Effect of Concentration and pH on the Size of Silver Nanoparticles Synthesized by Green Chemistry
Lakkappa B Anigol1, Jasmith S Charantimath1,2 and Prabhuodeyara M Gurubasavaraj1*
1Department of Chemistry, Rani Channamma University, Vidyasangama, Belagavi-591156, India
2Department of Chemistry, S. G. Balekundri Institute of Technology, Belagavi-590010, India
Submission: September 09, 2017; Published: September 19, 2017
*Corresponding author: Prabhuodeyara M Gurubasavaraj, Department of Chemistry, Rani Channamma University, Vidyasangama, PBNH-4, Belagavi-591156, India, Email: pmg@rcub.ac.in
How to cite this article: Lakkappa B A, Jasmith S C, Prabhuodeyara M G. Effect of Concentration and pH on the Size of Silver Nanoparticles Synthesized by Green Chemistry. Organic & Medicinal Chem IJ. 2017; 3(5): 555622. DOI: 10.19080/OMCIJ.2017.03.555622.
Introduction
Nanotechnology is versatile subject, which deals with biology, chemistry, physics and engineering. The term “Nano” is derived from Greek word which means dwarf and size of particle varies from 1-1QQnm [1]. Nanotechnology is the synthesis of nanoparticles which exhibit different sizes, shapes and morphology. Nanoparticles being very small in size posses large surface area to volume ratio due to which nanoparticles exhibit very different properties such as electrical , magnetic and optical properties than its bulk material [2]. In nanoparticle synthesis it is very important to control not only the particle size but also the particle shape and morphology as well. Therefore, controlling the shape and structure of nanoparticles seems to be the most attractive fields of current research.
Nanoparticles and nano devices contain potential to reduce pollutants such as water and air pollutants which can be used for efficient alternate energy production. Nanoparticles show activity against bacteria, fungus as well as Virus. Various chemical and physical methods are known for preparation of silver and other metal nanoparticles. These methods are very costly and toxic to the environment [3]. The use of plant parts like seed, fruit, bark, stem, leaf etc. for the synthesis of nanoparticles is quite novel method compared to the chemical and physical method [4]. This is cost effective and easily scaled up process. Photo chemicals present in the plant posses anti-oxidant or reducing properties which are responsible for reduction of metal compounds. Methods used for the synthesis of nanoparticles are eco-friendly, bio-compatible, non-toxic and clean [5].
During recent times several groups have achieved success in the synthesis of Ag, Au and Pd nanoparticles using extracts obtained from unicellular organisms like bacteria [6-9] and fungi [1Q-12] as well as extracts from plant parts e.g. geranium leaves [13], lemon grass [14], neem leaves [15], aloevera [16] and several others [17-21]. The spectacular success in this field has opened up the prospect of developing bio-inspired methods of synthesis of metal nanoparticles with tailor-made structural properties. Among the various bioreductants, coriandrum sativum leaves extract was chosen for the present study since they have minerals and vitamin contents including calcium, phosphorus, iron, carotene, thiamine, riboflavin and niacin. They also contain sodium and oxalic acid.
Techniques using citrate reduction or reduction by plant extracts are all termed as green synthesis methods. To distinguish from others we refer to our method as biosynthesis. Silver nanoparticles have an advantage over the metal nanoparticles (e.g. gold and copper) because the surface Plasmon resonance energy of Ag is located far from the interband transition energy The Ag nanoparticles-contained composites are investigated for the non-linear optical effects solely based on the surface Plasmon contribution. In situ growth of metal nanoparticles in polymer films and their nonlinear properties have been reported by group [22]. The advantage of this method is the use of aqueous medium for the fabrication process, employment of the polymer itself as the reducing agent, mild thermal annealing for generating the metal and in situ generation of the nanoparticles inside the polymer matrix which serves as the stabilizer as well.
Different types of nanoparticles like Copper, Iron, Titanium, Zinc, Gold, Magnesium and Silver have been come up but silver nanoparticles have proved to be the most effective as compare to other metal nanoparticles because the surface Plasmon resonance is located far from the inter transition energy. On the vast of available literature on this topic, we hypothesize inherently Capparis Moonii which is rich in photochemical and could form a basis for the synthesis of silver nanoparticles [9-15]. Capparis Moonii is a medically important plant which belongs to Capparis a flowering plant genus in the family. The whole plant contains a number of medicinally important compounds and it is an excellent analgesic used in the treatment of asthma, Bronchits, Dyspesia, Flatulene etc. In this paper we report, for the biosynthesis of the Ag nanoparticles using Capparius Moonii fruit extract as reducing agent. The nanoparticles are characterized by UV-Visible spectroscopy, XRD, IR, and TEM analysis. The usefulness of these biologically synthesized nanoparticles as antimicrobial agents are also discussed.
Preparation of Fruit Extracts
Preparation of Fruit Extracts
2gm of Rudanti fruit was taken and was crushed with help of mortar and pestle. And the crushed fruit was taken in a 250 ml beaker containing 100mL distilled water. It was kept in a microwave oven for about 30 minute. Prepared fruit extract was filtered with the help of Whatman filter paper no 41 and collected in a beaker.
Preparation of silver nanoparticles (AgNPs)
The fruit extract (50ml) was added to 250ml of 1mM of AgNO3 solution and kept in microwave oven. After 3 minutes added distilled water to maintain a constant volume of 300ml. This process continues up to 30 minutes. After heating color changes to dark brown this indicates the formation of silver nanoparticles [23] The bioreduced silver nanoparticles were collected and UV- Visible of the solution was measured. The nanoparticles were diluted to avoid errors due to high optical density.
Results and Discussions
UV-Vis Spectra Analysis
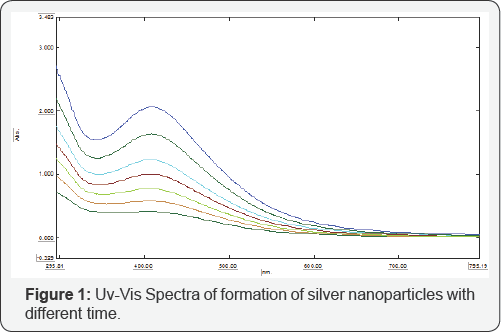
UV-visible spectroscopy analysis was carried out on a SHIMADZU-1800 visible absorption spectrometer with a resolution of 0.5nm between 190nm to 800nm. Equivalent amounts of the suspension (1ml) were diluted in a constant volume of de-ionized water (10 ml) and subsequently analyzed at room temperature. The progress of the reaction between metal ions and the leaf extract were monitored by UV-visible spectra of AgNP'S in aqueous solution with different reaction times as shown in (Figure 1). It was observed that the peak centered at 413nm at 5, 6, 7 minute samples on irradiating with microwaves. The reduction of silver ions and for the formation of stable nanoparticles occurred rapidly within an hour of reaction [24] (Figure 1).
Effect of Concentration of AgNO3
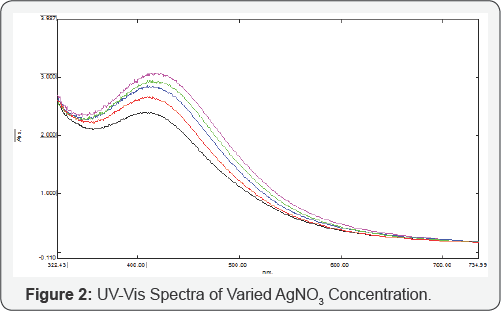
The UV-Vis spectrum shows the effect of silver nitrate concentration on the formation of silver nanoparticles using the fruit extract of Capparis Moonii. Characteristic surface Plasmon absorption band was absorbed at 430nm. Absorption spectra at different values of the concentration (1mM, 2mM, 3mM, 4mM, and 5mM) are presented in (Figure 2). A general trend is that the surface plasmon resonance peak shift towards the higher wavelength region as well as becomes narrower when the concentration value increases. Assuming that the shift in the plasmon resonance peak indicates a change in the size of AgNPs and hence any shift of the peak towards the higher wavelength is accompanied by a decrease in the size of the prepared AgNPs, we conclude that the raising concentration of the solution results in the formation of nanoparticles with smaller size [25-26]. On the other hand, the broadening of the surface plasmon resonance peak indicates the existence of the wider range of size in the solution (Figure 3).
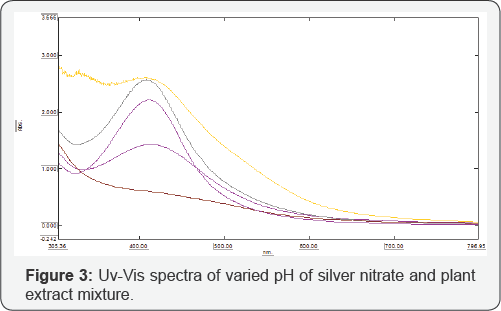
Effect of pH
At the alkaline pH range, the stability of cluster distribution and colloid formation is increased with a declined tendency for aggregation of the particles due to complete charging of the clusters which maximize the repulsive electrostatic/ electrosteric interactions. At elevated, monodispered and small spherical nanoparticles are formed as well as the amount of AgNPs was also higher. A possible reason for this result was that during the elevated pH, the reaction rate will be increased with subsequent crystallization into smaller particles, which involved the nucleation and growth processes of smaller particles from Ag nuclei. There are many factors that affect the size and shape of the silver nanoparticle. In this part we will address the impact of the pH on the size of AgNPs by monitoring the size variation as a result of changing the pH of solution. Absorption spectra at different value of the pH (3, 5, 7, 9, and 11) are presented in (Figures 4 & 5). In the acidic condition the peak becomes broader and the size of particle increases. With increase in pH the absorption increases and gives the narrow peak at pH 9 with uniform distribution of size. Hence we presume that the basic condition is favored for controlling the particle size. The pH is inversely proportional to size of AgNPs (Figure 3).

FTIR Spectra Analysis of AgNPs

FTIR spectroscopy (Figure 6) is performed to get the idea regarding various functional groups and their interactions with silver, which may be accountable for fabrication and stabilization of silver nanoparticles. Represents the absorption bands. The absorption range 346Q-35QQcm-1 indicates the presence of a carboxylic O-H group and 2933cm-1 range indicates the presence of Aldehyde C-H group present and 1746cm-1which represents the Carboxylic acid and C-O stretching in the organic moiety. And1632cm-1 which assigns an amide group present and characteristic of C-O stretching is observed. The bands 123Qcm- 1 represents the characteristics of multiple CO (Figure 4).
Particle Size Analysis of AgNPs
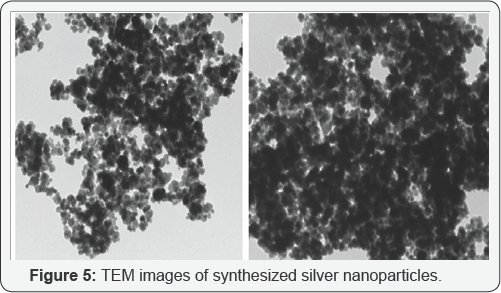
Particle size analysis at various concentration and pH shows different particle size. The sizes of nanoparticles are in the range of 540nm to 16nm (figure 5) and (figure 6).
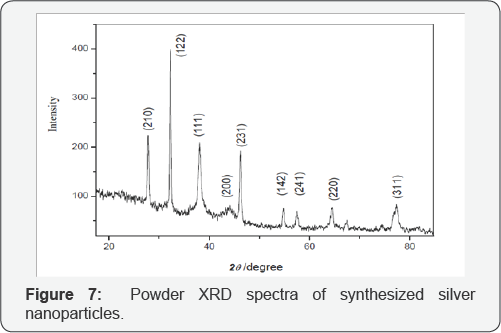
3.5.1.X-ray Diffraction Study: X-ray diffraction pattern of the synthesized silver nanoparticles is shown below (Figure 7). A number of Bragg reflection is observed at 20 values 27.81° 32.16° 38.12°, 44.3°, 46.21°, 54.83°, 57.39°, 64.42° and 77.45° which corresponds to (21Q), (122), (111), (2QQ), (231), (142), (241), (22Q) and (311) planes of pure silver based on the face-centered cubic structure (JCPDS file No. 04-0783). From XRD results, it is clearly observed that the silver nanoparticles synthesized by the plant extract are crystalline in nature.
Biological Activity Studies
Bacterial growth was reduced with an increase in AgNP concentration. Further increasing concentration of AgNP caused absence of bacterial growth. Their antibacterial activity can not solely be explained on the basis of size, since the shape of silver nanoparticles may have significant effect. The factors that influence bacterial activity are initial bacterial concentration, microbial strains and composition of culture media. More over the size, shape, crystallinity, surface chemistry and capping reagent of silver nanoparticles is likely to play an actual role and may cause variation in the anti-bacterial effect. Results showed that the anti-bacterial potential of AgNPs was greatly enhanced as their size was reduced.
Anti-bacterial activity
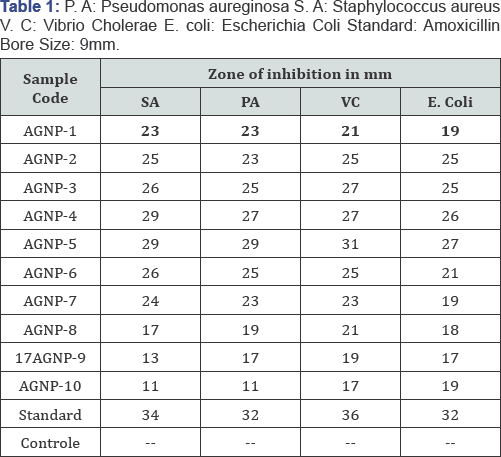
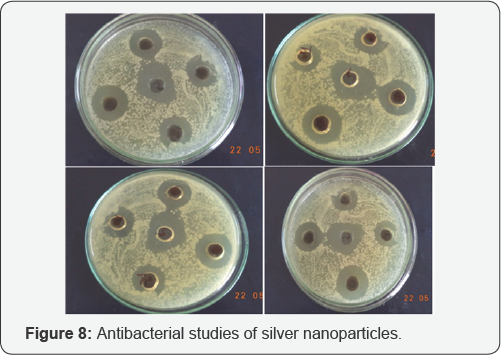
The result obtained is shown below in the table. The inhibition zones obtained indicates maximum anti-bacterial activity of the prepared test sample of silver nanoparticles at different concentration and pH. As the concentration of AgNPs increases anti-bacterial activity increases with the decrease in particle size. On the other hand the pH of AgNPs increases with decreasing anti-bacterial activity. The below table shows antibacterial activity of silver nanoparticles and is synergistic activity against P.A (Pseudomonas aurginosa) S.A (Staphylococcus aureus) V.C (Vibrio cholera) and E. Coli (Escherichia Coli), these are compared with standard values of Amoxicillin. In this S.A of AgNp-5 shows a higher anti-bacterial activity at 5mM concentration and shows a lower activity at AgNp-10,and P.A of AgNp-5 shows a higher ant-bacterial and lower antibacterial activity at AgNp-10, P.A of AgNp-5 have anti-bacterial activity near to standard value of Amoxicillin and it is favorable. Similarly a V.C shows a higher anti-bacterial activity at AgNp-5 it is near to standard value and low anti-bacterial activity at AgNp-10. Similarly E. coli has higher Antibacterial activity at AgNp-5 and low at AgNp-10(Higher pH) (Table 1) and (Figure 8).
Antifungal activity
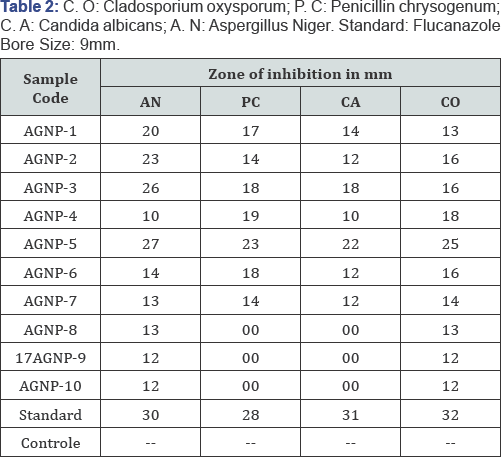
In the antifungal activity as the concentration of AgNPs increases with the increase in anti-fungal activity On the other hand the pH increases with the decrease in the anti-fungal activity or negligible. The AgNPs have higher anti-fungal activity and maintains pH in the acidic medium. Anti-fungal activity of silver nanoparticles and its synergistic activity against A.N (asperigillus niger), P.C (Penicillin chrysogeum), C.A (Candida albicans) and C.O (Cladosporium oxysporum), these are having standard values and comparable with above anti-bacterial activity of AgNPs having different concentration and different pH. In this table A.N of antifungal activity of AgNp-5(higher concentration) shows highest anti-fungal activity and low antifungal activity at AgNp-10(higher pH).And in case of P.C shows their anti-fungal activity at AgNp-5 and negligible or zero antifungal activity at AgNP-8, AgNp-9, AgNp-10. Similarly in C.A AgNp-5 having higher antifungal activity and low or negligible activity at AgNp-8, AgNp-9, AgNp-10. And finally C.O having higher anti-fungal activity at AgNp-5 and low at AgNp-9, AgNp- 10 (Table 2).
Conclusion
We have successfully synthesized silver nanoparticles using capparis moonii fruit extract. The nanoparticles were characterized by UV, XRD, IR, and Particle analyzer techniques. The size of the particles was varied depending upon the pH and Silver nitrate concentration. The synthesized silver nanoparticles were screened for antibacterial and antifungal activities. All samples showed both antifungal and antibacterial activities. The activity was size dependent and showed highest activity to lower sized nanoparticles which is in agreement with the literature. We are currently exposing the anticancer activity of the synthesized nanoparticles.
References
- Sing C, Sharma V, Naik P, KhandewalV, Singh H, et al. (2011) A green biogenic approach for synthesis of gold and silver nanoparticles using Zingiber officinale. Digest J Nanomat Biostruct 6(2): 535-542.
- Kim J ,Yu K ,Park S, Lee H, Kim S, et al. (2007) Antimicrobial effects of silver nanoparticles. Nano med 3(1): 95-1Q1.
- Kalaiarasi R, PrasannarajaG, Venkatachalama P (2013) A rapid biological synthesis of silver nanoparticles using leaf broth of Rauvolfia tetraphylla and their promising antibacterial activity Indo Am J pharm Res 3(1Q): 8Q52-8Q62.
- Y Sun, Y Xia (2002) Shape-controlled synthesis of gold and silver nanoparticles. Science 298(56Q1): 2176-2179.
- Sharma V, Yangard R (2009) Silver nanoparticles: green synthesis and their antimicrobial activities. J Colloid Interface Sci 145(1-2): 83-96.
- Ahamad A, Senapati S, MI Khan, R Kumar, M Shastri, et al. (2003) Extracellular Biosynthesis of Monodisperse Gold Nanoparticles by a Novel Extremophilic Actinomycete, Thermomonospora sp. Langmuir 19: 355Q.
- B Nair, T Pradeep (2002) Coalescence of Nanoclusters and Formation of Submicron Crystallites Assisted by Lactobacillus Strains. Crys Growth Des 2(4): 293-298.
- TK Joerger, R Joerger, E Olsson, CG Granqvist (2001) Bacteria as workers in the living factory: metal-accumulating bacteria and their potential for materials science. Trends Biotechnol 19(1): 15-20.
- AR Shahverdi, Seminarian, HR Shahverdi, H Jamalifar, AA Nohi, et al. (2007) Rapid synthesis of silver nanoparticles using culture supernatants of Enterobacteria: A novel biological approach. Proc Biochem 42: 919.
- P Mukherjee, A Ahmad, D Mandal, S Senapati, S R Sainkar, et al. (2011) Alkene Hydrogenations by Soluble Iron Nanocluster Catalysts. Angew Chem Int Ed Engl 56(13): 3585-3589.
- A Ahmad, P Mukharjee, S Senapati, D Mandal, MI Khan, et al. (2003) Extracellular biosynthesis of silver nanoparticles using the fungus Fusarium oxysporum. Colloids Surf B Bionterf 28(4): 313-318.
- A Ahamad, P Mukharjee, S Senapati, D Mandal, MI Khan, et al. (2002) Extracellular synthesis of gold nanoparticles by the fungus Fusarium oxysporum. Chem Biochem 3(5): 461-463.
- S Shivshankar, A Ahamad, R Pasricha, M Sastry, J Matter, et al. (2003) Bioreduction of chloroaurate ions by geranium leaves and its endophytic fungus yields gold nanoparticles of different shapes. Chem 13: 1822.
- S Shivsshankar, A Rai, A Ahamad, M Sastry, J Colloidinterf, et al. (2004) Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. Sci 275 (2): 496-5Q2.
- S Shivshankar, A Rai, A Ahamad, M Sastry (2005) Controlling the Optical Properties of Lemongrass Extract Synthesized Gold Nanotriangles and Potential Application in Infrared-Absorbing Optical Coatings. Chem mater 17 (3): 566-572.
- SP Chandran, M Choudhary, R Pasricha, A Ahamad, M Sastry, et al. (2006) Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol Prog 22(2): 577-583.
- NA Begum, S Mandal, S Basu, RA Laskar, D Mandal, et al. (2009) Biogenic synthesis of Au and Ag nanoparticles using aqueous solutions of Black Tea leaf extracts. Colloids and Surface B: Biointerfaces 71(1): 113-118.
- D Phillip, Specrochemica (2009) Biosynthesis of Au, Ag and Au-Ag nanoparticles using edible mushroom extract. Acta part A 73(2): 374381.
- K Badri Narayanan, N Saktivel (2008) Coriander leaf mediated biosynthesis of gold nanoparticles. Mat Lett 62: 4588.
- D Phillip (2009) Biosynthesis of Au, Ag and Au-Ag nanoparticles using edible mushroom extract. Spectrochem Acta A: Mol biomol Spectrosc in press 73(2): 374-81.
- J Haung, Q Li, D Sun, Y Lu, Y su, et al. (2007) Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology 18: 1Q51Q4.
- S porel, N Venktram, D NarayanRao, TP Radhakrishanam (2007) Journal of Nanoscience and Nanotechnology 7:1.
- Gupta A, bonde S, Gaikwad S, Ingle A, Gade a, et al. (2014) Lawsonia inermis-mediated synthesis of silver nanoparticles: activity against human pathogenic fungi and bacteria with special reference to formulation of an antimicrobial nanogel. IET Nanobiotechnol 8(3): 172-178.
- Gaikwad S, ingle A, Gade A, Maracato P, Rai M, et al. (2013) A New Generation of Chemist Leaders for a Chemistry without Borders. J Brazchemsoc 24(12): 1974-1982.
- RS Patil, MR kokate, CL jambale, SM Pawar, SH Han, et al. (2012) One- pot synthesis of PVA-capped silver nanoparticles their characterization and biomedical application. Adv Nat sci Nano sci Nanotechnol 3: 7.
- H Bar, DH Bhui, PG Sahoo, P Sarakar, PS De, et al. (2009) Green synthesis of silver nanoparticles using latex of Jatropha curcas. Colloids Surf A Physicochem Eng Asp 339: 134-139.






























