Biological Activity of Pyrimidine Derivativies: A Review
Ajmal R. Bhat*
Department of Chemistry, S. B. B.S. University, India
Submission: March 20, 2017; Published: April 03, 2017
*Corresponding author: Ajmal R Bhat, Department of Chemistry S. B. B.S. University,Punjab-144030, India, Tel: 91-9797354802; Email: bhatajmal@gmail.com
How to cite this article: Ajmal R. B. SBiological Activity of Pyrimidine Derivativies: A Review. Organic & Medicinal Chem IJ. 2017; 2(2): 555581. DOI: 10.19080/OMCIJ.2017.02.555581
Abstract
The Pyrimidine derivativies in the chemistry of biological systems has attracted much attention due to availability in the substructures of therapeutic natural products. As a result of their prominent and remarkable pharmacological activity, pyrimidine derivatives has been found the most prominent structures in nucleic acid. The present review gives brief information about biological activity of annulated pyrimidine derivatives.
Keywords: Pyrimidine derivativies; Anti-inflammatory drugs; anticancer activity; Anti-HIV agents; Antihypertensive drugs
Introduction
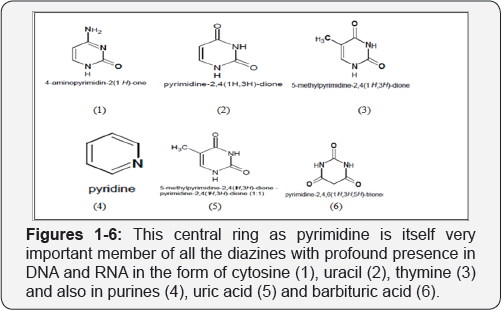
Progressive and prospective research in the field of pharmaceutical chemistry is having most important focus for the design and formulation of new and effective drugs and their successful application in applied field. The main concern to every research work is to develop and prepare pharmaceutical substances and preparation, which are new, effective and original and to overcome with more accuracy over a drug already known. Preparation and development for these drugs may have its effect either quantitatively or by qualitatively, the absence of undesirable side effects, lower toxicity, improved stability or decreased cost is preferred. Dihydropyrimidines are important member of heterocyclic family and its large number of novel derivatives possess effective pyrimidine ring as central core/ ring with heteroatoms as nitrogen [1]. This central ring as pyrimidine is itself very important member of all the diazines with profound presence in DNA and RNA in the form of cytosine (1), uracil (2), thymine (3) and also in purines (4), uric acid (5) and barbituric acid (6). They have gained much interest in the field of drug research to design and discovery of new physiologically and pharmacologically active compounds. Annulated pyrimidine derivatives have received great attention during the past years, because they exhibit a wide range of biological activities such as antibacterial and antifungal activity [2], antiasthmatics, antiallergic [1], antihypertensive [3], cardiotonic [4], bronchodilator [5] or antitumor [6] activity.
The preparation of the heterocycles containing uracil ring poses significant synthetic challenges. Moreover, several alkaloids obtained from marine source also constituted of these moieties which also impart pharmacological properties (Figures 1-6). The wide applicability associated with these heterocycles and its novel compounds encouraged the chemists to contribute and synthesis large number of biologically active novel drugs and introduce some efficient methods.
For proceeding to drug preparation and design rather safe and effective drug, the primary condition is to have sufficient knowledge about the various metabolic reactions and endogenous compounds undergo in the organism. The selection and choice for a particular functional group associated to molecular structure while preparing a drug is of great importance and its proper pharmaceutical effectiveness rather inferring the internal system of organism is primary task for medicinal chemists. In some cases drugs have adequate pharmacological activity but have an inadequate pharmacokinetic profile (i.e. absorption, distribution, metabolism and excretion) and these are tested repeatedly in laboratory to find their exact and effective pharmaceutical value. The reactivity of a drug is believed to be due to the interaction of the drug with enzymes, receptors and other molecules found in the biological system and its pharmaceutical effect depend on the stability of the drug- enzyme complex as well as the fraction of active and allosteric sites occupied by the drug and is guaranteed by proper binding of drug molecules with the enzymes.
Heterocycles have great active involvement in pharmaceutical phenomena shared by their specific chemical reactivity and provide false synthons in biosynthetic process or block the normal functioning of biological receptors. Most of the nitrogenous bases which occur as natural alkaloids are present in plants and many antibiotics including penicillin and streptomycin have also heterocyclic ring system. All classes and groups with different heteroatom in core ring exhibit highest level of proficiency. Pyrimidines are six membered compounds have wide range of biological and Pharmaceutical activities, like their inhibitory action against many fatal diseases.
Biological Activity of Heterocyclic Compounds
The extraordinary role of pyrimidine ring in dihydropyrimidine presumes as essential part in nucleic bases, vitamins, enzymes, chlorophyll, hemoglobin, hormones and moreover the broad spectrum of pharmaceutical properties, subsequently gives birth to new biologically active anti-dots in drug industries. The biological investigation of these various modified and manipulated derivatives exhibits vast activities are briefly discussed as under with some known examples and their respective structures.
Anti-HIV agents
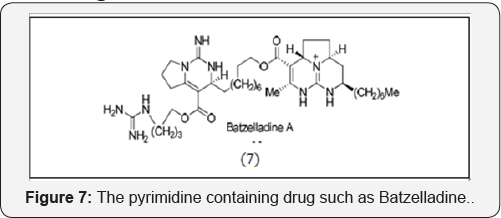
The pyrimidine containing drug such as Batzelladine (7) obtained from marine natural source and inhibits the binding of HIV gp-120 to CD4cells [7] (Figure 7).
Antitumor activity
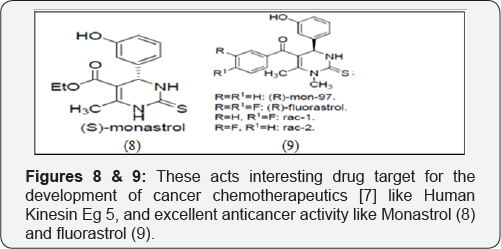
These acts interesting drug target for the development of cancer chemotherapeutics [8] like Human Kinesin Eg 5, and excellent anticancer activity like Monastrol (8) and fluorastrol [9] (Figures 8 & 9).
Antimalarial drugs
The application of these drugs kills the plasmodium causing malaria. Combination of sulphamethoxazole with pyrimethamine is a novel antimalarial drug [9]. Pyrimidinone-amides derivatives of dihydropyrimidines 50- (Hsp70 modulators), could inhibit the replication of the pathogenic P. falciparum stages in human red blood cells (Figures 10 & 11).

Anti-microbial drugs
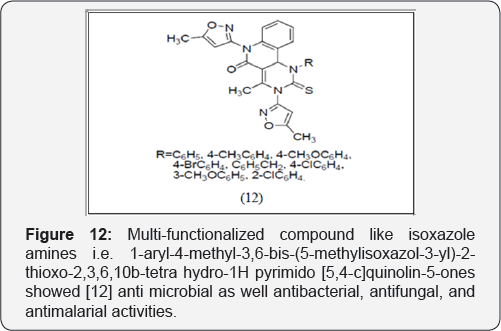
Some Biginelli multi-functionalized compound like isoxazole amines i.e. 1-aryl-4-methyl-3,6-bis-(5-methylisoxazol-3-yl)-2- thioxo-2,3,6,10b-tetra hydro-1H pyrimido [5,4-c]quinolin-5- ones showed anti microbial as well antibacterial, antifungal, and antimalarial activities [10] (Figure 12).
Anti-inflammatory drugs
A series of compounds 3-(4,6-disubtituted-2-thioxo- 1,2,3,4-tetrahydro pyrimidin-5-yl) propanoic acid derivatives were examined for their anti-inflammatory activity using rat paw edema method and most of them showed significant antiinflammatory activity [2] (Figures 13-15).

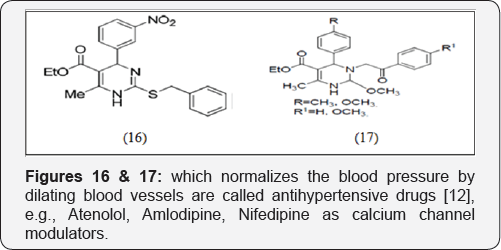
Antihypertensive drugs
Drugs, which normalizes the blood pressure by dilating blood vessels are called antihypertensive drugs [11], e.g, Atenolol, Amlodipine, Nifedipine as calcium channel modulators [12] and Biginelli compounds viz SQ 32926, SQ 32547 (effective orally active antihypertensive agents) (Figures 16 & 17).
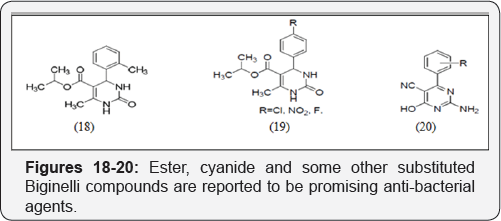
Anti-bacterial drugs
Ester, cyanide and some other substituted Biginelli compounds are reported to be promising anti-bacterial agents [13] (Figures 18-20).
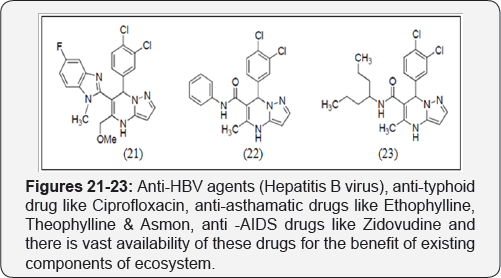
Potassium Channel Antagonists
There are some drugs which work as Potassium channel antagonists like Annulation of benzimidazole ring, showed potassium channel antagonists activity [14] (Figures 21-23). Besides all above large number of cited pharmaceutical drugs, there is presence of some other active drugs which equally play their respective role in this drug arena are, anti-HBV agents (Hepatitis B virus), anti-typhoid drug like Ciprofloxacin, anti- asthamatic drugs like Ethophylline, Theophylline & Asmon, anti -AIDS drugs like Zidovudine and there is vast availability of these drugs for the benefit of existing components of ecosystem [15-22].
Conclusion
Annulated pyrimidine derivatives are one of the most prominent structures found in nucleic acid including uracil, thymine, cytosine, adenine, and guanine are fundamental building blocks for deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Therefore Pyrimidine derivativies are the important heterocyclic compounds because it is an essential constituent of all cells and potential therapy for the treatment of large number of diseases, This review focus on the various potent annulated Pyrimidine derivatives and observed that cyclic N-type structure with different electron withdrawing and electron donating substitution groups showed broad biological act activity.
References
- A R Bhat, R S Dongra, R S Selokar (2014) Potent In-Vitro Antibacterial And Antifungal Activities Of Pyrano[2,3-D]Pyrimidine Derivatives With Quantitative Yield. Int J Pharma Bio Sci 5: 422-430.
- E Klein, S DeBonis, B Thiede, D A Skoufias, F Kozielskib, et al. (2007) New chemical tools for investigating human mitotic kinesin Eg5. Bio Org Med Chem 15(19): 6474-6488.
- K Furukawa, T Hasegawa (1996) ChemAbstr124: 289568c.
- JW Ellingboe, NJ Princeton (1996) Chem Abstr124:176134q.
- D Heber, C Heers, U Ravens (1993) Pharmazie Positive Inotropic Activity of 5-Amino-6-Cyano-1,3-Dimethyl-1,2,3,4-Tetrahydropyrido[2,3-D] pyrim Idine-2,4-Dione in Cardiac Muscle From Guinea-Pig and Man. Part 6: Compounds With Positive Inotropic Activity. Pharmazie 48(7): 537-541.
- WJ Coates (1990) Chem Abstr 113: 40711 J Eur Pat 351058.
- EM Grivsky, S Lee, CW Sigel, DS Duch, CA Nichol (1980) Synthesis and antitumor activity of 2,4-diamino-6-(2,5-dimethoxybenzyl)-5- methylpyrido[2,3-d]pyrimidine. J Med Chem 23(3): 327-329.
- C D Cox, M J Breslin, B J Mariano (2004) Two-step synthesis of ß-alkyl chalcones and their use in the synthesis of 3,5-diaryl-5-alkyl-4,5- dihydropyrazoles. Tetrahedron Lett 45(7): 1489-1493.
- C O Kappe, O V Shishkin, G Uraya, P Verdinoa (2000) X-Ray Structure, Conformational Analysis, Enantioseparation, and Determination of Absolute Configuration of the Mitotic Kinesin Eg5 Inhibitor Monastrol. Tetrahedron 56(13): 1859-1862.
- B K Karale, C H Gill, M Khan, V P Chavan, A S Mane, et al. (2002) Synthesis of 3-methyl-4-[(chromon-3-yl) methylene]-1-phenylpyrazolin- 5(4H)-ones and their conversion into 2-thio-5-hydroxy-5H [1]-benzopyrano[4,3-d] pyrimidines. Indian J Chem 41B(9): 19571959.
- R W Lewis, J Mabry, J G Polisar, K P Eagen, B Ganem, et al. (2010) Dihydropyrimidinone Positive Modulation of 6-Subunit-Containing Y-Aminobutyric Acid Type A Receptors, Including an Epilepsy-Linked Mutant Variant. Biochemistry 49(23): 4841-4851.
- K S Atwal, B N Swanson, S E Unger, D M Floyd, S Moreland, et al. (1991) Dihydropyrimidine calcium channel blockers. 3. 3-Carbamoyl-4-aryl- 1,2,3,4-tetrahydro-6-methyl-5-pyrimidinecarboxylic acid esters as orally effective antihypertensive agents. J Med Chem 34(2): 806-811.
- Ila Sircar Eva K, Gregor K, R Anderson, Stehen J Haleen, et al. (1991) Calcium channel blocking and positive inotropic activities of ethyl 5-cyano-1,4-dihydro-6-methyl-2-[(phenylsulfonyl)methyl]-4-aryl- 3-pyridinecarboxylate and analogs. Synthesis and structure-activity relationships. J Med Chem 34(7): 2248-2260.
- J C Barrow, P G Nantermet, H G Selnick, K L Glass, K E Rittle, et al. (2000) In Vitro and in Vivo Evaluation of Dihydropyrimidinone C-5 Amides as Potent and Selective a1A Receptor Antagonists for the Treatment of Benign Prostatic Hyperplasia. J Med Chem 43(14): 2703-2718.
- B C Ranu, A Hajra, U Jana (2000) Indium(III) Chloride-Catalyzed One-Pot Synthesis of Dihydropyrimidinones by a Three-Component Coupling of 1,3-Dicarbonyl Compounds, Aldehydes, and Urea: An Improved Procedure for the Biginelli Reaction. J Organic Chem 65(19): 6270-6272.
- A Hantzsch (1882) Ann Liebigs Chem 1: 215.
- List, B Angew (2010) Emil Knoevenagel and the Roots of Aminocatalysis. Chem Int Ed 49(10): 1730-1734.
- P Biginelli (1893) Gazz Chim Ital 23: 360-416.
- AN Chiang, JC Valderramos, R Balachandran, R J Chovatiya, B P Mead, et al. (2009) Select pyrimidinones inhibit the propagation of the malarial parasite, Plasmodium falciparum. Bioorg Med Chem 17(4): 1527-1533.
- A. S N Mokale, S S Shinde, R D Elgire, J N Sangshetti, D B Shinde (2010) Synthesis and anti-inflammatory activity of some 3-(4,6-disubtituted- 2-thioxo-1,2,3,4-tetrahydropyrimidin-5-yl) propanoic acid derivatives. Bio org Med Chem Lett 20(15): 4424-4426.
B. S S Bahekar, D B Shinde (2004) Synthesis and anti-inflammatory activity of some [4,6-(4-substituted aryl)-2-thioxo-1,2,3,4-tetrahydro- pyrimidin-5-yl]-acetic acid derivatives. Bioorg Med Chem Lett 14(7): 1733-1736.
C. S S Bahekar, D B Shinde (2003) Synthesis and anti-inflammatory activity of some 2-amino-6-(4-substituted aryl)-4-(4-substituted phenyl)-1,6-dihydropyrimidine-5-yl-acetic acid derivatives. Acta Pharm 53: 223-229. - S Batra, S Nag, R Pathak, M Kumar, P K Shukla (2006) Synthesis and antibacterial evaluation of ureides of Baylis-Hillman derivatives. Bioorg Med Chem 16(14): 3824-3828.
- A. H A Stefani, C B Oliveira, R B Almeida, C M P Pereira, R C Braga, et al. (2006) Dihydropyrimidin-(2H)-ones obtained by ultrasound irradiation: a new class of potential antioxidant agents. Eur J Med Chem 41(4): 513-518.
B. LIsmaili,A Nadaradjane, L Nicod, C Guyon, AXicluna,etal.(2008) Synthesis and antioxidant activity evaluation of new hexahydropyrimido[5,4-c] quinoline-2,5-diones and 2-thioxohexahydropyrimido[5,4-c] quinoline-5-ones obtained by Biginelli reaction in two steps. Eur J Med Chem 43(6): 1270-1275.






























