Vitamin B12 Regulates the Expression of Methotrexate-Induced Increased Markers of Autophagy: An Immunohistochemical Study
Derya Karabulut1*, Nurhan Kuloğlu1,2, Emin Kaymak3, Emel Öztürk4, Ali Tuğrul Akın5, Meryem Sayan1 and Birkan Yakan1
1Department of Histology Embriology, Erciyes University, Turkey
2Department of Health Care Services Elderly Care, Niğde Ömer Halisdemir University, Turkey
3Department of Histology-Embriology, Bozok University, Turkey
4Department of Histology-Embriology, Harran University, Turkey
5Department of Biology, Erciyes University, Turkey
Submission:November 17, 2020; Published: January 19, 2021
*Corresponding author: Derya Karabulut, Department of Histology Embriology, Faculty of Medicine, Erciyes University, Kayseri, Turkey
How to cite this article: Derya K, Nurhan K, Emin K, Emel Ö, Ali Tuğrul A, et al. Vitamin B12 Regulates the Expression of Methotrexate-Induced Increased Markers of Autophagy: An Immunohistochemical Study. Open Acc J of Toxicol. 2021; 4(5):555648. DOI: 10.19080/OAJT.2021.04.555648.
Abstract
In this study, the immunohistochemical evaluation of autophagy in brain tissue in the presence and absence of folate was aimed. Therefore, Vitamin B12, a water-soluble folic acid, and Methotrexate (MTX), a folate antagonist, were used in the study. 28 rats were used in the study. Control group (n=7), Vitamin B12 group (n=7) 3μg/kg/ip B12 (during 15 days), MTX group (n=7) injected with 20mg/kg/ip MTX on 8th day of experiment, MTX+Vitamin B12 group (n=7) injected with a single dose of 20mg/kg ip MTX on 8th day of experiment + 3μg/kg/ip Vit B12 (15 days) per. After the histological procedure, microtubule-associated protein 1 Light Chain 3 (LC3B), microtubule-associated protein 1 Light Chain 3 (LC3AB) and glucose-regulated protein 78 (GRP78) immunohistochemistry staining were performed on the brain tissue. According to our results, all three protein expressions were observed in the cerebral cortex. Expressions of LC3AB, LC3B and GRP78 were statistically significantly increased in the MTX group compared to the control. Vitamin B12 administration showed a statistically significant improvement in the expression results of all proteins. Vitamin B12 to be used before MTX treatment may prevent abnormal autophagy in brain tissue.
Keywords: Autophagy; Endoplasmic reticulum stress; Methotrexate; Vitamin B12
Abbreviations: SAM: Adenosylmethionine; BBB: Blood-Brain Barrier; CNS: Central Nervous System; ER-Stress: Endoplasmic Reticulum-Stress; GRP78: Glucose-Regulated Protein 78; MXT: Methotrexate LC3: Microtubule-Associated Protein 1 Light Chain 3
Introduction
Vitamin B12 is a water-soluble vitamin produced by microorganisms in nature, which we take into our bodies through diets [1]. It is the cofactor of methionine synthase, which is involved in the synthesis of methionine, which is the precursor of S-Adenosylmethionine (SAM) [2]. Therefore, it plays an important role in DNA synthesis, erythropoiesis and healthy neurological functions [3,4]. Vitamin B12 deficiency is a common cause of macrocytic anemia, and also causes clinical disorders including peripheral and central neurological manifestations [3,5]. Therefore, bone marrow and nerve tissue are considered to be the organs most susceptible to vitamin deficiency [6]. Methotrexate (MTX) is a folate antagonist used for Central Nervous System (CNS) cancers [7]. However, the presence of the Blood-Brain Barrier (BBB) in the CNS causes the drug to be prevented from entering the brain [8]. Various doses are administered intravenously or intrathecally in order to obtain effective treatment [7]. MTX exerts its effect by limiting one-carbon fragments required for the synthesis of purines in DNA synthesis and cell proliferation and inhibiting dihydrofolate reductase [9]. It has been reported that MTX used for the CNS causes neuron degeneration in different parts of the brain [10]. MTX has been reported to induce apoptosis, which is programmed cell death in brain tissue [11,9]. There is limited information about the effects of MTX on autophagy, another programmed cell death in brain tissue. Autophagy is a highly complex physiological process that recycles cellular components. This process is important in maintaining cellular homeostasis in certain tissues such as the brain and liver muscle [12]. Autophagy can cause cell survival, as well as irregular autophagy can cause cell death. Autophagy occurs under the influence of the Autophagy-Related gene (Atg) family [13]. The yeast Atg8 homologue is rat microtubuleassociated protein 1 Light Chain 3 (LC3) [12]. Three subfamilies of LC3 (LC3A, LC3B and LC3C) have been identified in humans [14]. While autophagy occurs in the cell, first cytosolic LC3-I is produced in the cytoplasm, then transformed into LC3-II, which can localize to autophagosomal membranes [15]. Incorporation of LC3-II into the membrane is associated with Atg activation and the process ends with the formation of mature autophagosomes [12]. Thus, the amount of LC3-II correlates with the number of autophagosomes [16]. Therefore, LC3s are important in showing autophagy in tissue. Information about the relationship of MTX used in CNS cancers with autophagy is quite limited. In this study, we aimed to demonstrate immunohistochemically the role of autophagy in the changes that MTX will cause in brain tissue. In order to eliminate the inhibition of MTX on folate, we aimed to show immunohistochemically whether autophagy is affected by using Vitamin B12. In addition, we evaluated glucose-regulated protein 78 (GRP78) immunohistochemically in the tissue in order to evaluate the possible Endoplasmic Reticulum-Stress (ERstress) that will occur in the tissue if autophagy is affected.
Material And Methods
Animals
Twenty-eight adult male Wistar albino rats (2-3 months/ 8-12 weeks, weighing 200-300 g) was used from Erciyes University Experimental and Clinical Research Center (DEKAM), Kayseri, Turkey. Rats were housed in plastic cages in a well-ventilated rat house and allowed ad libitum access to food and water and kept at a 12-h light: dark cycle. All the animals received humane care according to the standard guidelines. Ethical approval for the study was obtained from Erciyes University Animal Research Local Ethics Committee and the ethic at regulations were followed in accordance with national and institutional guidelines (date 2018, decision no 18/116).
Chemicals
Methotrexate (MTX) (500mg/20ml, Koçak Farma, Turkey) and Vitamin B12 (Vit B12) (Vitamin B12 Rubranova, 5,000 μg; Bristol-Myers Squibb) was used as medicine.
Experimental design
In the study, 28 Wistar albino rats were divided into 4 equal groups. Control group (n=7) injected was applied İntraperitoneally (ip) saline during the experiment to this group. Vitamin B12 group (n=7) 3μg/kg/ip B12 (15 days) per day throughout the experiment. Methotrexate group (MTX) (n=7) injected with a single dose of 20 mg/kg/ip MTX on 8th day of experiment. Methotrexate+Vitamin B12 group (MTX+Vitamin B12) (n=7) injected with a single dose of 20mg/kg ip methotrexate on 8th day of experiment + 3μg/kg/ip Vit B12 (15 days) per day throughout the experiment.
Preparation of brain tissues
At the end of the experimental, the animals were killed by decapitation under ip ketamine (75mg/kg) + xylazine (10mg/ kg) anesthesia. The brain tissues were quickly removed and were fixed in 4% formaldehyde fixative for histological examination. Following dehydration (50%, 70, 80, 96, 3 times absolute alcohol) and clearing (xylene) embedded in parafin. Sections were stained with immunohistochemistry. Photographs were taken with an light microscope (Olympus BX51, Center Valley, PA, USA).
Immunohistochemistry procedure
Immunohistochemistry was applied to the brain tissue sections. The kit (TA-125-HDX, Thermo Fisher Scientific, Waltham, MA, USA) was used to determine the expression intensities of the antibodies. Antibodies are as follows: microtubule-associated protein 1 light chain 3 (LC3AB) (D3U4C, Cell Signaling, USA), microtubule-associated protein 1 Light Chain 3 (LC3B) (NB100-2220, Novus, USA) and glucose-regulated protein 78 (GRP78) (bs1219R, Bioss, USA). A citrate buffer was used for antigen recovery. Phosphate buffered saline (PBS) was preferred for washings. The sections were visualized using 3,30-p-diaminobenzidine tetrahydrochloride (TA-060-HDX, Thermo Fisher Scientific, Waltham, MA, USA) lightly counterstained with Gill hematoxylin. Under the light microscope (Olympus BX51, Center Valley, PA, USA) and images were obtained. Antibody’s immunoreactivities were measured with the Image j program [17].
Statistical analysis
All statistical analyses were carried out by using GraphPad Prism version 7.00 for Mac, GraphPad Software, La Jolla, California, USA. D’Agostino Pearson omnibus test was used to identify the normal distribution of the data. In the case of normal distribution, quantitative variables were compared using one-way analysis of variance and Tukey’s posthoc test. The data were expressed as the mean of normalized data±standard deviation of the mean. p<0.05 was considered as statistically significant.
Results
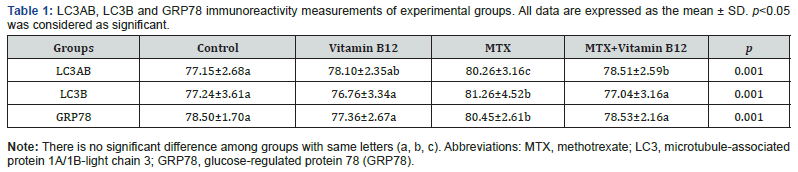
Evaluation of LC3AB and LC3B expressions LC3AB and LC3B expressions were observed in the brain tissue of all groups. Expression intensities were mostly in neuron cytoplasms located in the midline of the cerebral cortex. LC3AB expression showed a statistically significant increase in both the Vitamin B12 group and the MTX group compared to the control. LC3B expression was similar to control and Vitamin B12. It increased statistically significantly in the MTX group. In the MTX + Vitamin B12 group, it decreased, showing a result similar to the control. There was also a statistically significant difference between the vitamin B12 group and the MTX group. LC3AB expression decreased in the MTX + Vitamin B12 group, showing a similar result to the control. Between-group differences in LC3AB and LC3B expressions are shown in Table 1, Figure 1.
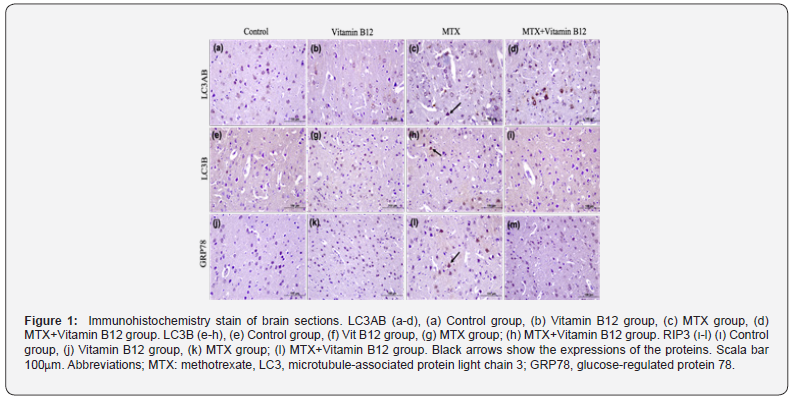
Evaluation of GRP78 expression
GRP78 expression was observed especially in the neuron cytoplasm with a pyramidal shaped cell body. There was no statistical difference between the control group and the Vitamin B12 group. There was a statistically significant increase in the MTX group compared to the control. The MTX + Vitamin B12 group was also similar to the control. GRP78 expression is shown in Table 1, Figure 1.
Discussion
MTX shows its effect on cells by inhibiting dihydrofolate reductase, indirectly inhibiting purine synthesis, by inhibiting both DNA synthesis and cell proliferation [18]. Because MTX is a folate antagonist. Intracellular folate deficiency indirectly causes anemia and nerve damage as vitamin B12 deficiency causes inhibition of both purine and pyrimidine synthesis [19]. In our study, we preferred MTX because of its effects on folate and evaluated autophagy in nerve tissue and related ER-stress. In order for the normal physiological process to continue, the cell performs many activities together and regularly. However, under adverse conditions, cell death mechanisms can be triggered. Apoptosis and autophagy are processes that play an important role in maintaining homeostasis. Autophagy, the mechanism of programmed cell death, is essential for normal development and cellular activity, and LC3 (also known as Atg8) is a marker for autophagosomes that occur in autophagy formation [20]. As long as the physiological process continues normally, autophagy is a process that only removes old and degraded cellular components. However, increased abnormal autophagy can also cause cell death. The main difficulty in using MTX in the treatment of brain tumors is the presence of a blood-brain barrier that does not allow tissue passage. For this reason, high doses, various drugs and methods have been developed to obtain the effective concentration of the drug in the tissue [8,21,22]. In our study, we evaluated the autophagy marker LC3 and ER-stress marker GRP78 in brain tissue by applying 20mg/kg MTX. Both LC3AB and LC3B expressions in brain tissue increased in MTX group. LC3 expressions were in the cell cytoplasm of neurons. Neurons are the cells of the nervous system that hear, interact, and transmit. Pyramidal neurons of the cerebral cortex are large cell bodied neurons with the same morphological prototype [23]. LC3 expressions were stained specifically in the cytoplasm of these neurons, not Betz cells. Cisplatin, a chemotherapeutic agent such as MTX, has been reported to up-regulate autophagy-related genes in the breast cancer cell line [20]. In a study in which neoadjuvant was used to increase the effect of MTX on Ehrlich acid carcinoma, it was reported that the autophagic gene beclin1, up-regulated by MTX, decreased after neoadjuvant drug use [24]. It has been reported that MTX increases LC3 expression in the spermatocyte cell line and is induced in apoptosis due to this increase [25]. In this study, we can say that the decreased folate concentration after MTX administration causes an increase in both LC3 expressions in the brain tissue. Because we obtained similar results to the control in both LC3 expressions in MTX + Vitamin B12 group, which we used to complement the reduced folate. In parallel with the increase in LC3 expressions in the MTX group, we also found an increase in GRP78 expression. GRP78, as one of the important chaperones on ER, participates in the ER protein folding and assembly process [26]. In response to stress conditions, GRP78 overexpression is a harbinger of the presence of large amounts of unfolded protein [27]. It has been reported that substances such as lead impair GRP78 function in the nervous system, increasing the unfolded protein response [28]. It has been reported that low doses of MTX cause a decrease in the number of apoptotic cells by reducing ERstress in vitro and in vivo [29]. On the contrary, it is also suggested that even its topical application promotes ER-stress mediated apoptosis [30]. We found that MTX increased GRP78 expression in brain tissue after 20mg/kg dose administration. This increase was parallel to LC3 expressions. We think that the MTX dose likely increases ER-stress in neurons, causing an increase in the amount of misfolded/unfolded protein, thereby increasing LC3 expressions in response to the cell in order to eliminate these accumulated proteins. These results are not only dependent on them, but also related to more genes and proteins.
The effect of the folate antagonist MTX on LC3 and GRP78 expressions was statistically significantly improved when a folate derivative Vitamin B12 was used. In this case, it can be concluded that regular methionine synthesis has an effect on both autophagy and ER-stress. Because unlike a folic acid antagonist MTX, Vitamin B12 is a water-soluble folic acid [31]. Exogenous completion of depleted endogenous Vitamin B12 indicates that it is necessary for the normal physiological process to continue in the tissue. There is very limited literature information on vitamin B12 and GRP78. Only in the diabetic retinopathy model, Vitamin B12 supplementation has been reported to reduce the various ERstress markers contained in GRP78 [32]. In addition, Vitamin B12 is very important for the nervous system, and neuronal degeneration occurs in its deficiency [33]. Vitamin B12 has also been shown to reverse impaired ER-stress and cellular autophagy after stroke [34]. In our study, we found improvement in autophagic marker LC3 expression and ER-stress marker GRP78 expression with Vitamin B12 administration. This result should not be ignored in order to prevent possible abnormal autophagy.
Conclusion
In study, we demonstrated the autophagy in the brain tissue immunohistochemically using MTX and Vitamin B12. In addition, increased GRP78 expression showed us that increased autophagy can also trigger ER-stress. We think that autophagy and ERstress should not necessarily be ignored in studies on improving the effect of MTX on CNS cancers. In addition, we believe that Vitamin B12 should be essential supplementary food for all drugadministered diseases that may cause folate deficiency.
Acknowledgement
This work was supported by Erciyes University the Scientific Research Projects Unit, TSA-2019-8673 project code.
References
- Romain M, Sviri S, Linton DM, Stav I, Van Heerden PV (2016) The role of Vitamin B12 in the critically ill--a review. Anaesth Intensive Care 44(4):447-452.
- Guéant JL, CaillerezFofou M, BattagliaHsu S, Alberto JM, Freund JN, et al. (2013) Molecular and cellular effects of vitamin B12 in brain, myocardium and liver through its role as co-factor of methionine synthase. Biochimie 95(5):1033-1040.
- Oh R,Brown DL (2003) Vitamin B12 deficiency. Am Fam Physician 67(5):979-986.
- GreibeE, Kornerup LS, Juul CB, Fedosov SN, Heegaard CW, et al. (2018) The tissue profile of metabolically active coenzyme forms of vitamin B12 differs in vitamin B12-depleted rats treated with hydroxo-B12 or cyano-B12. Br J Nutr 120(1):49-56.
- GuillandJC,AimoneGastin I (2013) [Vitamin B12 (cobalamin)]. Rev Prat 63(8):1089-1090.
- Birn H, Nexø E, Christensen EI,Nielsen R (2003) Diversity in rat tissue accumulation of vitamin B12 supports a distinct role for the kidney in vitamin B12 homeostasis. Nephrol Dial Transplant 18(6):1095-1100.
- Ogushi N, Sasaki K,Shimoda M (2017) CAN a P-gp modulator assist in the control of methotrexate concentrations in the rat brain? -inhibitory effects of rhodamine 123, a specific substrate for P-gp, on methotrexate excretion from the rat brain and its optimal route of administration. J Vet Med Sci 79(2):320-327.
- Ogushi N, Sasaki K,Shimoda M (2015) Effect of cyclosporin on distribution of methotrexate into the brain of rats. J Vet Med Sci 77(9):1171-1173.
- Sun J, Sugiyama A, Inoue S, Takeuchi T,Furukawa S (2014) Effect of methotrexate on neuroepithelium in the rat fetal brain. J Vet Med Sci 76(3):347-354.
- Moore IM, Merkle CJ, Byrne H, Ross A, Hawkins AM, et al. (2016) Effects of Intraventricular Methotrexate on Neuronal Injury and Gene Expression in a Rat Model: Findings From an Exploratory Study. Biol Res Nurs 18(5):505-514.
- Hirako A, Furukawa S, Takeuchi T,Sugiyama A (2016) Effect of methotrexate exposure at late gestation on development of telencephalon in rat fetal brain. J Vet Med Sci 78(2):213-220.
- Russo M,Russo GL (2018) Autophagy inducers in cancer. Biochem Pharmacol 153:51-61.
- Onorati AV, Dyczynski M, Ojha R,Amaravadi RK (2018) Targeting autophagy in cancer. Cancer 124(16):3307-3318.
- He H, Dang Y, Dai F, Guo Z, Wu J, et al. (2003) Post-translational modifications of three members of the human MAP1LC3 family and detection of a novel type of modification for MAP1LC3B. J Biol Chem 278(31):29278-29287.
- Kabeya Y, Mizushima N, Yamamoto A, OshitaniOkamoto S, Ohsumi Y, et al. (2004) LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci 117(Pt 13):2805-2812.
- Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, et al. (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. Embo J 19(21):5720-5728.
- Öztürk E, Kaymak E, Akin AT, Karabulut D, Ünsal HM, et al. (2020) Thymoquinone is a protective agent that reduces the negative effects of doxorubicin in rat testis. Hum Exp Toxicol 39(10): 1364-1373.
- Sugiyama A, Sun J, Ueda K, Furukawa S,Takeuchi T (2015) Effect of methotrexate on cerebellar development in infant rats. J Vet Med Sci 77(7):789-797.
- Scott J,Weir D (1994) Folate/vitamin B12 inter-relationships. Essays Biochem 28:63-72.
- Shen M, Duan WM, Wu MY, Wang WJ, Liu L, et al. (2015) Participation of autophagy in the cytotoxicity against breast cancer cells by cisplatin. Oncol Rep 34(1):359-367.
- Wang X, Liu P, Yang W, Li L, Li P, et al. (2014) Microbubbles coupled to methotrexate-loaded liposomes for ultrasound-mediated delivery of methotrexate across the blood-brain barrier. Int J Nanomedicine 9:4899-4909.
- Sane R, Wu SP, Zhang R,Gallo JM (2014) The effect of ABCG2 and ABCC4 on the pharmacokinetics of methotrexate in the brain. Drug Metab Dispos 42(4):537-540.
- Brat DJ (2018) Normal brain histopathology. In:Practical surgical neuropathology: a diagnostic approach. Elsevier pp.19-37.
- ElMagd MA, Khamis A, Nasr Eldeen SK, Ibrahim WM,Salama AF (2017) Trehalose enhances the antitumor potential of methotrexate against mice bearing Ehrlich ascites carcinoma. Biomed Pharmacother 92:870-878.
- Xiong S, Song D, Xiang Y, Li Y, Zhong Y, et al. (2020) Reactive oxygen species, not Ca2+, mediates methotrexate-induced autophagy and apoptosis in spermatocyte cell line. Basic Clin Pharmacol Toxicol 126(2):144-152.
- Wu YY, Yang YP,Li ZQ (2009) [Glucose regulated protein 78 kD]. Sheng Li Ke Xue Jin Zhan 40(2):135-141.
- Ibrahim IM, Abdelmalek DH,Elfiky AA (2019) GRP78: A cell's response to stress. Life Sci 226:156-163.
- Qian Y,TiffanyCastiglioni E (2003) Lead-induced endoplasmic reticulum (ER) stress responses in the nervous system. Neurochem Res 28(1):153-162.
- Rong F, Gao X, Liu K,Wu J (2018) Methotrexate remediates spinal cord injury in vivo and in vitro via suppression of endoplasmic reticulum stress-induced apoptosis. Exp Ther Med 15(5):4191-4198.
- Chen H, Yan L, Wang J, Sun Y, Li X, et al. (2017) Methotrexate prevents epidural fibrosis through endoplasmic reticulum stress signalling pathway. Eur J Pharmacol 796:131-138.
- Gisondi P, Fantuzzi F, Malerba M,Girolomoni G (2007) Folic acid in general medicine and dermatology. J Dermatolog Treat 18(3):138-146.
- Reddy SS, Prabhakar YK, Kumar CU, Reddy PY,Reddy GB (2020) Effect of vitamin B12 supplementation on retinal lesions in diabetic rats. Mol Vis 26:311-325
- Adachi K, Izumi M, Osano Y, Miura N, Takatsu S, et al. (2003) Polyamine concentrations in the brain of vitamin B12-deficient rats. Exp Biol Med (Maywood) 228(9):1069-1071.
- Tripathi M, Zhang CW, Singh BK, Sinha RA, Moe KT, et al. (2016) Hyperhomocysteinemia causes ER stress and impaired autophagy that is reversed by Vitamin B supplementation. Cell Death Dis 7(12):e2513.






























