Age Related Heavy Metal Accumulation in Sediment and Mangrove Roots in the Niger Delta Coastal Fringes, Nigeria
Miebaka Moslen*, Cynthia Nwoha, John Onwuteaka
Department of Animal and Environmental Biology, Rivers State University, Port Harcourt, Nigeria
Submission:December 2, 2019; Published: January 13, 2020
*Corresponding author: Miebaka Moslen, Department of Animal and Environmental Biology, Rivers State University, Port Harcourt, Nigeria
How to cite this article: Miebaka M, Cynthia N, John O. Age Related Heavy Metal Accumulation in Sediment and Mangrove Roots in the Niger Delta Coastal Fringes, Nigeria. Open Acc J of Toxicol. 2020; 4(2):555632. DOI: 10.19080/OAJT.2020.04.555632.
Abstract
The aim of this study was to determine the concentration of heavy metals (Ni, Pb and Cd) in sediments, root of mangrove plants and relate it to the age of plant root. Three sites (St.1, St.2 and St.3) on the coastal fringes of Niger Delta mangrove were established. Composite samples of sediment and respective plant (Rhizophora and Avicennia) roots were collected for analysis. Heavy metal content of sediment and root were analysed using atomic absorption spectrophotometry while the age of root was assessed using rings/growth zones under microscope. Maximum concentrations (μgg-1) of Pb, Ni and Cd in sediments were 7.3, 7.1 and 0.6 respectively. Avicennia roots were found to concentrate higher level of metals (μg/g) (Pb: 8.7 Ni: 6.6 and Cd:1.1). General spatial order of heavy metal contamination was St.3>St.1>St.2 attributed to differential anthropogenic activities. Highest age of plant root (years) examined was in the order Avicennia (7.3)>Rhizophora (4.3). Spatial and temporal variations were significant (p<0.05) for some metals in sediment and roots examined. The level of Ni in sediments around Avicennia had significant positive correlation (p<0.01) with accumulations of Ni in the root. However, level of Cd in root of Rhizophora had a significant negative relationship with the age of the root while the reverse was the case of Avicennia with respect to Pb accumulation. Comparatively, metal levels in this study were within and also higher than WHO/FAO standards suggesting incremental potentials. The study concluded that different plant roots contain different levels of heavy metals irrespective of the concentrations in surrounding sediment. Avicennia roots accumulated more metals with increase in surrounding sediment and age of root. Correlation further indicated Avicennia has better potential for phytoremediation over Rhizophora in order to minimize metal bioaccumulation and toxicity. Observed discrepancies in metal concentration with respect to study site was mainly human factor induced but specific to each station studied.
Keywords: Heavy metal contaminants; Bioaccumulation; Sediment; Mangrove Root; Age; Niger Delta.
Introduction
Globally, marine and coastal ecosystems continue to be subjected to heavy metal pollution from municipal wastes, runoffs from agricultural and industrial sources [1-8] thereby, making coastal vegetations have a key function of trapping and storing pollutants [9-11]. The Niger Delta region remains a major concern in terms of heavy metal and hydrocarbon pollution due to increasing anthropogenic activities [12,13]. Accumulation of heavy metals in natural ecosystems is a threat to biodiversity and human health because of their persistence and toxicity [14-17] due to bioaccumulation and biomagnification effects [18,19]. The mangrove environment is very sensitive and vulnerable to pollution in view of its rich flora and fauna community. Mangroves are one of the most biologically important and productive ecosystems in the world [20]. MacFarlane reported that mangrove forests serve a key function of primary production in estuarine ecosystems and are an essential habitat for a wide variety of species such as birds, insects, mammals and reptiles [21,22].The proximity of mangroves to urban centers make them recipients of heavy metal contamination [23,24]. However, Mackey AP et al. [25] stated that mangroves are poor indicators of trace metals but found that large amounts of heavy metals are found in mangrove soils while few are found in plant tissues [26]. It is pertinent to state that even at low concentrations, heavy metals are poisonous due to bioaccumulation [27,28]. Mangrove forests are found in 118 countries around the globe with Nigeria’s Niger Delta area having the largest in Africa and fourth largest in the world in the order Indonesia>Brazil>Australia>Nigeria [29,30].
Biologically, six mangrove species make up these forests, three species in the family Rhizophoraceae (Rhizophora racemosa (red mangrove; tall), Rhizophora harrisonii (red mangrove; dwarf), Rhizophora mangle (red mangrove; dwarf)), and species in the family Avicenniaceae (white mangrove) and Combretaceae [31]. The mangrove forest of the Niger Delta is fast being depleted partly by Nypa palm invasion and wholly due to urbanization and industrialization. Accumulation of contaminants especially heavy metals occur in the roots but restricts its translocation to aerial portions of the plant hence less amount of heavy metals are found in the leaf compared to stem and root [26,32,33]. Heavy metals are not degradable but accumulate in plant tissues from soil which could cause long-term damage to plants particularly for mangrove soil with small grain size capable of accumulating such contaminants [34-36]. Most studies in the Niger Delta area had focused on the concentration of heavy metals in mangrove root and soil. The aim of this study was to assess the concentration of heavy metals (Pb, Cd and Ni) in the sediment and roots of two species of mangrove plants (Rhizophora, and Avicennia) in relation to the age of the plant root examined.
Materials and Methods
Site description
The study was conducted in mangrove forest areas on the coastal fringes of the Bonny estuary in the Niger Delta. The study areas included st.1: (Eagle island), St.2: (Bundu Ama) and St.3 (Borikiri) all in the southern Niger Delta region of Nigeria (Figure 1). The vegetation in the study area is mangrove with a mix of Avicennia, Rhizophora, Laguncularia and Nypa fruticans with Rhizophora as most dominant. The scanty mangrove trees in the area were irregularly disturbed with most at the fringes appearing under regenerative conditions of young age. Anthropogenic activities such as dredging, metal fabrication/maintenance works, oil servicing company activities, illegal oil bunkering activities and discharge of wastewater into nearby creeks characterized the study area.
Sample Collection
Mangrove Root Samples
Six (6) mangrove root samples were collected on a monthly basis for six months (December 2017 - May 2018). Samples for heavy metal determination were composited from triplicate samples to enhance wider coverage. Mangrove root (Rhizophora, Avicennia) samples were collected with sharp stainless-steel knife from three different sites studied. The plant roots were carefully collected from the part above the soil and divided into two parts. One part of each root sample was used to determine heavy metal concentration in roots while the corresponding part was used for age determination. The samples were properly labeled and immediately taken to the laboratory for analysis.
Sediment
Sediment samples were collected from the same stations as the root samples using soil/sediment sugar. Three spots around the root were sampled and composited. Samples were collected close to the root of the mangrove plant in order to correlate heavy metal content in both sediment and root samples. Sediment samples were put in properly labeled polythene bag and taken to the laboratory for analysis. All samples were preserved in mobile coolers while in transit.
Laboratory Analysis
Plant root samples were dried, grinded and digested with HCl/ HNO3 using the method of the American Society for Testing and Materials [37]. The concentration of heavy metals in plant root was determined with an Atomic Absorption Spectrophotometer (GB Avanta PM AAS, S/N A6600). Metal concentration was blank corrected and expressed as μgg-1 dry weight of sample for quality control.
Sediment
Samples were wrapped in properly labeled aluminum foils and put in ice coolers before taken to the laboratory for analysis. One (1g) of air-dried sediment was digested with Equia-Regia (mixture of HCI and HNO3 in the ratio of 3:1). The digested sediment samples were filtered with 20 ml of de-ionized water and the filtrates were stored in clean acid- washed and appropriately labeled 30 ml sample containers. Heavy metal analysis was done using Atomic Absorption Spectrophotometer.
Estimation of Age of Mangrove Root
A section of the mangrove root used for heavy metal analysis was air dried and surface polished to enable visualization of growth zones/ring bands. Triplicate samples were examined to obtain average age. Macroscopic and microscopic observations were made and ring-like formations (concentric circles) counted to estimate age of root. This is similar to methods used by other researchers to determine age of mangrove plants [38,39].
Data Analysis
Analysis of variance (ANOVA - General Linear Model) was used to test significant difference in metal concentrations across stations and also between the months of study. Tukey test was used for posting hoc analysis. Pearson correlation coefficient was used to determine relationship between metal concentration in root, sediment and age of plant root. The software Minitab 16 was used for the statistical analysis.
Results and Discussion
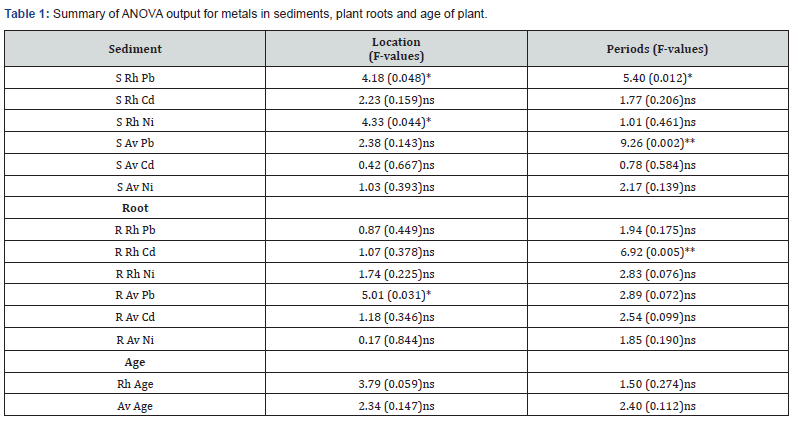
The concentration of heavy metals in sediments, roots of mangrove plants (Rhizophora-Rh and Avicennia- Av) and the age of plant roots examined is given in (Figure 2a-f). Table 1 gives a summary of the ANOVA output for metals in sediments, plant roots and age of roots. Table 2 has correlation of metal concentrations in sediments and plant roots while Table 3 presents that of metal concentrations in plant roots and the age of the plant root.


Heavy metals in Sediments
Both temporal and spatial variations were quite visible in terms of the variables examined. For purposes of clarity, trends and comparison, graphs were plotted to highlight spatial variations on monthly distinctions in line with discussions. First set of samples collected in December, only Ni was considerably observed in root and sediments while Pb and Cd were < 0.02 and <0.001 μgg-1 respectively across study stations. The concentration of Ni in sediments (<0.018 - 5.0 μgg-1) and those in root (<0.018 - 6.5 μgg-1) showed differences in the accumulation of the same heavy metal by different mangrove plants root within the same ecosystem. This implies that level of Ni observed in sediment did not transmit to a corresponding proportion in the root of the respective plants in the same month.
The sediments around the root of Rhizophora had elevated
values of Ni compared to actual Ni concentration in the root.
Interestingly, Avicennia roots bioaccumulated more of the heavy
metal relative to the surrounding sediments. This is an implication
for phytoremediation of such metal in polluted environments. This
finding agrees with that of [40] who also recorded higher values
(μgg-1) of Ni-33.12, Cd-0.33 and Pb-5.01 in the root of Avicennia
compared to the respective values (27.42, 0.02 and 0.53) of the
metals in sediments. The age of the plant roots also varied (2.3 -
4.2 years) as Avicennia root of higher age concentrated elevated
amount of Ni unlike those of Rhizophora. The second set of samples
in January also had metal levels in sediment generally higher than
values in mangrove plant roots with St.1 and St.3 having higher
values compared to St.2. But Ni (7.0 μgg-1) and Cd (0.6 μgg-1) were
only observed in sediments at St.3 and St.1respectively while
Pb in sediment (2.3 - 7.3 μgg-1) and root (1.2 - 7.3 μgg-1) were
recorded in all stations examined. The levels of Pb obtained in this
study were generally higher compared to those (0.34 mg/kg) for
polluted soils and 0.001mg/kg for unpolluted mangrove soils in
the Niger Delta [41] but Cd values in the sediment of this study
were comparable to concentrations reported by [41]. Avicennia
root also concentrated more of Pb relative to the surrounding
sediment regardless of root age unlike Rhizophora suggesting
a relationship with intake from the surrounding environment.
However, Rhizophora with the highest age (4.3 years) at St.1 did
not accumulate the highest metal but Avicennia at St.3 had the
highest concentration of Pb (7.3 μg/g) at the mean age of 3.3 years.
Age difference between periods of sampling was quite minimal.
At the 3rd sampling in February, all three metals were recorded in
all stations in the order Pb>Ni>Cd with St.3 Metal concentration in sediment was Pb (<0.02 - 7.6 μgg-1),
Ni (<0.018 μgg-1), Cd (<0.001 μgg-1) while those in root were Pb
(<0.02 - 8.7 μgg-1), Ni (<0.018 - 2.2 μgg-1) and Cd (<0.001 - 1.0 μgg-1).
Plant roots accumulated more metals particularly Avicennia roots
when compared to the root of Rhizophora and the surrounding
sediments. Avicennia root with higher age generally accumulated
more metals relative to Rhizophora roots with no clear pattern in
relation to age of plant root. Increase in age with corresponding
rise in metal concentration in the root was shown more in
Avicennia plant compared to other mangrove plants examined.
This result agrees with the findings of other researchers that
Avicennia is a highly efficient plant for bioaccumulation of heavy
metal contaminant [17,33,40,42]. In the month of March only Pb
(1.2 μgg-1) was observed in sediment around the root of Avicennia
at St.1 but the roots of the plants indicated higher levels of Pb, Ni
and Cd compared to values in the surrounding sediment across
the study sites. The oldest plant root (Av - 6.3yrs) however, did
not accumulate the highest concentration of metal at specific
locations. The preference for root was clearly demonstrated by
Pb, Ni & Cd across study sites where such metals were detected in
the root but not in the surrounding sediments. This pattern was
also observed in April samples with values in sediment as follows
Pb: <0.02 - 6.5 μgg-1, Ni: <0.018 - 4.2 μgg-1, Cd: <0.001μgg-1) and
values in root (Pb: 1.3 - 6.2 μgg-1, Ni: 0.1 - 3.2 μgg-1, Cd: <0.001
- 0.4 μgg-1). The levels of Cd and Ni of this study corroborate the
findings of Gbosidom VL, Obute GC and Tanee FBG who reported
similar ranges within the Niger Delta mangrove (Rhizophora) but
at variance in terms of Pb content [43]. However, researchers
elswhere had reported Ni values (mg/kg) in sediments around
Avicennia as 25.08, 54.12 and 1.9 - 7.7 [42,44,45]. The observed
trend shows Avicennia roots indicating higher accumulation of
metals compared to Rhizophora roots with St.3 having elevated
levels of metals in plant roots and sediments. This is due to St.3
having more input of upland drainage and other anthropogenic
activities compared to other sites. The Pb content of Rhizophora at St.3 indicated a corresponding
increase with the Pb content (6.2 μg/g) of the surrounding
sediment but did not show a proportional increase with root age
(2.7yrs). This implies that mangrove plant roots with lower age
can also accumulate higher levels of heavy metal contaminants
than older plant roots with respect to mangrove type. The heavy
metals (Pb, Cd and Ni) content of both sediment and plant root
in this study were lower compared to the findings of [46] who
reported (sediment= Cd; 28.10, Pb:41.53, Ni:28.08 μgg-1 and root
- Cd; 343.08, Pb:502.04, Ni:609 μgg-1) in a crude oil polluted area
within the Niger Delta. The last set of samples in May examined
also affirmed higher accumulation of heavy metals in Avicennia
root than the surrounding sediments compared to values found in
Rhizophora root. Metal concentrations in sediments and roots was
in the order Pb>Ni>Cd with variations across sites studied, hence,
Pb and Ni generally remained higher in concentration compared
to Cd. In another study, Nazli MF et al. [47] reported higher
concentrations of Pb (83.1±3.1) and Cd (0.8±0.5) in sediments
above the values of this study but had Cd values (0.6) in mangrove
plant roots was comparable to that of this study. The value of Pb
(92.9) in the root of mangrove plant reported by is at variance
with those of this study [47]. The age of the plant root also
varied across sites but incremental concentrations of metals with increase in age of root was more apparent in Avicennia. However,
one deviation occurred at St.2 where the root of Avicennia with
the highest age (7.3yrs) did not accumulate the highest metal
level, interplay of other environmental factors may be responsible
for such difference. The concentration of Pb in sediments surrounding the
root of Rhizophora showed significant spatial and temporal
variations (p<0.05). Post hoc analysis showed actual
concentration to occur thus: location (St.3 The concentration of Pb in the root of Avicennia was also
significantly different (p<0.05) between stations examined
with post hoc analysis indicated as St.3 Pearson correlation in Table 2 was used to determine the
magnitude and direction of relationship between levels of heavy
metals in sediment and those in plant roots since the variables in
question are independent. Other significant correlations existed
due to interaction of different heavy metals but fell outside our
study interest. The level of Ni in sediments around Avicennia had
significant positive correlation (p<0.01) with the level of the metal
in the root of the plant. This implies an increase in Ni content of the
surrounding sediment translated into an increase in the Ni content
of the Avicennia plant root. The implication is that Avicennia plant
accumulated more metals with corresponding increase in the
environment, but this trend was different for the Rhizophora plant
root. The higher concentration of metals in Avicennia sp root in
this study also consents with the findings of who reported a value
range of 0.6 - 5.5μg/g. in similar environments [51]. S Rh - Sediment around Rhizophora root, S Av - Sediment
around Avicennia root R Rh - Root of Rhizophora, R Av - Root of Avicennia ns - not significant, ** - significant (p<0.01) The strength and direction of linear relationship between
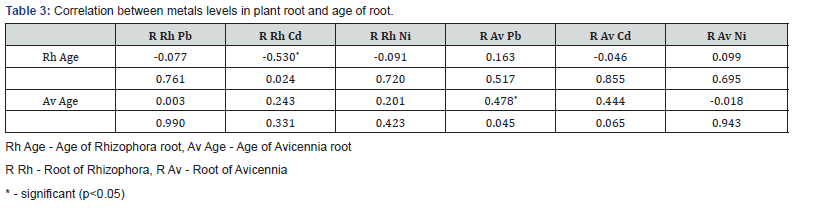
the level of metal in the root of the plant and the age of the plant
was also examined with Pearson correlation as given in (Table
3). The level of Cd in the root of Rhizophora had a significant
(p<0.05) linear negative relationship with the age of the plant
root while the concentration of Pb in the root of Avicennia had
a strong positive correlation. This implies that as the age of the
root of Rhizophora increased it absorbed less Cd and possibly
other metals while increase in the age of the root of Avicennia
correspondingly increased intake of Pb and likely other metals
depending on metal interactions. The effective intake of heavy
metals by the root of mangrove plants may be governed by the
nature of the plant, availability of the metal in surrounding soil,
metal interactions and other environmental factors. Rh Age - Age of Rhizophora root, Av Age - Age of Avicennia root R Rh - Root of Rhizophora, R Av - Root of Avicennia * - significant (p<0.05)
Sediment and mangrove plant root samples were collected
from three different stations in the southern region (Port
Harcourt) of Nigeria for the analysis of heavy metals (Pb, Ni and
Cd). The age of the plant roots were also examined with a view to
establishing relationship between heavy metal levels in sediment, plant root and age of root. Results indicated spatial and temporal
variations in the heavy metals accumulated in the roots and the
sediment/soil surrounding plant roots examined. Heavy metal
concentration both in sediment and plant root was generally in
the order Pb>Ni>Cd with higher concentrations observed at St.3
followed by St.1 and St.2. due to the anthropogenic activities at the
various stations. The study concluded that different plant roots
contain different concentrations of heavy metals irrespective of
the concentrations within their surrounding sediment. Avicennia
concentrated more metals with increase in the metal content of
the surrounding sediment but this relationship was not observed
for Rhizophora root and sediment metal content. Though, there
were variations in the age of the plants, there was no clear
pattern that the plant root with the highest age also had highest
accumulation of metal. Importantly, the root of Rhizophora
showed that increase in age was met with reduced metal (Cd)
concentration while Avicennia generally accumulated more heavy
metal (Pb) with increase in root age. Observed discrepancies in
heavy metal concentration with respect to study site was mainly
human factor induced. Correlation (Heavy metals in sediment and root of plants)
Correlation (heavy metals in plant roots and age of roots)
Conclusion
References
- Silva CAR, Lacerda LD, Rezende CE (1990) Heavy metal reservoirs in a red mangrove forest. Biotropica. 22: 339-345.
- Windom HL (1992) Contamination of the marine environment from land-based sources. Mar Pollut Bull 25(1): 32–36.
- Danovaro R (2003) Pollution threats in the Mediterranean Sea: an overview. Chem Ecol 19(1): 15–32.
- De Mora S, Fowler SW, Wyse E, Azemard S (2004) Distribution of heavy metals in marine bivalves, fish and coastal sediments in the Gulf and Gulf of Oman. Mar Pollut Bull 49 (5-6): 410–424.
- Defew LHJ, Mair M, Guzman HM (2005) An assessment of metal contamination in mangrove sediments and leaves from Punta Mala Bay, Pacific Panama. Mar Pollut Bull 50(5): 547–552.
- Crain CM, Halpern BS, Beck MW, Kappe CV (2009) Understanding and managing human threats to the coastal marine environment. Annal New York Acad Sci 11(62): 39–62.
- Moslen M, Miebaka CA, (2017a) Hydrocarbon Contamination of Sediments in the Niger Delta Region: a case study of the Azuabie creek, upper reaches of the Bonny Estuary, Nigeria. IOSRJ Environ. Sci Toxicol Food Technol. 11(9): 26-32.
- Moslen M, Aigberua A (2018a) Sediment contamination and ecological risk assessment in the upper reaches of the Bonny Estuary, Niger Delta, Nigeria. J Environ Toxicol Pub Health 3: 1-8.
- Barling RD, Moore ID (1994) Role of buffer strips in management of waterway pollution: a review. Environ Manage. 18(4): 543–558.
- MacFarlane GR, Pulkownik A, Burchett MD (2003) Accumulation and distribution of heavy metals in the grey mangrove, Avicennia marina (Forsk.)Vierh. Biological indication potential. Environ Pollut 123(1): 139–151.
- Reboreda R, Cac¸ador I (2007) Halophyte vegetation influences in salt marsh retention capacity for heavy metals. Environ Pollut. 146(1): 147–154.
- Moslen M, Ekweozor IKE, Nsirim-Dimkpa N (2017) Assessment of heavy metals pollution in surface sediments of a tidal creek in the Niger Delta, Nigeria. Archives of Agric. Environ. Sci. 3(1): 81-85.
- Moslen M, Aigberua A (2018b) Heavy Metals and Hydrocarbon Contamination of Surface water in Azuabie Creek within Bonny Estuary, Nigeria. J Appl Sci Environ Manage 22(7): 1083–1088.
- MacFarlane GR, Burchett MD (2000) Cellular distribution of Cu, Pb and Zn in the Grey Mangrove Avicennia marina (Forsk.) Vierh. Aqua Bot 68: 45-59.
- Burt JA (2014) The environmental costs of coastal urbanization in the Arabian Gulf. City 18(6): 760–770.
- Singh RN, Gautam AM, Gupta R (2011) Heavy metals and living systems: an overview. Indian J Pharm 43(3): 246–253.
- Dias HQ, Nayak GN (2016) Geochemistry and bioavailability of mudflats and mangrove sediments and their effect on bioaccumulation in selected organisms within a tropical (Zuari) estuary, Goa, India. Mar Pollut Bull 105(1): 227–236.
- Moslen M, Miebaka CA (2017b) Heavy Metal Contamination in Fish (Callinectis amnicola) from an Estuarine Creek in the Niger Delta, Nigeria and Health Risk Evaluation. Bull Environ Contam and Toxicol 99(4): 506-510.
- Moslen M, Miebaka CA (2017c) Concentration of Heavy Metals and Health Risk Assessment of Consumption of Fish (Sarotherodon melanotheron) from an Estuarine Creek in the Niger Delta, Nigeria. IOSR J. Environ. Sci Toxicol Food Tech. 11(3): 68-73.
- Panda SS, Chaturvedi N, Dhal NK, Rout NC (2013) An assessment of heavy metal accumulation in mangrove species of Bhitarkanika, Odisha, India. Res. Plant Bio. 3(6): 01-05.
- MacFarlane GR, Koller CE, Blomberg SP (2007) Accumulation and partitioning of heavy metals in mangroves: A synthesis of field-based studies. Chemosphere, 69(9): 1454-64.
- Nagelkerken I, Blaber SJM, Bouillon S, Green P, Haywood M, et al. (2008) The habitat function of mangroves for terrestrial and marine fauna: A review Aqua Bot 89: 155-85.
- Mackey AP, Hodgkinson M, Nardella R (1992) Nutrient levels and heavy metals in mangrove sediments from the Brisbane River, Australia. Mar Pollut Bull 24: 418-420.
- Lacerda LD, Carvalho CEV, Tanizak KF, Ovalle ARC, Rezende CE (1993) The biogeochemistry and trace metals distribution of mangrove rhizospheres. Biotropica, 25(3): 251-256.
- Kathiresan K, Bingham BL (2001) Biology of mangroves and mangrove ecosystems. Adv Mar Bio 40: 81-251.
- Silva CAR, Lacerda LD, Rezende CE (1990) Metals reservoir in a red mangrove forest, Biotropica 22(4): 339–345.
- Goyer RA, (2001) Toxic Effect of Metals. In: Casarett and Doull’s Toxicology, Klassen C D (Ed) 891-867.
- MacFarlane GR, Burchett MD (2002) Toxicity, growth and accumulation relationships of copper lead and zinc in the grey mangrove Avicennia marina (Forsk.) Vierh. Mar Environ Res 54(1): 65–84.
- Giri C, A Singh, T Loveland, J Masek, N Duke, et al. (2011) Status and distribution of mangrove forests of the world using earth observation satellite data. Global Ecol Biogeogr 20: 154–159.
- Nandy S, Mitra S (2004) Features of Indian Sunderbans mangrove swamps. Environ Ecol. 22: 339 – 344.
- Abere SA, Ekeke BA (2011) The Nigerian Mangrove and Wildlife Development. 1st International Technology, Education and Environment Conference, Omoku-Rivers State, Nigeria , September 2011.
- Chiu CY, Chou CH (1991) The distribution and influence of heavy metals in mangrove forests of the Tamushi estuary in Taiwan. Soil Sci plant nut 37: 659-669.
- Tam NF, Wong YS (1999) Mangrove soils in removing pollutants from municipal wastewater of different salinities. J Environ Qual 28: 556-564.
- Forstner U (1989) Contaminated Sediments: Lecture Notes in Earth Sciences. (1st Edn)., Springer, Berlin, pp. 157.
- Clark MW, Conchie DM, Lewis DW, Saenger P (1998) Redox stratification and heavy metal partitioning in Avicennia-dominated mangrove sediments: A geochemical model. Chem Geol 149: 147-171.
- Lewis DW, McConchie D (1994) Practical Sedimentology. 1st Edn, Chapman and Hall, New York, USA, pp. 213.
- American Society for Testing and Materials (ASTM) (1986) Annual book of ASTM standards, 11.01, (1986) D3559-85, Philadelphia PA, 19103.
- Verheyden A, Kairo JG, Beeckman H, Nico K (2004) Growth Rings, Growth Ring Formation and Age Determination in the Mangrove Rhizophora mucronata. Annals of Bot 94(1): 59-66.
- Menezes M, Berger U, Worbes M (2003) Annual growth rings and long-term growth patterns of mangrove trees from the Bragança peninsula, North Brazil. Wetlands Ecol Manage 11: 233–242.
- Abohassan RA (2013) Heavy Metal Pollution in Avicennia marina Mangrove Systems on the Red Sea Coast of Saudi Arabia. Met Env Arid Land Agric Sci 24(1): 35-53.
- Numbere AO (2017) Comparison of Microbial and Heavy Metal Contents in Soils and Roots under Mangrove Forest Stands with Different Levels of Pollution in the Niger River Delta, Nigeria. American J of Appl Sci 15(2): 132-140.
- Einollahipeer F, Khammar S, Sabaghzadeh A (2013) A study on heavy metal concentration in sediment and mangrove (Avicennia marina) tissues in Qeshm Island, Persian Gulf. J Novel Appl Sci 2(10): 498–504.
- Gbosidom VL, Obute GC, Tanee FBG (2017) Evaluation of Heavy Metal Levels and the Distribution of Rhizophora racemosa and Nypa fruticans in the Niger Delta Mangrove Forest, Nigeria. J Nat Sci Res 7(20) 9-16.
- Dehghani M, Karbassi A (2015) Determining environmental sensitivity of mangrove forest at hara protected area. J Biodivers Environ Sci 6(1) 480– 488.
- Seedo KA, Abido MS, Salih AA, Abahussain A (2017) Assessing Heavy Metals Accumulation in the Leaves and Sediments of Urban Mangroves (Avicennia marina (Forsk), Vierh.) in Bahrain. Hindawi Inter J Ecol 3978216: 8.
- Erakhrumen AA (2015) Assessment of In-Situ Natural Dendroremediation Capability of Rhizophora racemosa in a Heavy Metal Polluted Mangrove Forest, Rivers State, Nigeria. J Appl Sci Environ Manag 19(1): 21-27.
- Nazli MF, Hashim NH (2010) Heavy Metal Concentrations in an Important Mangrove Species, Sonneratia caseolaris, in Peninsular Malaysia. Environ Asia 3 4: 50-55.
- Alloway BJ (1990) Heavy metals in soils. John Wiley and Sons, Inc, New York, USA.
- EU (Euoropean Union) (2002) Heavy metals in Wastes, European Commission on Environment.
- WHO/FAO (2007) Joint FAO/WHO Food Standard Program, Codex Alimentarius Commission, 13th
- Shete A, Gunale VR, Pandit GG (2007) Bioaccumulation of Zn and Pb in Avicennia marina (Forsk.) Vierh. and Sonneratia apetala Buch. Ham. from Urban Areas of Mumbai (Bombay), India. J Appl Sci Environ Manage 11(3): 109-112.






























