Radiopaque Contrast Agents Based on Bulk and Nanosized La1 Gd TaO4 and Gd2O3
Mikhail G Zuev1*, Vladislav G Ilves2, Sergey Yu Sokovnin2,3 and Elena Yu Zhuravleva2
1Institute of Solid State Chemistry, Ural Branch, Russia
2Institute of Electro physics, Ural Branch, Russia
3Ural Federal University, Russia
Submission: October 02, 2017; Published: October 17, 2017
*Corresponding author: Mikhail G Zuev, Institute of Solid State Chemistry, Ural Branch, Russia; Email: zuev@ihim.uran.ru
How to cite this article: Mikhail G Z, Vladislav G I, Sergey Y S, Elena Y Z. Radiopaque Contrast Agents Based on Bulk and NanosizedLa1-xGdxTaO4 and Gd2O3. Organic & Medicinal Chem IJ. 2017; 4(2): 555632. DOI: 10.19080/OMCIJ.2017.04.555632.
Abstract
In vitro tests of bulk- and nanosized radiopaque contrast agents based on solid solutions of La1xGdxTaO4 and Gd2O3 are presented. Methods for obtaining radiopaque contrast agents are described.
Keywords: Bulk; Nanosized; Radiopaque; Contrast Agents; RE Tantalates; Gadolinium Oxide
Abbreviations: RCA: Radiopaque Contrast Agents; NP: Nano Powder; PTC: Physical and Technical Conditions
Introduction
Currently, iodine-containing RCA have been extensively used for contrast studies. However, these agents have a number of drawbacks. They produce a toxic effect on blood, liver, kidneys, pancreas, the central nervous system, and the endocrine system (especially on the thyroid gland). They have a local irritant action on mucous tunics, including epithelium of tracheobronchial tree, hepatic and pancreatic ducts, endothelium of arterial, venous, and lymphatic vessels and heart, as well as arachnoids membranes. Besides, they exert various allergic reactions, anaphylactic shock included. Therefore, the search for new effective RCA without such drawback is being well under way. In this paper we describe new RCA in the form of liquid gels, which are suspensions based on bulk- and nano solid solutions La1-x Gdx TaO4 and Gd2 O3Tantalates La1-xGdxTaO4 has different K-absorption jumps of La, Gd and Ta. Therefore, using the substances that contain these chemical elements, it is possible to vary the radiation absorption and to change smoothly the image contrast [1,2].
Materials and Methods
Nanopowders (NPs) LaTaO4 and Gd2O3 were obtained using radiation technologies from targets made from bulk samples. The bulk substances La1-xGdxTaO4 (x = 0 - 0.18) were synthesized from the corresponding oxides in the solid phase. As raw materials used reagent Ta2O5, La2O3 and Gd2O3 with the maintenance of the basic substance not less than 99.99 % the blend was annealed at temperature 1450 to 1500 °C during 40 h with intermediate recrushing of blend. To synthesize La1-xGdxTaO4 samples used LaTaO4 and GdTaO4 compounds taken in the relations corresponding the stekhiometricheskikh. X-ray diffraction analysis was performed using a DRON-2.0 diffractometer (CuKα radiation) and ICDD database. The NPs were produced by evaporation of ceramic targets in air (residual pressure 4 Pa) using pulsed electron beam [3]. The targets - circular disks 20-30 mm in diameter, to 20 mm in height, made on a hand press from phosphor powders - were produced by annealing of disks at 1400 °C for 40 h. The target was fixed on a graphite substrate placed in a stainless-steel sample holder to eliminate possible contamination of NP in case of deep melting of the target or if the beam hits the surface of the metal holder. The electron energy was 40 keV, the electron beam pulse energy was 1.8 J, the pulse duration was 100 μs, and the pulse frequency was 100-200 Hz. The target evaporation time was 40-60 min. The target rotation rate was 8.3 rpm. The powders were deposited on large 4-mm-thick non-cooled glass substrates placed around the target. The distance between the target and the substrate was not less than 10-15 cm.
The obtained NPs of LaTaO4 and Gd2O3 are colored greyish powders with an average particle size of ~ 5 nm.The specific surface of the NPs was determined by the Brunauer-Emmett- Teller (BET) method on a Micromeritics TriStar 3000 facility. The microscopic analysis of NP was performed on a JEOL JEM 2100 transmission electron microscope. The contrast properties of the bulk- and NPs were estimated in vitro in comparison with urografin. The gel suspension was prepared with concentrations 10 % (La1-xGdxTaO4) The compared RCA were placed into 10 ml glass vials. RCA contains tantalate NPs, natural polysaccharide and water The main problem which managed to be solved consists in possibility of use as the agent for contrasting tantalates rare- earth elements in the form of nanoparticles with the average size of 5 nanometers in a wide energy interval of the x-ray radiation, covering all range of energy of x-ray radiation for medical x-ray diagnostics. There were experimentally established interaction conditions of tantalates nanoparticles and the liquid dispersive environment in the form of natural polysaccharide.
High sedimentative stability of means is apparently the consequence of generation on the surface of the adsorptive layer particles owing to sedimentation molecules of the natural polysaccharide which existence complicates possibility of particles adhesion and consequently also formations of agregates. The gel suspension of Gd2O3 was prepared with concentrations 0.75 %. For the preparation of PKC in gel form, the aqueous solution of the sodium salt of carboxymethylcellulose. X-ray studies were performed on an X-ray diagnostic digital complex APELEM BACCARA 90/20 under the following physical and technical conditions for LaTaO4 and Gd2O3: focal distance 50 cm, cassette size 18x24 cm. A RUM-20M X-ray unit was also used. The exposed X-ray film was treated using the usual technology
Results and Discussion
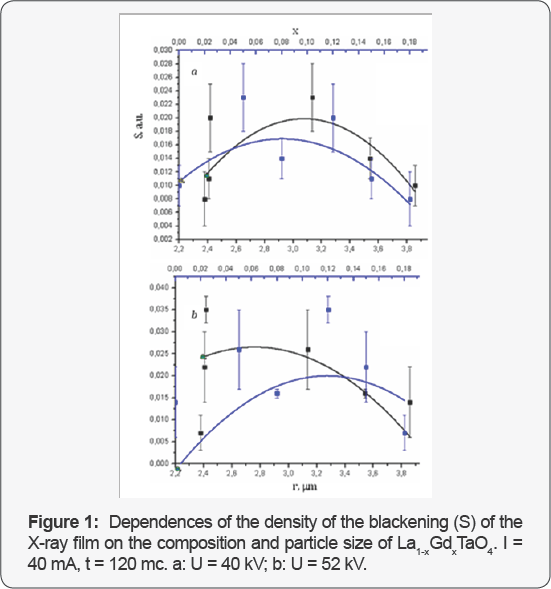
Figures 1 & 2 demonstrates the dependence density of the blackening (S) of the X-ray film with these compounds. Characteristic dependences between the average density ofthe blackening of images on the X-ray film of vials with the gel suspensions of the examined substances and the composition x and the average particle size r are presented in Figure 3. The vials were irradiated with X-ray quanta under various physical and technical conditions; the r(x) dependence is also given.
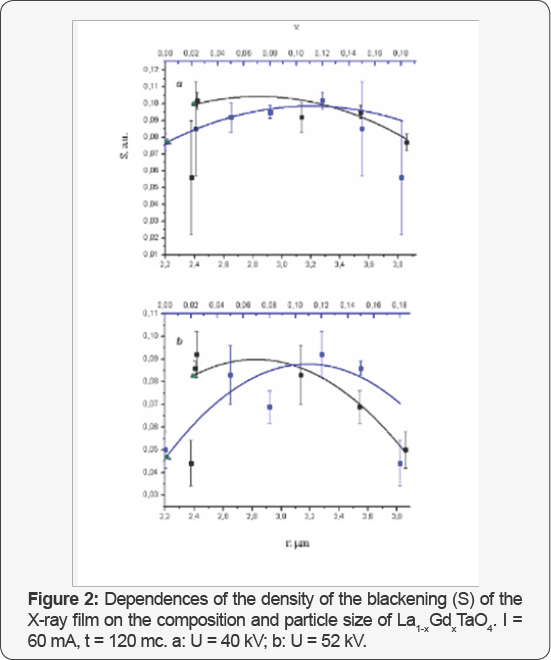

The least S value was observed for the solid solution La0.82Gd018TaO4. The physical and technical conditions also affect the S value. If the voltage on the X-ray tube increases from 40 to 52 kV, the opacity value of the X-ray film for this substance lowers. Therefore, this substance provides the highest X-ray contrast in the series of solid solutions. From the Figures 1 &2it is well seen that by changing the quantitative composition of RCA containing the same chemical elements it is possible to change the radiation attenuation and, consequently, to alter the contrast of the substances, which is very important for designing of new RCA. Thus, the specific efficiency of the substances can be continuously changed by varying smoothly their composition and the average particle size. From this Figures 1 & 2 it can be also concluded that the X-ray contrast of the substances can be controlled not only by variation of their composition, but also by alteration of the physical and chemical conditions of filming.

In Table 1 are given to the degree of blackening of X-ray film after irradiation of vials with bulk and NP based on LaTaO4 (5%). It is seen that the density of the blackening of films obtained by irradiating an NP gel based on LaTaO4 is lower than for bulk LaTaO4. This confirms the higher NP contrast on the basis of LaTaO4 than bulk samples. Perhaps this is due to the aggregation of nanoparticles. In Figure 3 shows the dependence of the average density of the S images on an X-ray film of vials with gel suspensions containing NP Gd2O3with concentrations of 2.5, 5, 12.5 % and urographine. The vials were irradiated with X-ray quanta under various physical and technical conditions (PTC). It is seen that the values of S for urographine and NP Gd2O3 with a concentration of 12.5 % are especially close at PTC U = 45 kV, I = 25 mA, t = 160 mc. It is known that a shorter film exposure time corresponds to a shorter exposure time.
Conclusion
The gel suspensions based on the bulk solid solutions La1- xGdxTaO4 (x = 0 - 0.18) allow of continuous and smooth alteration of radiopaque contrast agents thus expanding the fields of their application. Gel substances based on NPs LaTaO4 and Gd2O3 have a higher contrast than bulk substance.
Acknowledgement
The authors are grateful to Dr. I. M. Strekalov and A. A.Shchelkanov for X-ray photography
References
- Zuev MG, Larionov LP, Strekalov IM (2007) Psychopharmacology and Biological Narcology. III Congress of Pharmacology Russia. Saint-Petersburg 23-27.
- Zhuravleva E Yu (2004) Tantalum X-ray Contrast Media (Solid-State Chemistry for Medicine Series), by MG Zuev, LP Larionov, Yekaterinburg: Ural Otd Ross Akad Nauk. Inorganic Materials 40(6): 671-672.
- Sokovnin S Yu, Il'ves VG, Zuev MG (2016) Production of complex metal oxide nanopouders using pulsed electron beam in low-pressure gas for biomaieriats application. In Engineering of Nanobiomaterials Applications of Nanobiomaterials. Edited by Alexandru Grumezescu, Elsevier, Oxford, USA, pp. 29-75.






























