Chemistry and Biological Potential of Pyrimidine Derivatives: Study of Nucleosides with Pyrimidines
*Mohammad Asif
Department of pharmacy, GRD (PG) Institute of Management and Technology, India
Submission: May 25, 2017; Published: June 30, 2017
*Corresponding author: Mohammad Asif, Department of pharmacy, GRD (PG) Institute of Management and Technology, Dehradun, 248009, India
How to cite this article: Mohammad A. Chemistry and Biological Potential of Pyrimidine Derivatives: Study of Nucleosides with Pyrimidines. Organic & Medicinal Chem IJ. 2017; 2(5): 555598. DOI: 10.19080/OMCIJ.2017.02.555598
Abstract
Much attention has been devoted to the preparation of nucleosides with pyrimidinic bases and their derivatives, especially uracils. Uracils are an important heterocycles because of their applications as bioactive compounds like 5-iodouridine, 2(S)-will-ardiine, zidovudine and tifluridine. This scaffold is of prime importance to chemists and biologists to yield diverse type of new molecules and screen these new entities for pharmacological activities. These synthesized molecules exhibited a variety of activities like antitumor, tuberculostatic, anti-diarrhea], anticonvulsants, antibacterial, antimicrobial, tyrosine kinase inhibitor, calcium channel antagonists, antileishmanial, diuretic, anti-inflammatory and analgesic etc. These observed activities systematic molecular manipulations have yielded several types of clinically used drugs like anticancer drug, nucleoside based anti-AIDs drugs, CNS acting drugs.
Keywords: Pyrimidines; Uracils; Drugs; Biological activities
Introduction
Most of commercially available biological active compounds are heterocycles and further N-heterocycles have attracted extensive attention of researchers, as a result of their exciting biological properties. Within nitrogen heterocycles, the preparation [1,2], reactions [3,4] and biological activities [5,6] of pyrimidine (1) containing compounds situate as a expanding area of research in hetero chemistry. Although compounds of this group were known as breakdown products of uric acid, the systematic study of the ring system began with the work of Pinner [7] who first used the name Pyrimidine to the unsubstituted parent unit (1). Pyrimidine or m-diazine is the parent ring system of a variety of compounds which play critical role in biological processes [8]. Various drugs and naturally occurring compounds like vitamins, coenzymes, purines, pterins, nucleotides and nucleic acids.
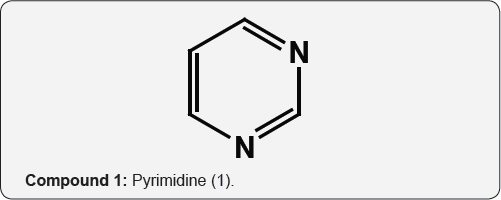
Natural occurrence of pyrimidines
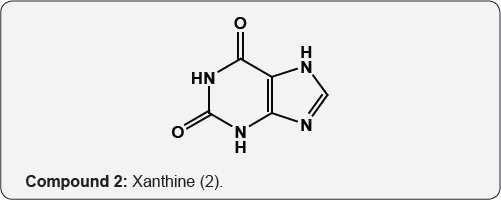
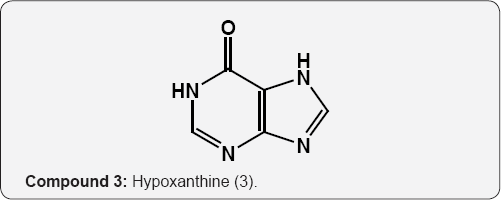
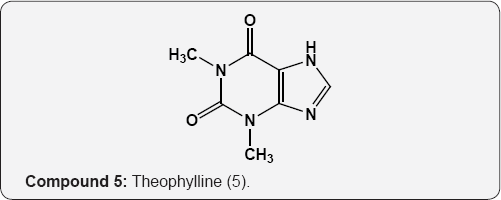
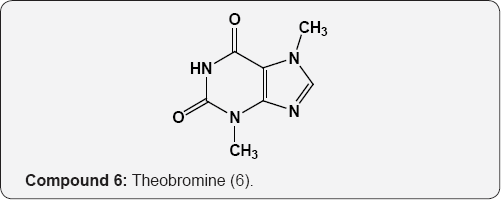
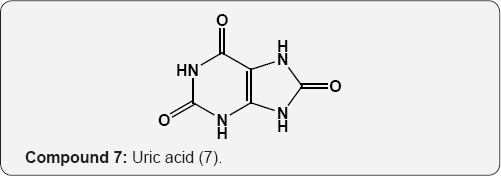
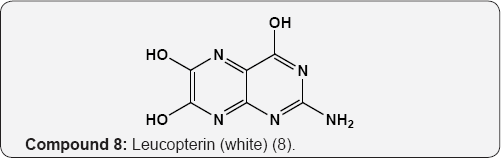
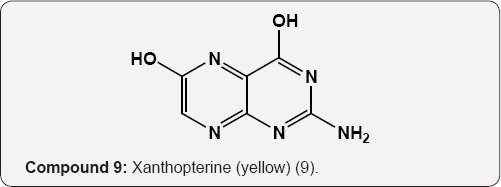
The pyrimidine nucleus has an ample occurrence in nature namely in nucleic acids, nucleotides, alkaloids from tea, coffee, cocoa and in uric acid. The majority of the natural pyrimidines are hydroxyl and amino derivatives. Xanthine (2) was firstly discovered by Marget in 1817 in bladder stone also occurs in tea and in animal tissues along with hypoxanthine (3). The coffee bean and tea leaves contains about 1.5% caffeine (4) and some theophylline (5) [9,10] cocoa bean contains about 1.3% theobromine (6) [11]. These methylated derivatives of xanthines have stimulating effect on the central nervous system (CNS). The uric acid (7) discovered by Swedish chemist Scheele in 1776, is a ingredient of human urinary calculi (pebbles) and is a metabolite of purine nucleoside [12]. Isolated some white (8), yellow (9) and red (10) pigments from the wings of butterflies which were later recognized as pteridine (11) i.e. pyrimido-pyrazine [13,14].


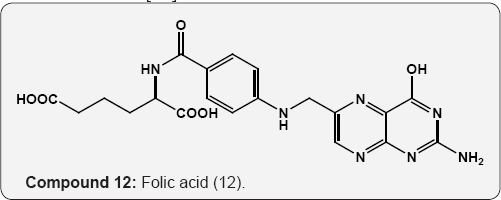
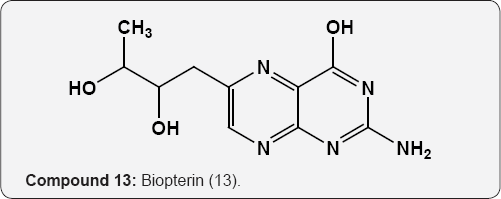
Folic acid 12 (pteroyl glutamic acid, vitamin B10) has been isolated from liver and yeast is an main growth factor for many organisms [15]. Biopterin (13) is a growth factor for the protozoan Crithidia fasciculate [16]. Nucleosides, the pyrimidine and purine glycosides are obtained by hydrolysis of nucleotides and nucleic acids. The hydrolysis of ribonucleic acid (RNA) provided the purine nucleosides guanosine and adenosine together with the pyrimidine nucleoside uridine (14) and cytidine (15). These are β-D-ribofuranosides of guanine, adenine, uracil and cytosine respectively. The hydrolysis of deoxyribonucleic acid (DNA) gives purine and pyrimidine derivatives of deoxyribose (16 & 17) compounds. The phosphoric acid ester of nucleosides are called nucleotides [17].
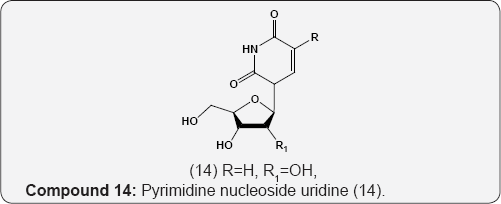

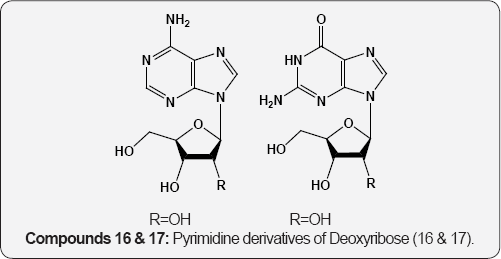
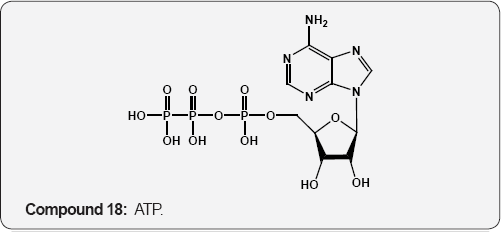

(14) R=H,R,1=OH,Uridine 15 R=OH,Cytidine (16) R= OH,Adenosine (17) R=OH, Guanosine R=CH3, Rj=H, Thymidine R=H, Deoxycytidine R=H, Deoxyadenosine R=H, Deoxyguanosine. The trinucleotide, adenosine triphosphate 18 (ATP) which is obtained from muscle, is involved in various reversible phosphorylations and is the unit of energy in living cells. Flavin- adenine dinucleotide 19 (FAD) was first isolated by Warburg and Christian from D-amino acid oxidase. It is the prosthetic group of flavoprotein enzymes active as oxidation-reduction catalyst in biological systems [18,19].
Riboflavin or vitamin B2 (20) was first isolated in 1933 from whey, is also present in milk, egg and liver [20]. Its absence in the diet of rats leads to weaken growth and rat acrodynia (rat pellagra). The co-enzyme A (21) or the adenine mononucleotide pantothenic acid complex is concerned in trans-acetylation reactions, synthesis and degradation reactions together with metabolism of fatty acids [21].


Pharmacologcal significances of pyrimidines
The significance of uracil and its derivatives have been investigated by biological chemists. The synthesis of naturally occurring complex molecules containing a uracil ring and its substrates continue to be a great interest due to their wide range of biological activities such as antibacterial [22,23], antitumor [24,25] hepatoprotective [26], antihypertensive [26], cardiotonic [26,27], bronchiodilator [28], and vasodilator [29], antialergic [30], antimalarial [31], analgesic [32,33], antifungal [34] activities. Some selected biologically active heterocyclic compounds containing uracil are discussed below:
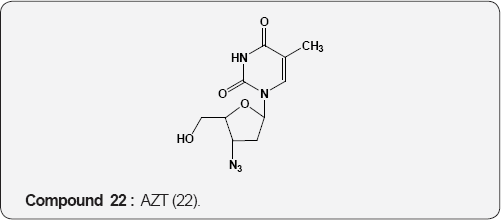
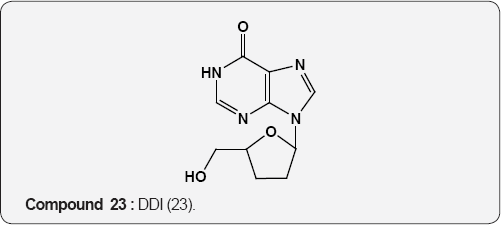
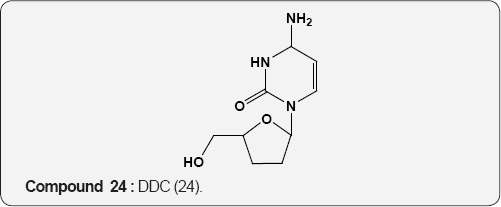
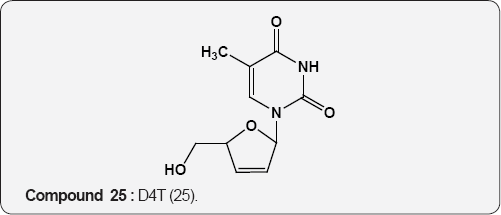
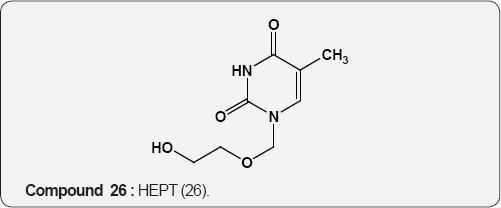
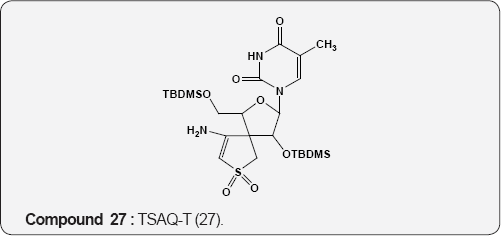
Pyirimidine As Anti-viral: A uracil analogue, 3'-azido-3'- deoxythymidine (AZT) 22 is the first drug to have been used in the treatment of AIDS [35,36]. Other nucleoside analogues of uracil like 2',3'-dideoxyinosine (DDI) 23 [37], 2',3'-dideoxycytidine (DDC) 24 [38], 2',3'-dideoxyhydro-3'-deoxythymidine [39] (D4T) 25 and 1-((2-hydroxyethoxy)methyl)-6-(phenylthio) thymine [40] (HEPT) 26 and TSAQ-T [41] 27 have been established for the treatment of AIDS. All these compounds act as inhibitors of viral reverse transcriptase (RT), an essential enzyme for the replication of human immunodeficiency virus. A series of 1-alkoxy-5-alkyl-6-(arylthio)uracil for anti-HIV-1 activity. Among these 6-((3,5-dimethyl-phenyl)thio)-5-isopropyl-1-propoxy uracil (R=i-Pr, R2=n-Pr, R3= 3,5-Me2) 28 showed the potent and selective anti-HIV-1 activity and also found to be chemically and metabolically stable [42].
<

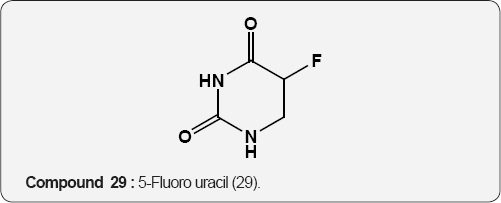
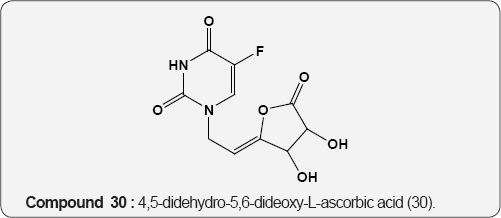
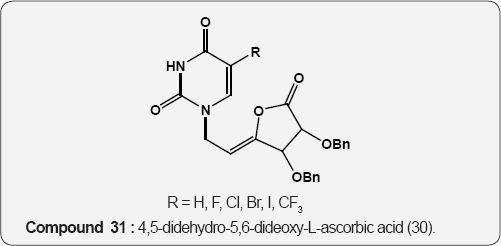
Pyrimidine as Anti-tumor: The 5-Fluoro uracil (29) is a anticancer agent and its derivative, 4,5-didehydro-5,6-dideoxy- L-ascorbic acid (30) act as antitumor activity, mainly against murine leukemia L1210/0 and murine mammary carcinoma FM3A/0 cell lines [43]. Cytostatic activities of pyrimidine derivatives of 5,6-diacetyl-2,3-dibenzyl-L-ascobic acid (31) against malignant cell lines: carcinoma (MCF7), cervical carcinoma (HeLa), Laryngeal carcinoma (Hep 2), murine leukemia (L1210/0), murine mammary carcinoma (FM3A) [44].
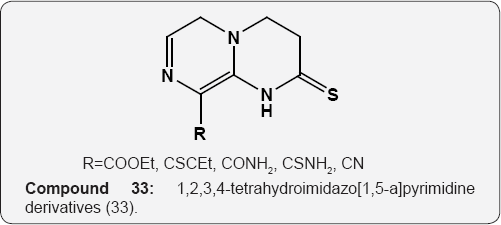
The 1H,3H-pyrido[2,1-f]purine-2,4-dione derivatives of arylpiperzine (32) were tested in vitro for their affinity for 5-HT1a, 5-HT2A, a1 and D2 receptor. These compounds were showed high affinity for 5-HT1A and a1 receptors and moderate to low affinity for 5-HT2A and D2 receptors [45]. Several 1,2,3,4- tetrahydroimidazo[1,5-a]pyrimidine derivatives (33)bearing electron withdrawing substituent, were tested for their activities against mouse leukemia L1210 and human oral epidermoid carcinoma KB cell lines. Among these compounds, 8-thiocarbamoyl-1,2,3,4-tetrahydroimidazo[1,5-a]pyrimidine- 2(1H)-thione (R=CSNH2) tested activity comparable to that of 5-fluoro uracil against both L1210 and KB cells [46].

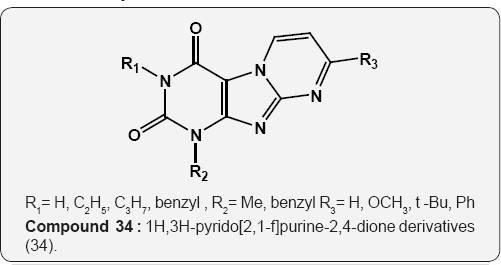
The 1H,3H-pyrido[2,1-f]purine-2,4-dione derivatives (">34), as fused xanthines structures have been tested for their affinities for the human adenisine Aj, A2A and A3 receptors have been tested in radioligand binding studies. Most of the compounds showed moderate antagonist effects at the level of A1 receptors, low or negligible activity at the level of A2A receptors and substantial affinity at the A3 adenisine receptors [47].
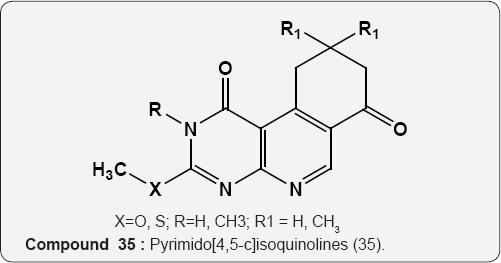
Pyrimidine As Anti-fungal: The pyrimido [4,5-c] isoquinolines (35) were tested in vitro for antifungal properties. The compound (X=S, R=CH3, Rj=CH3) is found to selectively inhibit the E. floccosum, a fungal species [48].
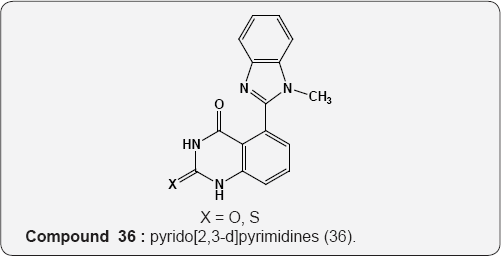
Pyrimidine As Anti-bacterial: The activity of pyrido[2,3-d] pyrimidines (36) for five bacterial species Escherichia coli, Bacillus subtilis, Staphylococcus albus, Neisseria gonorrheae, Enterococcus faecalis and Candida albicans (fungus). The compounds showed moderate activity to these species [49].
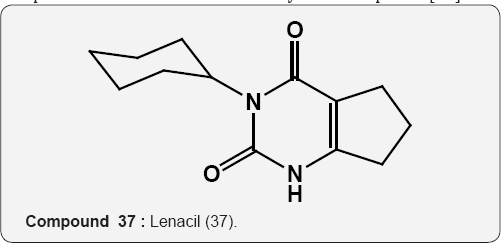
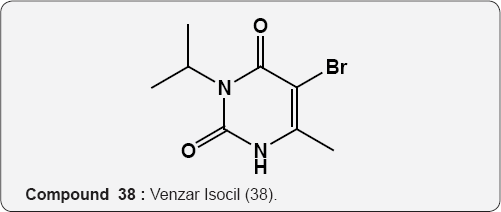
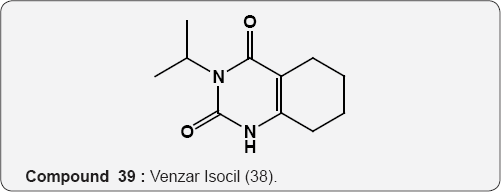
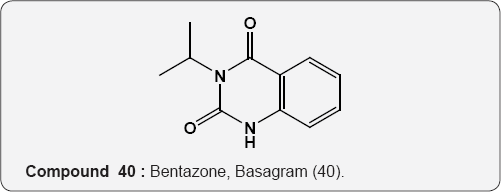
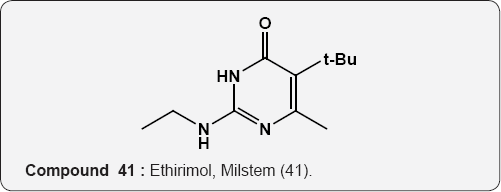
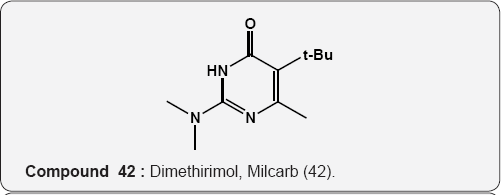
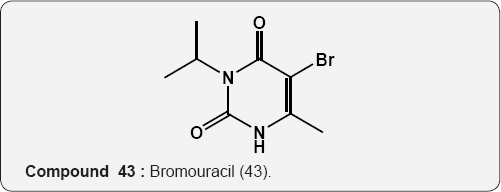
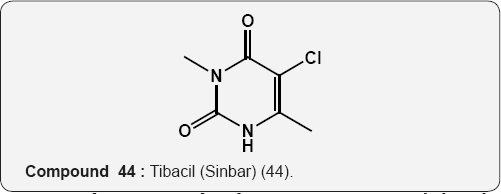
Pyrimidine as Herbicides: Many mono- and bicyclic uracils (37-42) are used to protect plants, mostly as herbicides. Pyrimidine herbicides are generally used for selective control of weeds in certain crops. Bentazone, Basagram (40) Ethirimol, Milstem (41) Dimethirimol, Milcarb (42) Bromouracil (43) is used for the non-selective control of weeds. Substituted uracil herbicides Bromouracil (Hyvarx) used for citrus, pineapple, weed control and soon after that “sinbar” (44) and Venzar” for alfalfa, fruit, mint and sugarbeets [50-54]. Bromouracil (43) Tibacil (Sinbar) (44).
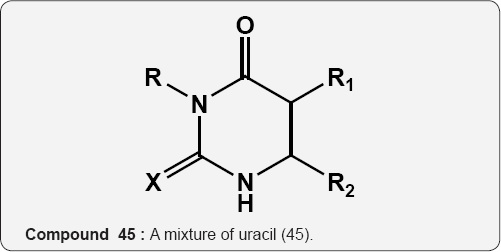
A mixture of uracil (45) and sulfonyl urea (46) acts as herbicides for the protection of orchard such as citrus, apple,sugarcane, grapes and other trees [54]. R=C1-C10 alkyl, C5-C10 aryl, R=Cj-C4 alkyl Substituted Ph, etc. R1=1,3-diazinederivs =Cl, F, Br, I, Me, Et etc. R2=H, R4=H R2=Cl, Br, CrC5, alkyl etc. R5=H, Me X = O and S.
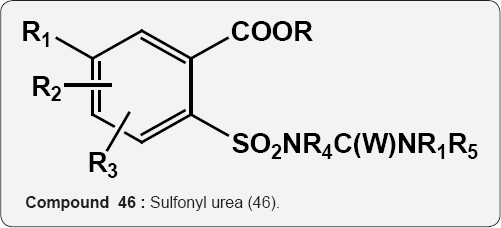
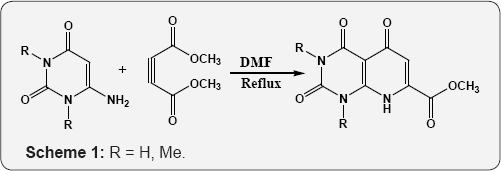
45 46 Pyrimidine (6-amino uracil) chemistry: Uracil is one of the components of nucleic acids in living organisms. Uracil was first isolated from hydrolysis of herring sperm in 1900 [55] and its derivatives such as monomethyl, dimethyl and corresponding thio-analogues were prepared. The enaminone character of 6-amino uracil by reacting this scaffold with dimethyl acetylene-dicarboxylate (DMAD) leading to synthesis of pyrido[2,3-d]pyrimidines derivatives (Scheme 1). Attack usually occurs at the triple bond in a Michael-type reaction pursued by cyclization either through the other carbon of the acetylene or through the β-carbomethoxy group [56].

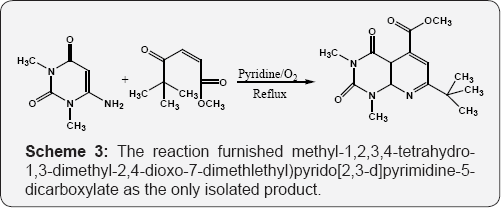
Reaction of dibenzolethylene (DBE) with 6-amino uracil in ethanol under reflux, pyrrolo[2,3-d] pyrimidines were obtained and solution of DBE and 6-amino uracil refluxed in the pyridine under oxygen bubbling, reaction furnished pyrido[2,3-d] pyrimidine-2,4-diones (Scheme 2) [57]. Extended the same reaction by reacting unsymmetrical unsaturated dicarbonyl compound i.e. 3-pivolylacrylate and 6-amino-1,3-dimethyl- uracil in refluxing pyridine. The reaction furnished methyl-1,2,3,4-tetrahydro-1,3-dimethyl-2,4-dioxo-7-dimethlethyl) pyrido[2,3-d]pyrimidine-5-dicarboxylate as the only isolated product (Scheme 3) [58].
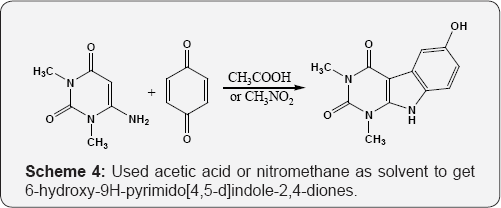
Extended the Nenitzescu reaction to cyclic enamino ketones i.e. 6-amino uracil by reaction of 6-amino-1,3-dimethyluracil with p-benzoquinone (PBQ). Used acetic acid or nitro methane as solvent to get 6-hydroxy-9H-pyrimido[4,5-d]indole-2,4- diones (Scheme 4) [59]. Reactions of electron rich uracils with electron deficient alkenes and to begin with selection were made of Nitroisothiocyanates and nitrostyrenes. When nitroisothio- cyanates reacts with 6-hydro-amino-1,3-methyluracil, afford cyclized product on C5-C6 double bond of uracil (Scheme 5) [60]. Reaction of 6-amino and 6-hydroxyamino uracils with nitrostyrene furnishes 5-alkylated uracil. Nitrostyrene undergoes Michael addition to C5 position of uracil (Scheme 6) [61].
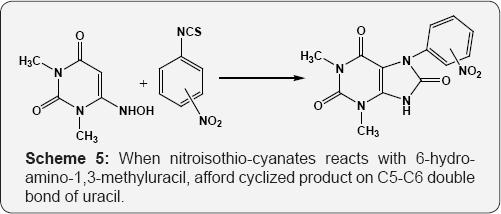
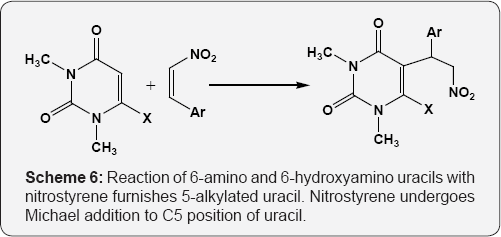
X = NH2, NHOH Ar=C6H5, 4-OCH3-C6H4 4-N(CH3)2 -C6H4, 4-HO-C6H4
Important class of alkenes from Knoevenagel reaction obtained from various aldehydes with cyano compounds mainly malononitriles, ethylcyanoacetate, cyanoacetamide etc. The reaction of functionalized uracils bearing amino and hydroxyamino group at C-6 position with strongly electrophillic cyano olefins affords the pyrido[2,3-d]pyrimidines. Hydrazine- substituted uracil furnishes pyrazolo [3,4-d]pyrimidines (Scheme 7) [62,63].
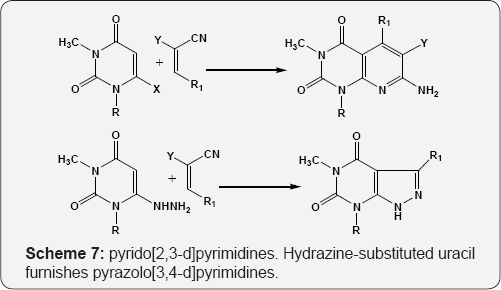
X = NH2, NH R=H, CH3 R1=Ph, 2-furyl, 2-thienyl Y=CN, COOC2H5, CONH2.
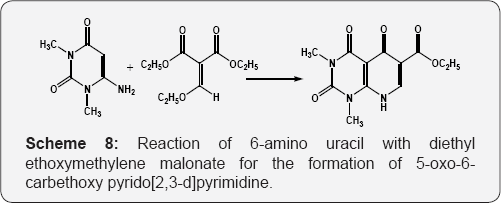
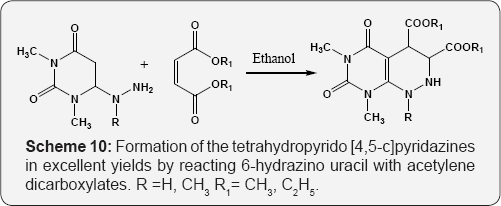
Using different alkenes variants or doing some other minor changes or multicomponent ways etc. Reaction of 6-amino uracil with diethyl ethoxymethylene malonate for the formation of 5-oxo-6-carbethoxy pyrido [2,3-d]pyrimidine (Scheme 8) [64]. The reaction of 6-amino uracil with allene gives the pyrimido [2,3-d]pyrimidines. The reaction proceeds via Michael addition of enamine carbon at C-5 on central carbon atom of allene i.e. dimethyl alkene-1,3-dicarboxylate (Scheme 9) [65]. Formation of the tetrahydropyrido [4,5-c]pyridazines in excellent yields by reacting 6-hydrazino uracil with acetylene dicarboxylates (Scheme 10) [66].

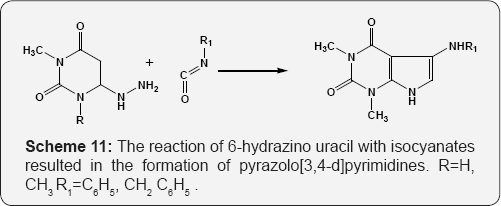
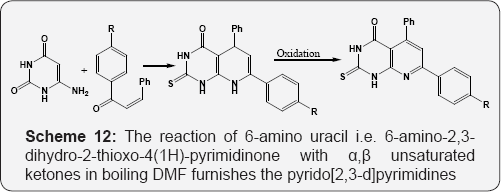
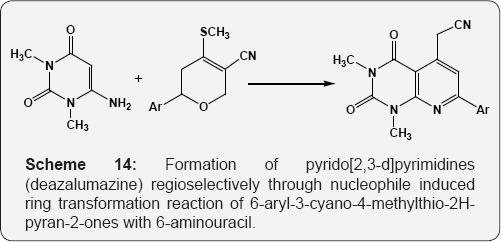
The reaction of 6-hydrazino uracil with isocyanates resulted in the formation of pyrazolo [3,4-d]pyrimidines (Scheme 11) [67]. The reaction of 6-amino uracil i.e. 6-amino-2,3-dihydro-2-thioxo- 4(1H)-pyrimidinone with α,β-unsaturated ketones in boiling DMF furnishes the pyrido [2,3-d]pyrimidines (Scheme 12) [68]. Formation of the pyrido[2,3-d]pyrimidines by reaction of amino pyrimidin-4-one with benzylidene Meldrum's acid derivatives (Scheme 13) [69]. Formation of pyrido[2,3-d]pyrimidines (deazalumazine) regioselectively through nucleophile induced ring transformation reaction of 6-aryl-3-cyano-4-methylthio- 2H-pyran-2-ones with 6-aminouracil (Scheme 14) [70].
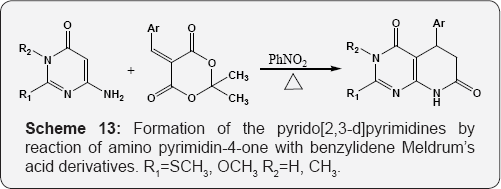
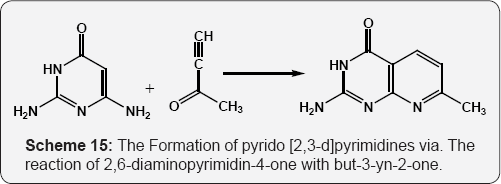
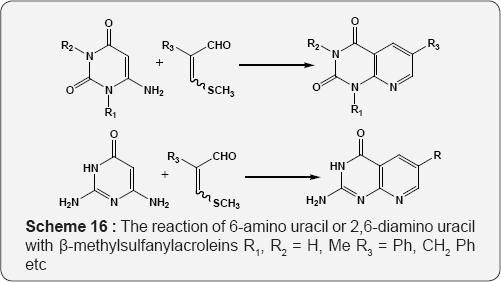
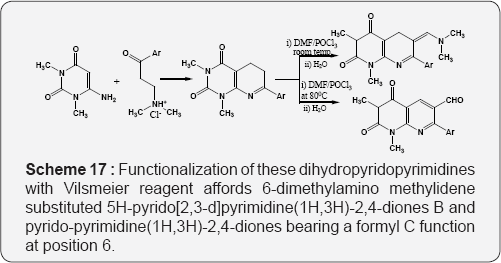
The Formation of pyrido [2,3-d]pyrimidines via. The reaction of 2,6-diaminopyrimidin-4-one with but-3-yn-2-one (Scheme 15) [71]. The reaction of 6-amino uracil or 2,6-diamino uracil with β-methylsulfanylacroleins, derived from alkenyl sulfides by the Vilsmeier reaction, provides to the 6-substituted pyrido [2,3-d] pyrimidines (Scheme 16) [72]. The reaction of 6-amino-1,3-dimethyl uracil with equimolar amount of arylalkane Mannich base leads to the formation of 7-aryl-5,6- dihydropyrido[2,3-d]pyrimidines A. Functionalization of these dihydropyridopyrimidines with Vilsmeier reagent affords 6-dimethylamino methylidene substituted 5H-pyrido[2,3-d] pyrimidine(1H,3H)-2,4-diones B and pyrido-pyrimidine(1H,3H)- 2,4-diones bearing a formyl C function at position 6 (Scheme 17) [73].
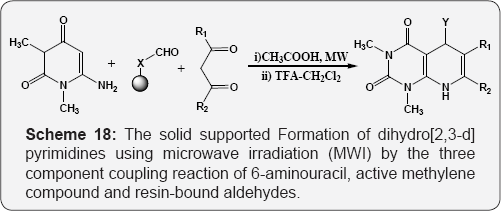
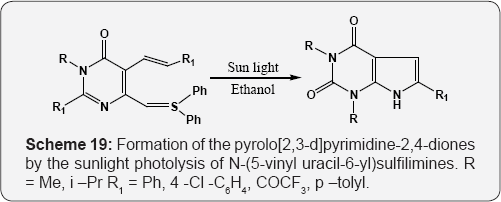
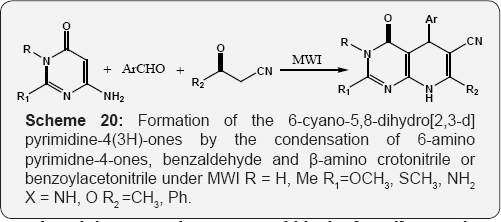
The solid supported Formation of dihydro [2,3-d]pyrimidines using microwave irradiation (MWI) by the three component coupling reaction of 6-aminouracil, active methylene compound and resin-bound aldehydes (Scheme 18) [74]. Formation of the pyrolo [2,3-d]pyrimidine-2,4-diones by the sunlight photolysis of N-(5-vinyl uracil-6-yl)sulfilimines (Scheme 19) [75]. Formation of the 6-cyano-5,8-dihydro[2,3-d]pyrimidine-4(3H)-ones by the condensation of 6-amino pyrimidne-4-ones, benzaldehyde and β-amino crotonitrile or benzoylacetonitrile under MWI (Scheme20) [76].
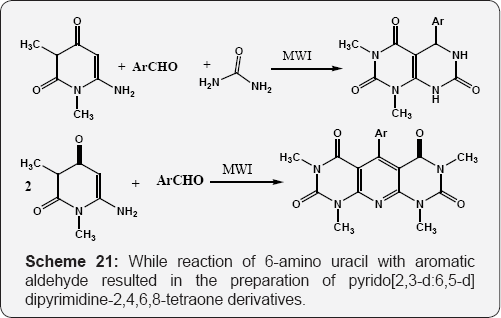
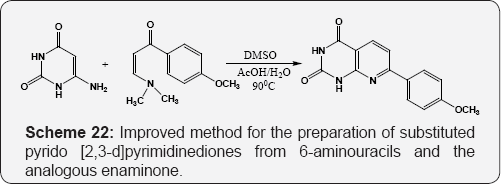
The formation of5,6-dihydro-1,3-dimethyl-5-phenylpyrido[4,5-d] pyrimidine-2,4,7(1H, 3H, 8H)-triones by the reaction of 6-amino uracil, benzaldehyde and urea. While reaction of 6-amino uracil with aromatic aldehyde resulted in the preparation of pyrido [2,3-d:6,5-d] dipyrimidine-2,4,6,8-tetraone derivatives (Scheme 21) [77]. Improved method for the preparation of substituted pyrido [2,3-d] pyrimidinediones from 6-aminouracils and the analogous enaminone [78] (Scheme 22).
Discussion
A series of pyrido [2,3-d]pyrimidines were prepared using 1,3-dimethyl uracil/3-methyl uracil with various substituted arylidene malonic acids. Uracil is essential unit of life cell structure. Nucleoside derivatives have been used as antineoplastic and antiviral agents [79-82]. Much attention has been devoted to the nucleosides with pyrimidinic bases, mainly uracils. Uracils are an important of their applications as bioactive compounds such as 5-iodouridine, 2(S)-will- ardiine [83], zidovudine [84] and tifluridine. This scaffold is importance to chemists and biologists; to chemists as synthon which gives diverse type of molecules and to biologists to screen these entities for biological activities. These compounds were exhibited various types of activities such as antitumor [83], antitubercular [84], anti-diarrhea [85], anti-convulsants [86], antibacterial [87], antimicrobial [88], tyrosine kinase inhibitor [89], calcium channel antagonists [90], antileishmanial [91], diuretic [92], anti-inflammatory and analgesic [93], and other activities. The molecular manipulations have given various types of drugs like 5-florouracil (anti-cancer drug), [94,95] AZT, DDI, DDC, BVDU etc. (nucleoside based anti-AIDs agents) [96-98] host of barbiturates (CNS acting drugs) [96] are indication to the commercial value of this motif [99,100].
References
- Brown DJ (1984) Comprehensive Heterocyclic Chemistry, Katritzky, A. R.; Rees, C. W. Pergamon Press, Oxford 3: 57.
- Lunt E (1974) Comprehensive Org Chem Bartan D, Oils W D Eds. Pergamon Press, Oxford 4: 493.
- Baruah B, Prajapati D, Sandhu J S, Ghosh A C (1996) Studies on pyrimidine-annelated heterocycles: synthesis of novel pyrazolo[3',4':4,5]pyrido[2,3-d]pyrimidines by intramolecular 1,3- dipolar cycloadditions. J Chem Soc Perkin Trans 1 pp: 1999.
- Ikuno K, Kobayashi T, Harada T, Noguchi M, Kakehi A (1995) Studies on fused pyrimidine derivatives. Part 14. Formation and transformation of [4 + 2] cycloadducts, cyclohepta[g]quinazoline derivatives, by the reaction of 5-(arylaminomethylene)-1,3-dimethyl- 6-methyleneperhydropyrimidine-2,4-diones with tropone. J Chem Soc Perkin Trans 1 pp: 1445.
- Jacobson K A (1990) In Comprehensive Medicinal Chemistry, Ernet, J. C. Eds. Pergamon Press, Oxford, 3: 623.
- A. Jones AS, Sayers J R, Walker R T, Clercq E D (1988) Synthesis and antiviral properties of (E)-5-(2-bromovinyl)-2'-deoxycytidine-related compounds. J Med Chem 31: 268.
B. Matsuya H, Yarchoan R, Broder S (1990) Science 249: 1533. - Pinner A Ber (1985) 19: 2519 .
- Brown DJ, Evans FF Fenn M D (1994)"The Pyrimidines”, Wiley, New York, USA.
- Merck Index, 11th Ed. Compd. No. 1635, Merck and Co. Rahway, New Jersy, 1989.
- Merck Index, 11th Ed. Compd. No. 9212, Merck and Co. Rahway, New Jersy, 1989.
- Ibid, Compd. No. 9202, Merck and Co. Rahway, New Jersy, 1989.
- Ibid, Compd. No. 9790, Merck and Co. Rahway, New Jersy, 1989.
- A. Hopkins F G (1889) Nature 40: 335.
B. Hopkins FG (1892) Nature 45: 197581. - Albert A (1952) Quart Revs (London) 6: 197.
- A. Woods DD, Fildes P J (1940) Soc Chem Ind 59: 133.
B. Woods D D (1940) Brit J Exptl Pathol 21: 74. - Patterson EL, Von Saltza MH, Stokstad E L R (1956) The Isolation and Characterization of a PteridineRequired for the Growth of Crithidia fasciculate. J Am Chem Soc 78: 5871.
- Kenner G W Fortschr (1951) Chem Org Naturstoffe 8: 96.
- Lepage G A (1949) Biochem Preparations 1: 5.
- Warburg D, Christian W (1938) Naturwiss 26: 235.
- See, Rosenberg H R (1942) "Chemistry and Physiology of the Vitamins” Interscience, New York, USA.
- Ibid, Compd. No. 8201, Merck and Co. Rahway, New Jersy, 1989.
- Gavrilov MY, Novoseleva G N, Vakhrin M I, Konshin M E KhimFarm Zh (1996) 30: 39.
- Ghorab M M, Hassan A Y (1998) Phosphorus Sulfur Silicon Relat Elem 141: 251.
- Broom A D, Anderson G L, Shim JL (1976) Pyrido[2,3-d]pyrimidines. IV. Synthetic studies leading to various oxopyrido[2,3-d]pyrimidines. J Org Chem 41: 1095.
- Grivsky E M, Lee S, Sigel C W, Duch DS, Nichol C A (1980) Synthesis and antitumor activity of 2,4-diamino-6-(2,5-dimethoxybenzyl)-5- methylpyrido[2,3-d]pyrimidine. J Med Chem 23: 327.
- Furuya S, Ohtaki T (1994) Pyridopyrimidine derivatives, their production and use. Eur Pat Appl EP Chem Abstr121: 205395.
- Heber D, Heers C, Ravens U (1993) Pharmazie 48: 537.
- Sakuma Y, Hasegawa M, Kataoka K, Hoshina K, Yamazaki N, et al. (1989)1,10-Phenanthroline Derivatives.. PCT Int. Chem Abstr 115: 71646.
- Coates W (1990) Pyrimidopyrimidine Derivatives. J Eur Pat 113: 40711.
- Bennett L R, Blankley CJ, Fleming R W, Smith RD, Tessonam D K (1981) Antihypertensive activity of 6-arylpyrido[2,3-d]pyrimidin-7-amine derivatives. J Med Chem 24: 382.
- Davoll J, Clarke J, Elslager EF (1972) Antimalarial substances. 26. Folate antagonists. 4. Antimalarial and antimetabolite effects of 2,4-diamino- 6-[(benzyl)amino]pyrido[2,3-d]pyrimidines. J Med Chem 15: 837.
- Kretzschmer E (1980) Pharmazie 35: 253.
- Shigo S, Hiroshi I (1969) Uracil Derivatives and Related Compounds. IX. : Synthesis of Bucolome's Related Compounds. Yakugaku Zasshi 89: 266.
- Ahluwalia V K, Bhatla R, Khurana A, Kumar R (1990) Indian J ChemSect B 29: 1141.
- Mitsuya H, Weinhold K J, Furman PA, St Clair MH, Nusinhoff Lehrman S, et al. (1985) 3'-Azido-3'-deoxythymidine (BW A509U): an antiviral agent that inhibits the infectivity and cytopathic effect of human T-lymphotropic virus type III/lymphadenopathy-associated virus in vitro. Proc Natl Acad Sci USA 82: 7096.
- Fischl M A, Richman DD, Grieco M H, Gottlieb MS, Volberding P A, et al. (1987) The Efficacy of Azidothymidine (AZT) in the Treatment of Patients with AIDS and AIDS-Related Complex. N Engl J Med 317: 185.
- Mitsuya H, Broder S (1986) Inhibition of the in vitro infectivity and cytopathic effect of human T-lymphotrophic virus type III/lymphadenopathy-associated virus (HTLV-III/LAV) by 2',3'-dideoxynucleosides. Proc Natl AcadSci U S A 83: 1911.
- Bozzette S A, Richman D D (1990) Am J Med 88 (Suppl. 5B): 24S.
- Dunkle L, Cross A, Gugliotti R, Martin R, Browne M, Murry H (1990) Antiviral Res Suppl 1 Abst 145.
- Miyasaka T, Tanaka H, Baba M, Hayakawa H, Walker RT, et al. (1989) A novel lead for specific anti-HIV-1 agents: 1-[(2-hydroxyethoxy) methyl]-6-(phenylthio)thymine. J Med Chem 32: 2507.
- Camarasa M J, Perez Perez M J, San Felix A, Balzarini J, De Clercq E (1992) 3'-Spiro nucleosides, a new class of specific human immunodeficiency virus type 1 inhibitors: synthesis and antiviral activity of [2', 5'-bis-O- (tert-butyldimethylsilyl)-.beta.-D-xylo- and -ribofuranose]-3'-spiro- 5”-[4"-amino-1” 2"-oxathiole 2'', 2”-dioxide] (TSAO) pyrimidine nucleosides. J Med Chem 35: 2721.
- Kim DK, Gam J, Kim YW, Lim J, Kim HT, Kim KH (1997) Synthesis and Anti-HIV-1 Activity of a Series of 1-Alkoxy-5-alkyl-6-(arylthio)uracils. J Med Chem 40: 2363.
- Raic Malic S, Svedruzic D, Gazivoda T, Marunovic A, Hergold Brundic A, et al. (2000) Synthesis and Antitumor Activities of Novel Pyrimidine Derivatives of 2,3-O,O-Dibenzyl-6-deoxy-l-ascorbic Acid and 4,5-Didehydro-5,6- dideoxy-l-ascorbic Acid. J Med Chem 43: 4806.
- Raic Malic S, Hergold Brundic A, Nagi A, Gardisa M, Pavelic K, et al. (1999) Novel Pyrimidine and Purine Derivatives of l-Ascorbic Acid: Synthesis and Biological Evaluation. J Med Chem 42: 2673.
- Jurczyk S, Kolaczkowski M, Maryniak E, Zajdel P, Pawlowski M, et al. (2004) New Arylpiperazine 5-HT1A Receptor Ligands Containing the Pyrimido[2,1-f]purine Fragment: Synthesis, in Vitro, and in Vivo Pharmacological Evaluation. J Med Chem 47: 2659.
- Matsumoto H, Ikeda K, Nagata N, Takayanayi H, Mizuno Y, Tanaka M, Sasaki T (1999) Synthesis of 2,8-Disubstituted Imidazo[1,5-a] pyrimidines with Potent Antitumor Activity. J Med Chem 42: 1661.
- Priego EM, Kuenzel J V F D, Ijzerman A P, Camarasa MJ, Perez Perez MJ (2002) Pyrido[2,1-f]purine-2,4-dione Derivatives as a Novel Class of Highly Potent Human A3 Adenosine Receptor Antagonists. J Med Chem 45: 3337.
- Quiroga J, Cisneros C, Inuasty B, Abonia R (2006) J Het Chem 43: 463.
- Shaaban MR Saleh T S, Farag AM (2007) Synthesis and Antimicrobial Evaluation of Novel Pyrazolo [1,5-a] pyrimidine, Triazolo[1,5-a] pyrimidine and Pyrimido[1,2-a]benzimidazole Derivatives. Heterocycles 71: 1765.
- Sherman H, Kaplan A M (1975)Toxicol Appl Phermacol 34: 189.
- Herbicide Handbook (1989) 6th Ed Weed Sci Soc Of American Champarign.
- Thomas WT (1989) Agriculture Chemical Book II, Herbicides 19891990, Rev., Thomson Publications, Fresno, Calif.
- Kirk Othmer Series on Encycl (1991) Chem Tech 4th Ed JohnWiley and Sons 13: 115.
- < Stewart SJ A U Patent (2001) Chem Abstr 134: 67474.
- Kossel A, Stendel H (1902) Hoppe Seyler's Physiol Chem 37: 246.
- Shim JL, Niess R, Broom AD (1972) Acylation of some 6-aminouracil derivatives. J Org Chem 37: 578.
- Tamura Y, Sakaguchi T, Kawasaki T, Kita Y (1975) A Regioselective Synthesis of Pyrrolo- and Pyrido[2,3-d]pyrimidine-2,4-diones. Heterocycles 3: 183.
- Bennect G B, Mason R B (1977) The regioselective behavior of unsaturated keto esters toward vinylogous amides. J Org Chem 42: 1919.
- Bernier JC, Henichart JP (1981) Extension of the Nenitzescu reaction to a cyclic enamino ketone. One-step synthesis of 6-hydroxy-9H- pyrimido[4,5-b]indole-2,4-dione. J Org Chem 46: 4197.
- Prasad A S, Sandhu JS, Baruah JN (1984) J Het Chem 21: 267.
- Prasad AS, Sandhu JS, Baruah JN (1984) J Het Chem 21: 1657.
- Gogoi M, Bhuyan PJ, Sandhu JS (1984) Arylidenemalononitriles in heterocyclic syntheses: a novel synthesis of pyrido[2,3-d]pyrimidines. J Chem Soc Chem Commun pp: 1549.
- Bhuyan P J, Boruah R C, Sandhu J S (1990) Studies on uracils. 10. A facile one-pot synthesis of pyrido[2,3-d]- and pyrazolo[3,4-d]pyrimidines. J Org Chem 55: 568.
- Anderson G L (1985) J Het Chem 1469.
- Doad G J S, Okor D I, Sceinmenn F (1998) J Chem Soc PerkinTrans 1 2993.
- Bhuyan P J, Lekhok KC, Sandhu J S (1999) Tetrahedron Lett 40: 1793.
- Bhuyan P J, Borah H N, Sandhu JS (2002) Tetrahedron Lett 43: 895.
- Quiroga J, Insuasty B, Sanchez A, Nogueras M, Meier H (1992) J Het Chem 29: 1045.
- Quiroga J, Hormaza A, Insuasty B, Nogueras M, Sanchez A, Hanold N, Meier H (1997) J Het Chem 34:521.
- Shrivastava P, Saxena A S Ram V J (2000) An Elegant Approach Towards the Regioselective Synthesis of Deazalumazines Through Nucleophile Induced Ring Transformation Reactions of 6-Aryl-3-cyano-4- methylthio-2H-pyran-2-ones. Synthesis 4: 541.
- Bagley M C, Hughes DD, Lloyd D, Powers VEC (2001) Tetrahedron Lett 42: 6585.
- Kuwada T, Harada K, Nobuhiro J, Choshi T, Hibino S (2002) A New Synthesis of 6-Substitiuted Pyrido[2,3-d]pyrimidines. Heterocycles 57: 2081.
- Girreser U, Heber D, Schutt M (2004) Tetrahedron 60: 11511.
- Aggarwal A, Chauhan P M S (2005) Tetrahedron Lett 46: 1345.
- Matsumoto N, Takahasmi M (2005) Tetrahedron Lett 46: 5551.
- Quiroga J, Cisneros C, Insuasty B, Abonia R J (2006) Het Chem 43: 299.
- Dabiri M, Arvin Nezhad H, Khavasi HR, Bazgir A (2007) Tetrahedron 63: 1770.
- Churchill G H, Raw S A, Powell L, (2011) Tetrahedron Lett 52: 3657.
- Suresh A Saini, D Kumar, JS Sandhu (2009) Multicomponent eco- friendly synthesis of 3,4-dihydropyrimidine-2-(1H)-ones using an organocatalyst Lactic acid.Green Chem Lett Rev 2: 29.
- Suresh D Kumar JS, Sandhu (2010) Microwave enhanced, solvent free green protocol for the production of 3,4-dihydropyrimidine-2-(1H)- ones using AlCl3.6H2O as a catalyst. Indian J Chem 49B: 360.
- A. Bhuyan P J, Borah H N, Sandhu J SJ (1999) Studies on uracils: an efficient method for the synthesis of novel 1-allyl-6-(1',2',3'-triazolyl) analogues of HEPT. Chem Soc Perkin Trans 1 pp: 3083.
- A. Mitsuya H, Yarchoan R, Broder S (1990) Science 249: 1533.
B. Barral K, Courcambeck J, Pepe G, Balzarini J, Neyts J, et al. (2005) Synthesis and Antiviral Evaluation of Cis-Substituted Cyclohexenyl and Cyclohexanyl Nucleosides.J Med Chem 48: 450. - A. Broom A D, Shim J L, Anderson G J (1976) Pyrido[2,3-d]pyrimidines. IV. Synthetic studies leading to various oxopyrido[2,3-d]pyrimidines. J Org Chem 41: 1095.
B. Dolman N P, Troop H M, More J C A, Alt A, Knauss J L, et al. (2005) Synthesis and Pharmacology of Willardiine Derivatives Acting as Antagonists of Kainate Receptors. J Med Chem 48: 7867. - A. Grivsky E M, Lee S, Sigel C W, Duch D S, Nichol C A (1980) Synthesis and antitumor activity of 2,4-diamino-6-(2,5-dimethoxybenzyl)-5- methylpyrido[2,3-d]pyrimidine. J Med Chem 23: 327.
B. Mai A, Sbardella G, Artico M, Ragno R, Massa S, et al. (2001) Structure- Based Design, Synthesis, and Biological Evaluation of Conformationally Restricted Novel 2-Alkylthio-6-[1-(2,6-difluorophenyl)alkyl]-3,4- dihydro-5-alkylpyrimidin-4(3H)-ones as Non-nucleoside Inhibitors of HIV-1 Reverse Transcriptase. J Med Chem 44: 2544. - Tanifum EA, Kots A Y, Choi BK, Murad F, Gilbertson SR (2009) Bioorg Med Chem Lett 19: 3067.
- Matsumoto J, Minami S (1975) Quinuclidine chemistry.3. .beta.-cis-2- (4'-Chlorobenzhydryl)-3-quinuclidinol, a new central nervous system stimulant. Importance of the benzhydryl configuration. J Med Chem 18:
- Narayana BL, Rao A R R, Rao P S (2009) Euro J Med Chem 44: 1369.
- Mohamed N R, El Saidi M MT, Ali Y M, Elnagdi M H (2007) Bioorg Med Chem 15: 6227.
- Thompson A M, Rewcastle G W, Bridges A J, Fry D W, Kraker A J, et al. (1995) J Med Chem 38: 3780.
- Pastor A, Alajarin R, Vaquero J J, Alvarez Builla J, Fau de Casa Juana M, et al. (1994) J Tetrahedron 50: 8085.
- Youssouf M S, Kaiser P, Singh G D, Singh S, Bani S, Gupta V K, Satti N K, Suri K A, Johri R K (2008) Int Immunopharmacol 8:1049.
- Bulicz J, Daniela C G, Bertarelli D C G, Baumert D, Fulle F, et al. (2006) Bioorg Med Chem 14: 2837.
- El Gazzar A BA, Hafez H N (2009) Bioorg Med Chem Lett 19: 3392.
- Hirota K, Kubo K, Sajiki H, Kitade Y, Sako M, et al. (1997) Reactivity of Thioaldehyde: Cyclization Reaction of 6-Amino-1,3-dimethyl-5- thioformyluracil with Enamines into Pyrido[2,3-d]pyrimidine-2,4- (1H,3H)-diones. J Org Chem 62: 2999.
- Heidelberger C, Arafield F J (1963) Experimental and Clinical Use of Fluorinated Pyrimidines in Cancer Chemotherapy. Cancer Res 23: 1226.
- A. Gavrilov M Y, Mardanova I G, Kolla V E, Konshin M E (1988) Synthesis, antiinflammatory and analgesic activities of 2-arylamino-5,6,7,8- tetrahydroquinoline-3-carboxamides. Pharm Chem J 22: 554.
B. Daniel Lednicer, Charles R Hauser(1958) Five- vs. Six-membered Ring Formation in the Cyclization of 2,3,4-Triphenylbutyric Acid; the Relative Importance of Stereochemistry. J Am Chem Soc 80: 3449. - Wamho H, Winfried S (1986) 2,7-Diphenyloxepin. J Org Chem 51: 2787.
- Broom A D, Shim J L, Anderson C L (1976) Pyrido[2,3-d]pyrimidines. IV. Synthetic studies leading to various oxopyrido[2,3-d]pyrimidines. J Org Chem 41(4): 1095.
- A. Gohain M, Prajapati D, Gogoi BJ, Sandhu JS (2004) A Facile Microwave Induced One-Pot Synthesis of Novel Pyrimido[4,5-d]pyrimidines and Pyrido[2,3-d]pyrimidines under Solvent-Free Conditions. Synlett 1179-1182.
B. Slatt J, Janosik T, Wahlstrom N, Bermang J (2005) Synthetic applications of 3-(cyanoacetyl)indoles and related compounds. J Heterocycl Chem 42: 141-145.
C. Slätt J, Romero I, Bergman J (2004) Cyanoacetylation of Indoles, Pyrroles and Aromatic Amines with the Combination Cyanoacetic Acid and Acetic Anhydride. Synthesis 2760.
D. Berghot M A, Moawad EB (2003) Eur J Pharm Sci 20: 173. - A. Suresh D Kumar, J S Sandhu (2009) New Efficient Protocol for the Production of Hantzsch 1,4-Dihydropyridines Using RuCl3. Synth Comm 39: 1957-1965.
B. Suresh, R S Bhatti, P Krishan J S Sandhu (2010) Indian J Chem Soc 87: 707;
C. Suresh, A Saini, JS Sandhu (2008) Li Br: A Mild Lewis Acid Catalyst for Efficient One-Pot Synthesis of a-Amino Nitriles. Synth Comm 38: 3655.
B. Loupy A, Song SJ, Sohn SM, Lee YM, Kwon TW (2001) Solvent-free bentonite-catalyzed condensation of malonic acid and aromatic aldehydes under microwave irradiation. J Chem Soc Perkin Trans 1 pp: 1220.
C. Chu C K, Baker D C Eds (1993) Nucleosides and Nucleotides as Antitumor and Antiviral Agents; Plenum Press: New York, USA.






























