH Public Access (2013) 6: 2289-2301.
- Bahattab M (2012) Comparative Properties of Hydrophilic and Hydrophobic Fumed Silica Filled Two-Component Polyurethane Adhesives J Adhes Sci Technol 26: 303-315.
- Brinker CJ, Scherer GW (1990) Sol-gel science: the physics and chemistry of sol-gel processing [Internet].
- Yaripour F, Baghaei F, Schmidt I (2005) Synthesis of dimethyl ether from methanol over aluminium phosphate and silica-titania catalysts Catal Commun 6: 542-549.
- Heck CA, Dos Santos J H Z, Wolf CR (2015) Waterborne polyurethane: The effect of the addition or in situ formation of silica on mechanical properties and adhesion. Int J Adhes Adhes 58: 13-20.
- Guo C, Xing Z, Ma X, L Xu, Y Qian, et al. (2008) Solvothermal Synthesis of Si3N4 Nanomaterials at a Low Temperature. J Am Ceram Soc [Internet] 91: 1725-1728.
- Taylor A C (2010) Advances in nanoparticle reinforcement in structural adhesives. Woodhead Publ Ser Weld Other Join Technol, Woodhead Publishing Limited, UK.
- Chavooshian M, Kamali R, Tutunchi A (2017) Effect of silicon carbide nanoparticles on the adhesion strength of steel-epoxy composite joints bonded with acrylic adhesives. J Adhes Sci Technol 31: 345-357.
- Mammeri F, Bourhis E Le, Rozes L (2005) Mechanical properties of hybrid organic-inorganic materials J Mater Chem 15: 3787.
- Bugnicourt E, Galy J, Gerard JF, Herbert Barthel (2007) Effect of submicron silica fillers on the mechanical performances of epoxy-based composites. Polymer 48: 1596-1605.
- T Imai, F Sawa, T Nakano, T Ozaki, T Shimizu, et al. (2006) Effects of nano- and micro-filler mixture on electrical insulation properties of epoxy based composites. IEEE Trans Dielectr Electr Insul 13(2): 319326.
- G Ragosta, M Abbate, P Musto, G Scarinzi, L Masciab, et al. (2005) Epoxy-silica particulate nanocomposites: Chemical interactions, reinforcement and fracture toughness. Polymer (Guildf) 46: 1050610516.
- A J Kinloch, R D Mohammed, A C Taylor, C Eger, S Sprenger, et al. (2005) The effect of silica nano particles and rubber particles on the toughness of multiphase thermosetting epoxy polymers. J Mater Sci 40: 50835086.
- Patel S, Bandyopadhyay A, Ganguly A, Anil K Bhowmick (2006) Synthesis and properties of nanocomposite adhesives J Adhes Sci Technol 20: 371-385.
- Vega baudrit J, Carballo SM, Jose Miguel Martin Martinez (2012) Thermoplastic Polyurethanes-Fumed Silica Composites: Influence of NCO / OH in the Study of Thermal and Rheological Properties and Morphological Characteristics. Thermoplast Compos Mater p. 12-24.
- Zhao H, Liang T, Liu B (2007) Synthesis and properties of copper conductive adhesives modified by SiO2 nanoparticles. Int J Adhes Adhes 27: 429-433.
- Huaxiang Lai, Xiuzhen Lu, Si Chen, Chune Fu, Johan Liu, et al. (2010) A novel isotropic conductive adhesive with Ag flakes, BN and SiC nanoparticles. Int Symp Adv Packag Mater Microtech APM: 49-53.
- Ariaee S, Tutunchi A, Kianvash A (2014) Modeling and optimization of mechanical behavior of bonded composite-steel single lap joints by response surface methodology. Int J Adhes Adhes 54: 30-39.
- Tutunchi A, Kamali R, Kianvash A (2014) Adhesive strength of steel- epoxy composite joints bonded with structural acrylic adhesives filled with silica nanoparticles. J Adhes Sci Technol 29: 195-206.
- S Bhowmik, R Benedictus, JA Poulis, HW Bonin, VT Buib, et al. (2009) High-performance nanoadhesive bonding of titanium for aerospace and space applications. Int J Adhes Adhes 29: 259-267.
- Bogue Robert (2011) Recent developments in adhesive technology: a review. Assem Autom 31: 207-211.
- Hu Chun Xiu, Zhao YN, Hou HB, Xia HZ (2015) 1200 Mesh Silicon Carbide Corona Protection Varnish with Epoxy/OMMT NanoComposite Adhesive. Adv Mater Res 1118: 166-169.
- Barkoula NM, Alcock B, Cabrera NO, T Peijs (2008) Fatigue properties of highly oriented polypropylene tapes and all-polypropylene composites. Polym Compos 16:101-113.
- Masson F, Decker C, Jaworek T, R Schwalm (2000) UV-radiation curing of water based urethane - acrylate coatings. Prog Org Coatings 39: 115126.
- Dispersions A Study AC Scale Curing 2000:72.
- Sow C, Riedl B, Blanchet P (2011) UV-waterborne polyurethane- acrylate nanocomposite coatings containing alumina and silica nanoparticles for wood: mechanical, optical, and thermal properties assessment. J Coatings Technol Res 8: 211-221.
- Belli R, Kreppel S, Petschelt A (2014) Strengthening of dental adhesives via particle reinforcement. J Mech Behav Biomed Mater 37: 100-108.
- Rose S, Prevoteau A, Elziere P, Dominique H, Alba Marcellen, et al. (2014) Nanoparticle solutions as adhesives for gels and biological tissue. Nature 505: 382-385.
- Pang B, Ryu C M, Kim H I (2013) Improvement of thermal stability of UV curable pressure sensitive adhesive by surface modified silica nanoparticles. Mater Sci Eng 178: 1212-1218.
- Zhenjiong Wang, Zhengbiao Gu, Yan Hong, Li Cheng, Zhaofeng Li, et al.(2011) Bonding strength and water resistance of starch-based wood adhesive improved by silica nanoparticles. Carbohydr Polym 86: 72-76.






























