Fulminant hepatic failure induced by antipsychotic drugs (a case report)
Raja A, Ezzouine H*, Mediouni K and Elabbasi R
Département of anesthésia ressuscitation, Médical emergency ressuscitation service, University Hospital of Casablanca, Hassan II University, Morocco
Submission:July 19, 2020; Published: July 31, 2020
*Corresponding author: Ezzouine H, Médical emergency ressuscitation service, University Hospital of Casablanca, Hassan II University, Faculty of Medicine and Pharmacy, Casablanca Morocco
How to cite this article: Raja A, Ezzouine H, Mediouni K, Elabbasi R. Fulminant hepatic failure induced by antipsychotic drugs (a case report). Open Acc J of Toxicol. 2020; 4(3):555638. DOI: 10.19080/OAJT.2020.04.555638.
Abstract
We report a case of fulminant hepatitis following triple neuroleptic poisoning complicated by multi-visceral failure, in a 16-year-old patient who presented a table associating a fever, asthenia, myalgia, intense mucocutaneous jaundice with incoercible vomiting following 3 neuroleptics. The assessment showed acute hepatic insufficiency (cholestasis, TP and factor V very collapsed). Despite initial management combining PFC transfusion and laxatives, the course was initially unfavorable, with a worsening of its hemostasis and hyperbilirubinemia. The administration of N-acetylcysteine for 48 hours associated with the infusion of PFC was followed by a rapid clinical and biological improvement. The modalities of use of NAC during acute IHC not related to paracetamol remains to be discussed.
Keywords: Acute liver failure; neuroleptics; N-acetylcysteine; Toxin-related hepatotoxicity
Introduction
Fulminant hepatitis is a rare condition but has a very poor prognosis in the absence of liver transplantation. It is important to identify the cause as soon as possible to start the etiological treatment, which may be drug poisoning, viral hepatitis, or alcoholic ... [1-3]. N-acetylcysteine (NAC), a glutathione precursor, was first used as a treatment for paracetamol overdose in 1979. Since then, it has been firmly established as an effective and safe treatment for paracetamol induced IHC prevention. NAC has also been shown to be effective outside paracetamol intoxication. It has been evaluated as an option for acute IHC other than paracetamol in adults and children. In a randomized clinical trial comparing NAC with placebo in adults with paracetamol-free IHC, NAC was associated with a marked improvement in survival without liver transplantation [4]. NAC has also been evaluated for non-hepatic clinical conditions, these indications include its use in lung diseases (COPD and pulmonary fibrosis), in the prevention of contrast-induced nephrotoxicity and for the treatment of certain cardiac diseases [3,5]. In this article, we report the case of a neuroleptic overdose in a 16-year-old who has rapidly progressed to fulminant hepatitis. The oral administration of N-acetylcysteine has allowed a dramatic improvement [3].
Observation
A 16-year-old man consulted at the emergency reception service (j1) for asthenia, disabling diffuse myalgia and uncontrollable vomiting, in the context of cutaneous-mucous jaundice, which had appeared for 4 days. Her antecedents included herpetic keratitis since the age of 3 years and chronic epigastralgia for 4 years. In the emergency department, the clinical examination found a sleepy, sleepy patient, a conjunctival subitem, muscle pain with manual pressure. The temperature was 37.8°C, and blood glucose was 2.2mmol/l. The blood pressure was 85/38 mmHg, the heart rate was 104 beats per minute and 95% saturation in the air. The biochemical assessment was very disturbed, with: an inflammatory syndrome (C-reactive protein at 150 mg/L, fibrinogen at 6.6g/L), renal insufficiency (urea at 14.4mmol/L, creatinine at 159 , 12mmol/L), cytolysis (ASAT: 10454 IU/L, ALA at 4408 IU/L), cholestasis (conjugated bilirubinemia at 151mmol/L, gamma GT at 489 IU / L, PAL at 657 IU/L ) and a disturbance of its hemostasis (TP <15%, a TCA at 96.4 s and a very collapsed factor V). The hemogram showed the following: white blood cells at 9210mm-3, platelets at 159 000mm-3 and hemoglobin at 11.2g/dl.
On the diagnosis side
The patient was admitted to intensive care (1st day). Etiologically, an infectious hypothesis has been ruled out by the negativity of his liver serologies (anti-HVA Ab, anti-HBV Ab and HBs antigen and anti-HCV antibodies) as well as the serology of CMV, the abdominal ultrasound was without particularities, blood ceruloplasmin level was normal, anti-smooth muscle and antimitochondrial antibodies were achieved returning normal. The preferred toxic hypothesis was a neuroleptic overdose because the interview reported a prescription of 3 different neuroleptics, by his doctor for his chronic epigastralgia of psychogenic origin (Olanzapine, Mainspring and Metoclopramide). Supported: Symptomatic treatment consisted of stopping neuroleptics, infusion of fresh frozen plasma, vitamin K, laxatives and ciprofloxacin were initiated associated with administration of N-acetylcysteine with a dose of oral load of 140mg/kg followed by a dose of 70mg/kg/day maintenance for 48 hours.
Evolution
The evolution was quickly favorable. Hyper-bilirubinemia was divided by five in three days, the state of consciousness improved rapidly with appearance of an asterixis, the correction of the hemostasis disorder was more progressive with normalization towards the 4th day (Figure 1), a dramatic improvement in hepatic transaminases was observed as early as the second day (Figure 2). The patient left the intensive care unit to the gastric department on day 6. The symptomatic treatment was continued until day 10, the PBH was performed on day 11 without abnormality then he left the hospital on day 17.
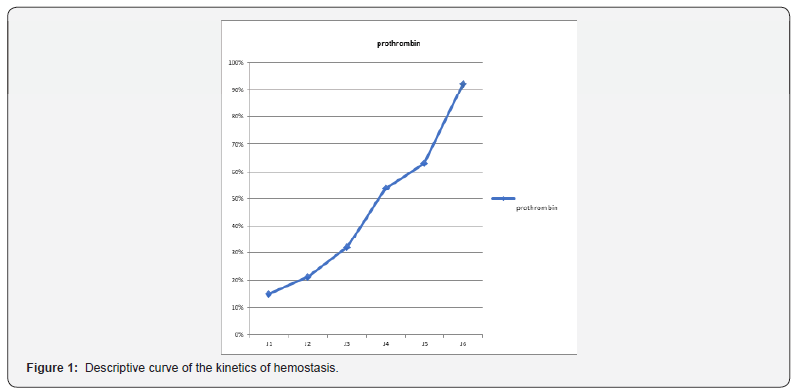
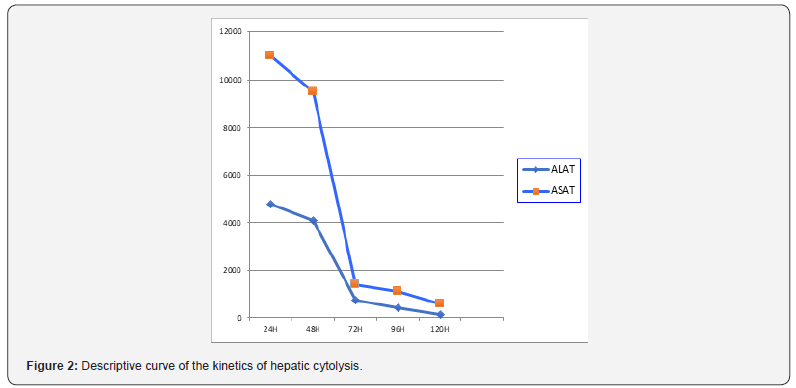
Discussion
IHA is defined as a sudden failure of liver function in a patient with no history of liver disease. The cardinal signs of hepatic failure include coagulopathies and hepatic encephalopathy of any grade in the context of acute liver injury [4]. Currently, there is no scientifically proven beneficial treatment in the treatment of IHC, apart from liver transplantation Lee [5]. More than 1000 drugs have been listed as being responsible of hepatic side effects; 16% of these agents were neuropsychiatric drugs. Antidepressant drugs (tricyclic agents or SSRI), mood stabilizing agents and neuroleptic drugs have been implicated in biological or/and clinical hepatotoxicity. For these reasons, some psychotropic agents have been withdrawn of the pharmaceutical, On the contrary, in case of clinical hepatotoxicity, challenge or maintenance is absolutely inadvisable. Mechanism of the hepatic troubles: precise mechanisms of the hepatotoxicity remain unclear. Contrary to phenothiazine drugs, no information is available on the respective rule of the agents and their metabolites. Hypersensitivity syndrome or eosinophilia has been reported, suggesting a possible immuno-allergic mechanism. Presence of risk factors: risk factors have been retrieved, in some observations, like high daily dosage, high plasmatic concentration, age, alcoholism, obesity or antecedent of hepatic disorders like Gilbert syndrome. [6] Special care is advisable with these patients. As hepatotoxicity has been observed after surd Osage (or suicide attempt), a hepatic check-up has to be performed in these clinical situations [7]. Co-medication with hepatotoxic drugs may increase the risk as it has been suggested. Acetylcysteine is a precursor of glutathione. It is well known as an antidote for acetaminophen overdose due to its ability to increase glutathione levels, which inactivates the toxic metabolite of acetaminophen [8]. N-acetyl-pbenzoquinone mine. Glutathione is a major antioxidant that can serve as a scavenger for free radicals; therefore, acetylcysteine may increase glutathione stock during periods of oxidative stress, increase nitric oxide production, which causes vasodilation and therefore tissue oxygenation, and may also have an antiinflammatory effect. by inhibition of pro-inflammatory factors (TNF alfa and IL8) [9,10]. The majority of studies evaluated the use of NAC in acute IHC secondary to acetaminophen poisoning. There is little research on the use of NAC in IHC secondary to other causes. Hu [8]. evaluated the efficacy of NAC in patients with non-acetaminophen-overdosed IHC (safety and efficacy of NAC in patients with ALF not caused by acetaminophen overdose), in a meta-analysis, which consisted of analyzing four assays prospective clinical trials evaluating NAC versus placebo in the treatment of non-acetaminophen-induced IHC[3].
Conclusion
NAC is a beneficial treatment in the context of nonparacetamol induced IHC, it can prolong the survival of patients with or without liver transplantation and survival after transplantation, but it cannot improve overall survival. Therefore, due to the lack of available scientific evidence, current data is unable to conclusively determine the role of NAC in patients with IHC without paracetamol. Thus, they are unable to make recommendations for clinical practice.
Disclosure of interest
The authors declare that they have no conflicts of interest concerning this article.
References
- Ostapowicz G, Fontana RJ, Schiødt FV, Larson A, Davern TJ, et al. (2002) Results of a prospective study of acute liverfailure at 17 tertiary care centers in the United States. Ann Intern Med 137(12): 947-954.
- 2 Lee WM, Hynan LS, Rossaro L, Fontana RJ, Stravitz RT, et al. (2009) Intravenous N-acetylcysteine improves transplant-free survival in early stage non-acetaminophen acute liverfailure. Gastroenterology 137(3): 856-864.
- 3.Stravitz RT, Sanyal AJ, Reisch J, Bajaj JS, Mirshahi F, et al. (2013) Acute effects of N-acetylcysteine on cytokinesin non-acetaminophen acute liver failure: potential mech-anism of improvement in transplant-free survival. Liver Int 33(9): 1324-1331.
- Squires RH, Dhawan A, Alonso E, Narkewicz MR, ShneiderBL, et al. (2013) Intravenous N-acetylcysteinein pediatric patients with nonacetaminophen acute liverfailure: a placebo-controlled clinical trial. Hepatology 57(4): 1542-1549.
- 5. Singh S, Hynan LS, Lee WM (2013) Improvements in hepatic serolog-ical biomarkers are associated with clinical benefit of intra-venous N-acetylcysteine in early stage non-acetaminophenacute liver failure. Dig Dis Sci 58(5): 1397-1402.
- 6. Mouton JP, Mehta U, Parrish AG, Wilson DP, Stewart A, et al. (2015) Mortality from adverse drug reactions in adult medical inpatients at four hospitals in South Africa: a cross-sectional survey. Br J Clin Pharmacol 80(4): 818-826.7. Björnsson ES, Bergmann OM, Björnsson HK, Kvaran RB, Olafsson S (2013) Incidence, presentation, and outcomes in patients with druginduced liver injury in the general population of Iceland. Gastroenterology 144(7): 1419-1425.8. Hu J, Zhang Q, Ren X, Sun Z, Quan Q (2015) Efficacy and safety of acetylcysteine in “nonacetaminophen” acute liver failure: A meta-analysis of prospective clinical trials. Clinics and Research in Hepatology and Gastroenterology 39(5): 594-599.9. Kortsalioudaki C, Taylor RM, Cheeseman P, Bansal S, Mieli-Vergani G, et al. (2008) Safety and efficacy of N-acetylcysteine in children with non-acetaminophen induced acute liver failure. Liver Transplantation 14(1): 25-30.10. Vilstrup H, Amodio P, Bajaj J, Cordoba J, Ferenci P, et al. (2014) Hepatic encephalopathy in chronic liver disease: 2014 practice guideline by the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver. Hepatology 60(2): 715-735.






























