Mitochondrial Toxicity can be Assessed by Measuring Mitochondrial Membrane Potentials at Single Cell Level
Ozge Tuncel and Serdar Ozcelik*
Department of Chemistry, Izmir Institute of Technology, Turkey
Submission: August 16, 2021; Published: August 31, 2021
*Corresponding author: Serdar Ozcelik, Department of Chemistry, Izmir Institute of Technology, Turkey
How to cite this article: Ozge Tuncel, Serdar Ozcelik. Mitochondrial Toxicity can be Assessed by Measuring Mitochondrial Membrane Potentials at Single Cell Level. Open Acc J of Toxicol. 2021; 5(1):555654. DOI: 10.19080/OAJT.2021.05.555654.
Abstract
Biological responses of individual cells to carcinogen and toxic chemicals may vary due to sub-cellular heterogeneity or biological state of individual cells. Mitochondrial membrane potentials of individual cells vary, therefore mitochondrial toxicity should be assessed at single-cell level in real-time and living cell conditions to better understand mitochondrial heterogeneity.
Keywords: Adhesion molecules; Pro-inflammatory cytokines; Matrix metalloproteinase; Chemotactic cytokines; Chemokine ligands; Glycosaminoglycans
Abbreviations: Mitochondrial membrane; Mitochondrial toxicity; Human bronchial epithelium; Reactive oxygen species; Glutathione
Introduction
Cadmium is a toxic element and exists as Cd2+ in biological systems and physiologically non-essential ion [1,2]. It is also listed as class I carcinogen [3]. The charge to diameter ratio of Cd2+ is 2.06 that is very close to the ratio of Ca2+ ion. This dense charge exerts strong electrostatic forces on biological molecules, particularly on thiol sites of proteins [4,5]. It is considered that Cd2+ ions diffuse cellular membrane by help of Ca2+ ion channels and some receptors because of similar charge to size ratio [1,2]. Cd2+ ions can accumulate in mitochondria due to their highly negative membrane potentials. Accumulated Cd2+ ions may interfere with electron transfer processes and cause mitochondrial disruption leading to cell death that can be assessed by measuring Mitochondrial Membrane Potential (MMP) as a method.
In-vitro assessment of Cd2+ ion exposure uses bulk measurement of MMPs as total response of a cell. However, biological responses of individual cells to the carcinogen/toxic exposure may vary due to sub-cellular heterogeneity and/or biological state of individual cells. Therefore, measuring MMPs at single cell level is important to assess biological responses and to reach reliable conclusions. Here, we aim to demonstrate this fact, by quantitatively evaluating MMPs of individual cells.
Lung is an organ that accumulates Cd2+ ions upon their inhalation as an entry mode to human body, hence BEAS-2B as a non-tumorigenic cell line established from normal human bronchial epithelium, and A549 as a human non-small lung cancer cell (adenocarcinoma alveolar epithelial cell) are used as model systems to determine mitochondrial responses at single cell level. Cd2+ ion exposure generates Reactive Oxygen Species (ROS) - oxygen containing charged species- in various cells [6,7]. Here, Cd2+ ion is used as stressor to induce ROS and then MMPs at single cell level are quantitatively evaluated by microscopy. The findings show that MMP of individual cells greatly varies, suggesting that MMPs should be assessed at single-cell level to withdraw reliable conclusions regarding mitochondrial toxicity.
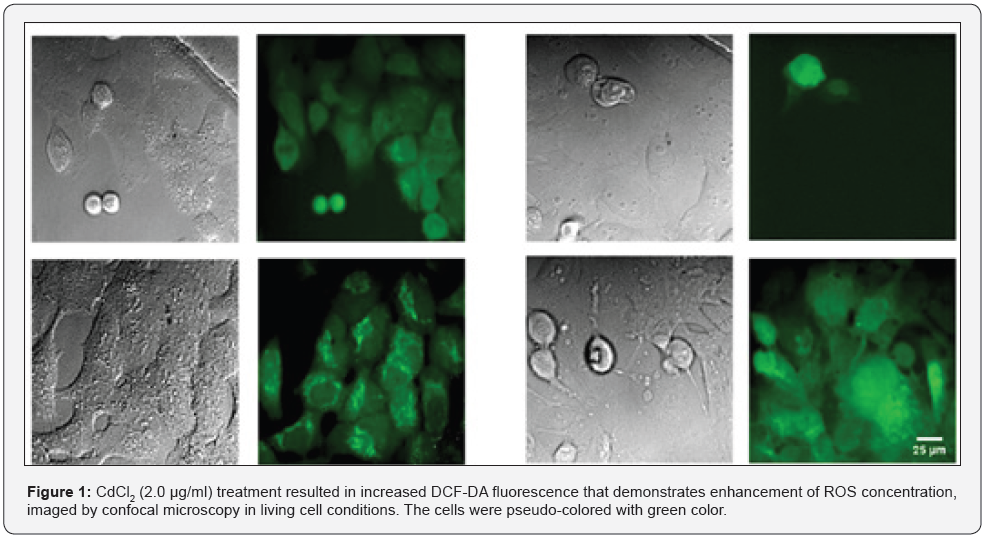
(Figure 1) shows enhanced DCF-DA fluorescence suggesting ROS generation induced by CdCl2 (10 μg/mL) treatment. Both cell lines at single cell level are strongly affected by the presence of Cd2+ ions when compared to control images. It was noted that both cells have high level basal ROS activity. Cd2+ ion shows high affinity to thiols as a primary target for free Cdions. Glutathione (GSH) possesses thiols and are highly abundant in cells. Cd2+ ion is nonredox-active therefore it cannot induce ROS production directly, but it displaces redox-active ions such as Ca2+ and Fe2+ ions from GSH and reduced GSH leads to an oxidative environment [8]. These free ions enhance the production of hydroxyl radicals through the Fenton reaction [1,9,10].
(Figure 2) demonstrates dose dependent alterations of MMPs averaged over multiple wells. The MMP is directly proportional to the fluorescence intensity of JC1, a mitochondrial membrane potential probe. The bar-chart type graphic is the common representation that shows the cumulative MMP responses. The MMPs of the treated cells were compared with control.
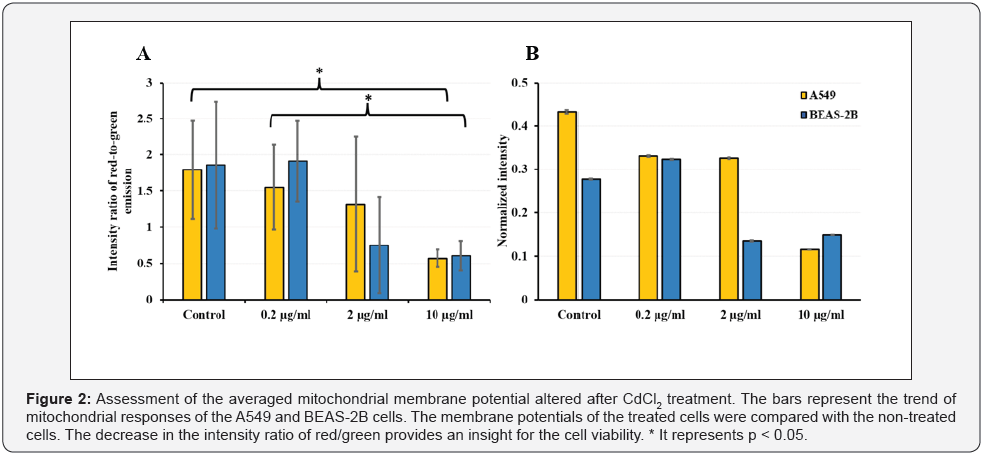
The decrease in the intensity ratio of red to green channels provides an insight for the cells’ viability at bulk-level (a singlewell) but not at individual cell level. Therefore, assessing MMPs at single cell level is required. However, there is a complexity at single mitochondrion level because mitochondria exist as a network, making trustworthy assessment of MMP at individual organelle level problematic. But measurement of MMPs of each cell individually can be easily and consistently carried out to determine the mitochondrial responses and fate of cells. When the intensity ratio is normalized to the cell area, the error bars are dramatically smaller, pointing to strong heterogeneous cellular responses.
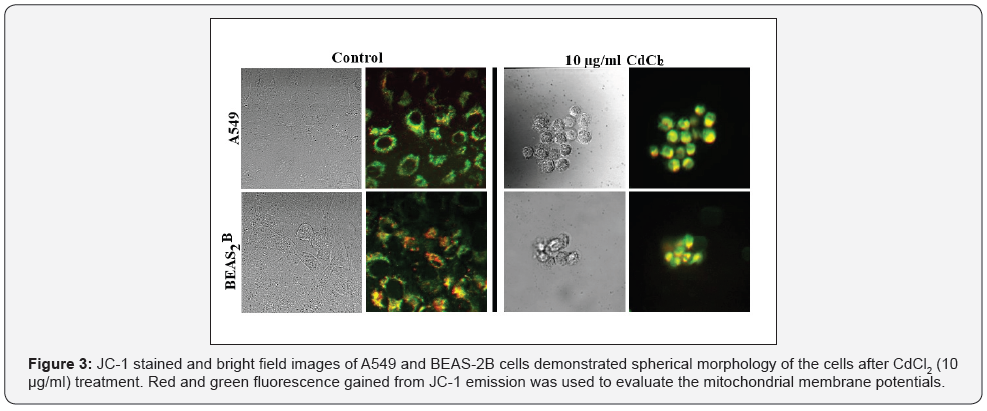
(Figure 3) is an image panel of JC1 stained cells following CdCl2 uptake. The cells were stained and imaged with JC1 fluorescence having two emissions: the green emission represents mitochondria with low membrane potentials because of disruption of mitochondrial functions and the red emission is originating from the aggregated form of JC1 indicating high membrane potential of functioning mitochondria. This way of imaging of the cells in real time and under living-cells conditions allows us to track changes in mitochondria through MMP measurements. In addition, this method may also provide evaluation of other mitochondrial parameters such as mass, size, change of network, mitochondrial fusion and fission in real time. Since obtaining the total MMPs of a single cell is the aim, we do not focus on getting the other mitochondrial parameters currently, but it is certainly doable.
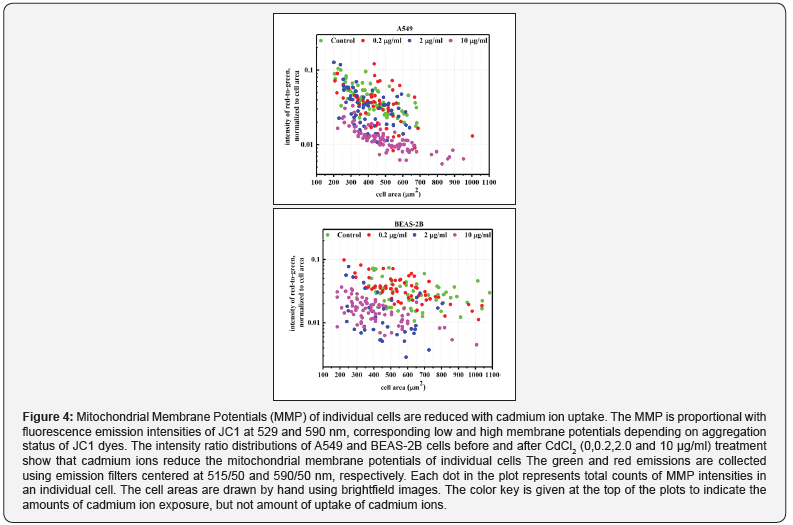
(Figure 4) a scatter type graphic- shows MMP distribution before and after CdCl2 uptake (0.0, 0.2, 2.0 and 10.0 μg/mL that correspond to 0.0, 10, 20 and 50 μM) of A549 and BEAS2B cells at single cell level. The MMP is proportional with fluorescence emission intensities of JC1 at 529 (the green emission) and 590 nm (the red emission), corresponding to the low and high membrane potentials. The green and red emissions are collected using emission filters centered at 515/50 and 590/50 nm, excited at 488 and 532 nm respectively. The area normalized intensity ratio of red to green emissions is indicating MMP changes at single cell level. As shown, the mitochondrial responses greatly vary and demonstrate strong dependency to the cell area, requiring normalization of intensities to the cell area. The normalized intensity ratio eliminates contribution of area to the scale of intensity, but still showing scattered data. The cell area may vary with cell division and as a biological response to Cd2+ ion exposure. Amount of Cd2+ ion uptake by individual cells may contribute to this fact but to measure that amount by analytical techniques at single cell level is rather difficult. However, using fluorescent Ca2+ ion indicators it may be feasible to estimate at least the amount of Cd2+ ions up taken by individual cells [11].
A closer inspection of Figure 4 reveals that there are heterogeneous mitochondrial responses, reduction in normalized intensity ratio and cell area, even after normalization of the intensity ratio. Regardless of cell type, the trend is similar in that increased Cd2+ ions exposure led to almost 10-fold reduction in the MMPs and cell areas. Figure 4 clearly demonstrates each cell individually responds to the stressor. Combined findings of ROS and MMPs suggest that cells are heterogeneously responding to the stressor, therefore assessing a cell’s biological responses at single cell level microscopically may help to reach detailed reliable conclusions, complementing next generation sequencing.
As an opinion, we suggest that microscopic assessment of individual cell responses in real time and living-cell conditions may decipher intra-cell and inter-cell dynamics of biological functions, and particularly relating mitochondrial dynamics to cell physiology. Furthermore, tracking locations and movements of individual cells and their correlation with single organelles may provide richer dynamical information.
References
- Choong G, Liu Y, Templeton DM (2014) Interplay of calcium and cadmium in mediating cadmium toxicity. Chem Biol Interact 211: 54-65.
- Meyer JN, Hartman JH, Mello DF (2018) Mitochondrial Toxicity. Toxicol Sci 162(1): 15-23.
- Joseph P (2009) Mechanisms of cadmium carcinogenesis. Toxicol Appl Pharmacol 238(3): 272-279.
- Chao SH, Suzuki Y, Zysk JR, Cheung WY (1984) Activation of calmodulin by various metal cations as a function of ionic radius. Mol Pharmacol 26(1): 75-82.
- Jacobson KB, Turner JE (1980) The interaction of cadmium and certain other metal ions with proteins and nucleic acids. Toxicology 16(1): 1-37.
- Cuypers A, Plusquin M, Remans T, Jozefczak M, Keunen E, et al. (2010) Cadmium stress: an oxidative challenge. Biometals 23(5): 927-940.
- Liu J, Qu W, Kadiiska MB (2009) Role of oxidative stress in cadmium toxicity and carcinogenesis. Toxicol Appl Pharmacol 238(3): 209-214.
- López E, Arce C, Oset Gasque MJ, Cañadas S, González MP (2006) Cadmium induces reactive oxygen species generation and lipid peroxidation in cortical neurons in culture. Free Radic Biol Med 40(6): 940-951.
- Casalino E, Sblano C, Landriscina C (1997) Enzyme activity alteration by cadmium administration to rats: the possibility of iron involvement in lipid peroxidation. Arch Biochem Biophys 346(2): 171-179.
- Dorta DJ, Leite S, DeMarco KC, Prado IM, Rodrigues T, et al. (2003) A proposed sequence of events for cadmium-induced mitochondrial impairment. J Inorg Biochem 97(3): 251-257.
- Hinkle PM, Shanshala ED 2nd, Nelson EJ (1992) Measurement of intracellular cadmium with fluorescent dyes. Further evidence for the role of calcium channels in cadmium uptake. J Biol Chem 267(35): 25553-25559.






























