Should a Toxicological Risk Assessment of Ubiquitous Chemicals Be Done Using Data from Only One Source of Exposure?
Di Nardo JC* and CA Downs
1 Retired Toxicologist, Vesuvius, USA
2 Executive Director, Haereticus Environmental Laboratory, Clifford, USA
Submission: February 12, 2020; Published: February 27, 2020
*Corresponding author: Di Nardo JC, Retired Toxicologist, Vesuvius, USA
How to cite this article: Di Nardo JC, CA Downs. Should a Toxicological Risk Assessment of Ubiquitous Chemicals Be Done Using Data from Only One Source of Exposure?. Open Acc J of Toxicol. 2020; 4(2):555633. DOI: 10.19080/OAJT.2020.04.555634.
Abstract
Keywords: Toxicological; Chemicals; Dioxybenzone; Octocrylene; Oxybenzone; Sulisobenzone
Introduction
After much discussion and petitioning by several individuals, non-governmental organizations and scientists alike, the Food & Drug Administration (FDA) re-opened the sunscreen drug monograph on February 26, 2019 to review the safety of sunscreen actives considered Generally Recognized as Safe and Effective (GRASE) since 1978 [1]. After their review, the FDA concluded that the public record “does not” currently contain sufficient data to support the safety of a dozen drug actives currently in use (several of which are benzophenone based chemicals - avobenzone, dioxybenzone, octocrylene, oxybenzone and sulisobenzone) and, therefore, are requesting industry to provide additional data (mainly toxicokinetics, carcinogenicity and developmental reproductive toxicology) to substantiate GRASE status. Since that time, industry has responded in a typical manner suggesting that much of this data can be generated and/or already exists but will require an additional 2 to 3 years to provide the information.
With that said, concerns that a political and not a scientific decision will be made to support the $10 billion in annual sales made by the sunscreen industry as well as protect other industries that also use these materials (inks, plastics, cardboard, medications … etc.). One major point of contention is how data is manipulated or ignored to demonstrate human safety especially when governments have varying opinions. Case in point would be the carcinogenic effect of the chemical benzophenone being used as a food additive or a plasticizer in plastic food wraps. The FDA decided to ban benzophenone in only foods and the European Union (EU) decided to rely on their previous risk assessment to re-conclude that the chemical is not of concern when added to foods. Neither group mentions exposure to the carcinogen in any other consumer product(s), possibly misleading consumers to believe that there is no further risk associated with the chemical.
Data
Additionally, in September 2017 the EU reapproved the existing benzophenone levels as safe for chronic dietary exposure of adults and children in foods, which was set at 10 – 20 ug/Kg and remarked that a Tolerable Daily Intake (TDI) of 0.03 mg/Kg body weight for benzophenone is appropriate to cover the non-neoplastic effects in the chronic toxicity studies and the neoplastic effects induced in the rodent carcinogenicity studies [5].
Issue
Analytical testing (HPLC method) of a 3 ounce bottle of sunscreen containing 10% octocrylene and 6% oxybenzone identified the chemical benzophenone at a level of 75.95 ppm (mg/Kg) or a total available dose of 75,950 ug/Kg (1,000 times higher than the daily exposure level per Kg believed to be from foods).
Problem
Applies to most SPF 70’s – 100’s, but exposure to any combination of these actives are concerning
a) 3 oz bottle of sunscreen containing 10% octocrylene and 6% oxybenzone yielded 75.95 ppm (mg/Kg) of benzophenone. If 1 oz is applied after every 80 minutes of sweating or swimming the bottle would last 4 hours OR If 1 oz is applied every 120 minutes of sitting on the beach or at the pool without sweating or swimming one bottle would last 6 hours. Therefore, the following supposition represents the benzophenone exposure to just “one” bottle of product for just “one” day at the beach/pool.
b) One bottle of sunscreen contained 75.95 mg, if we divide that, like the FDA did, by 60 Kg that would give us a dose of approximately 1.27 mg/Kg/day of benzophenone.
c) Considering what is absorbed into the body would yield 1.27 mg/Kg/day * 10% Absorption (Worse Case Scenario) or 0.127 mg/Kg/day of benzophenone; which is the same as saying 127 ug/Kg/day.
d) Therefore, after only one day of sunscreen application at the beach or pool, exposure to benzophenone is 127 ug/Kg/day or “169 times more” than the FDA exposure level that established the ban on benzophenone in foods and food wraps or “6 to 12 times greater” than the EU exposure level thought to have an adequate safety margin or “4 times higher” than the TDI which was thought to cover the non-neoplastic and neoplastic effects found in the animal study.
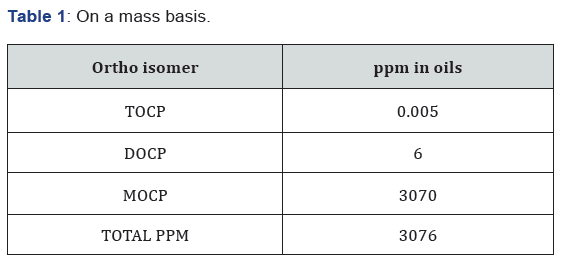
Additionally, this calculation does not include benzophenone exposure from other sources like canned, aqua- cultured or wild caught fish (see table 1 below); wastewater treatment systems that cannot remove these chemicals from the water and, therefore, returns in the tap water [6], printing inks, cardboard, plastic bottles/jars, dentures and even emissions from residential oil burners, via the atmospheric oxidation of diphenylmethane, can form benzophenone that can be inhaled [7] non-steroidal anti-inflammatory drugs like Ketoprofen are benzophenone based, cross-reacting with other benzophenone chemicals causing atopic dermatitis [8] and lastly, numerous personal care and household products also contain benzophenones with the former listing such chemicals in the ingredient label unfortunately most household products are not ingredient labeled, so there is no way to avoid exposure based on product(s) used.
Conclusion
We eat it, drink it, breathe it and rub it all over our body … it is time to stop under estimating and guessing what many industries expose us to by looking at only one source of exposure (i.e.: foods), especially when it comes to chemicals that are carcinogenic and/or endocrine disrupting pseudo-Persistent Organic Pollutants like benzophenone based chemicals. Scientists when testing chemicals, more times than not, incorrectly conclude that the “effects observed” or the “levels of exposure” are “far below toxicological reference values” based on only one point of exposure. This short-sighted logic is like trying to cross a two-way multi-lane superhighway during rush hour but insisting that one only needs to look to the left to cross safely … not a very wise or prudent decision! Regardless, we accept the conclusion that all is well and that the “risk outweighs the benefits” without truly evaluating the cost of what we are doing to the environment and ourselves [9] (Table 1).
References
- (2019) Sunscreen Drug Products for Over-the Counter Human Use. Food and Drug Administration 84: 38.
- Judith Kidwell (2018) Center for Food Safety and Applied Nutrition (HFS-265), Food and Drug Administration. USA.
- Rhodes MC, Bucher JR, Peckham JC, Kissling GE, Hejtmancik MR (2007) Carcinogenesis studies of benzophenone in rats and mice. Food Chem Toxicol 45(5): 843-851.
- https://oehha.ca.gov/proposition-65/chemicals/benzophenone
- Vittorio Silano (2017) Safety of benzophenone to be used as flavouring. Efsa journal 15(11): 5013.
- Díaz-Cruz MS, Gago-Ferrero P, Llorca M, Barceló D (2012) Analysis of UV filters in tap water and other clean waters in Spain. Anal Bioanal Chem 402(7): 2325-233.
- http://inchem.org/documents/iarc/iarcmono/v101iarc.pdf
- Devleeschouwer V, Roelandts R, Garmyn M, Goossens A (2008) Allergic and photoallergic contact dermatitis from ketoprofen: results of (photo) patch testing and follow-up of 42 patients. Contact Dermatitis 58(3): 159-666.
- Cunha et al (2018) UV-filters and musk fragrances in seafood commercialized in Europe Union: Occurrence, risk and exposure assessment. Environ Res 161: 399-408.






























