Sorption of Heavy Metals with Natural Adsorbents: A Review
P Barragán-Peña12* and MG Macedo-Miranda2
1Tecnológico Nacional de México/Instituto Tecnológico de Nogales, Mexico
2Instituto Tecnológico de Toluca, México
Submission: December 20, 2017; Published: April 06, 2018
*Corresponding author: P Barragán-Peña, Tecnológico Nacional de M&3233;xico/Instituto Tecnológico de Nogales & Instituto Tecnológico de Toluca, Mexico, Tel: +52 6313111870; Fax: +52 6313111871; Email: perfectobarragan@itnogales.edu.mx
How to cite this article: P Barragán-P, MG Macedo-M. Sorption of Heavy Metals with Natural Adsorbents: A Review. Open Acc J of Toxicol. 2018; 3(1): 555602. DOI: 10.19080/OAJT.2018.03.555602
Abstract
Sorption technologies to remove heavy metals in aqueous systems were analyzed in detail in this review. Lead, chromium, cadmium, zinc and nickel have been investigated a great deal to propose better treatments that guarantee their elimination or reduction in polluted waters. The best sorption capacities obtained with modified natural adsorbents are discussed thoroughly, and the toxicology premises exposed. As for zeolites, the trend is to produce synthesized materials whose pore diameter is uniform. However, to modify natural clinoptilolite remains less costly and therefore still an alternative in water treatment, worldwide. Bentonite certainly complies with ionic exchange capacity to take up heavy metals as well.
Keywords: Sorption; Heavy metal; Zeolites; Clinoptilolite; Bentonite; Oligo-Element
Introduction
Heavy metals in underground water are a major concern to health and environmental research since they can become part of the food chain easily. It has been proven that some plants can absorb heavy metals like cadmium present in runoff water. Cadmium big concern, as targeted carcinogenic in a variety of studies, makes it one of the most investigated heavy metals in the last decade [1-3]. Since cadmium is usually present in lead and zinc ores, some studies include these three metals to compare behaviors and differentiate selectivity [4].
Tannery, plating, and poultry are three major industries that account for chromium and therefore contribute a great deal with the load of this metal to wastewater. Although chromium is necessary in human diet, it is still targeted as toxic.
A variety of technologies can remove heavy metals from aqueous systems: electro-chemical, membrane-filtration, chemical precipitation, ion-exchange, and sorption. Sorption is a technology of low cost; it includes adsorption and chemisorptions, when a chemical bond occurs. The use of natural materials to remove heavy metals by a sorption process has been investigated intensively. Furthermore, the modification of these natural adsorbents in order to improve their sorption capacity has fulfilled specific needs. As water scarcity urges to recycle and reuse the vital liquid, there is still a lot to do regarding removal of heavy metals if health and environmental safety are a priority.
Pollutant transport
Heavy metals represent a serious problem to living organisms due to their non-biodegradable condition. Electroplating, mining, polymer stabilizing, and pigment production, among other anthropogenic activities can deposit them in soil and water. They are capable of staying in the environment, accumulating for indefinite time. Lead, mercury and cadmium are considered a high priority risk to the environment, while chromium is labeled as of medium environmental risk [5].
The potential risk of heavy metals to living organisms is attributable to their capacity to bind to organic molecules, accumulating eventually. A study with rice crops, for instance, revealed that cadmium can be absorbed from soil, and after a series of transport mechanisms it accumulates into grains [6]. Lead, on the other hand, once absorbed in the body, is distributed to several organs, even bones, where takes over calcium [4].
Toxicology
The adverse effects of heavy metals on humans vary according to their chemical properties. As for cadmium, it deposits in the liver and it is bioacumulable [7]. Cadmium also binds to the essential respiratory enzymes which causes oxidative stress and cancer [8]. A blood or urine test can determine cadmium exposure of patients.
Lead oxide is a pollutant present in air in developing countries where unleaded gasoline is not obligatory to vehicle users. Lead causes neurotoxic effects on brain [9].
Zinc and chromium are, in fact, oligo-elements for human- beings, a regular intake is necessary in trace amounts. However, Zinc oxide causes metal fume fever, a disease related to galvanization, and symptoms include sore muscles, fatigue, nausea, and cough [10]. Some studies conducted with people, mostly electroplaters, revealed these workers developed asthma and other signs of respiratory distress when exposed to chromium (VI). Irritation and ulcerated nasal septum were reported frequently after 89 days of exposure at 0.025mg/m3 of chromium [11].
Sorption of heavy metals with natural modified adsorbents
A number of natural adsorbents have been used in the last decade to treat water polluted with heavy metals. Bentonite, zeolites, plant debris, etc [12-14]. Some of them were modified to improve their sorption capacity. Such modifications depended on the pollutant of interest; if ammonium or dyes were on target, the modification involved nonpolar organic molecules that could interact with them and therefore remove the substance [1,15].
The use of zeolites in sorption operations is extended worldwide. These minerals are highly selective and cation exchangers. The clinoptilolite type hosts sodium and calcium cations in its micropores, besides water. A disadvantage might be its average pore diameter, which is very small (about 18.6 nm)3, and its surface area, compared to activated carbon. Nevertheless, zeolites have been proved to take up heavy metals from aqueous solutions successfully [16-18].
Table 1 shows the different sorption capacities of natural adsorbents in heavy metal removal. Each material improved its sorption capacity after modification. To test interference in chemical family metals, boron enrichment process waste was used to remove a solution with Zn and Cd [19].
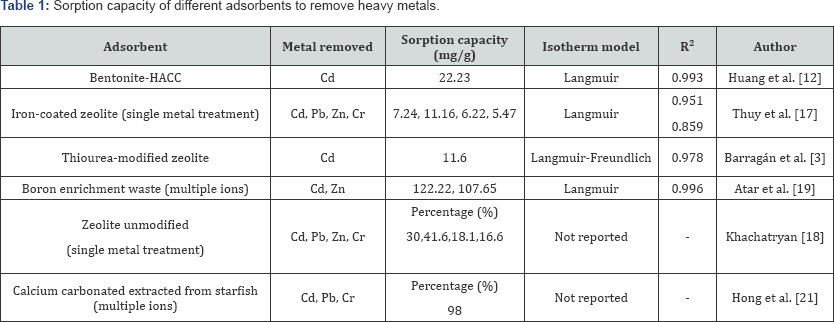
The surface area of adsorbents is crucial to the sorption process: Bentonite modified with N-2 Hydroxypropyltrimethyl Ammonium Chloride Chitosan (HACC) has 37.6 m2/g [12] While thiourea-modified zeolite, which showed a sorption capacity of 11.6mg/g, has 5.5 m2/g3 Zn and Cr are the metals with least affinity for the minerals as it can be seen from Table 1. Initial concentration of metals and pH influenced the uptake rate in the sorption process. Zeolite modified with thiourea (from Mexico) removed cadmium more efficiently than iron-coated zeolite from Australia. This can be explained in terms of the mechanism involved when an organic molecule is fixed to the surface of the adsorbent: the thiourea can interact in the sulfide part with the cation (Cd2+), they make a complex, a soft base binds to a soft acid (Pearson theory) [20].
The advantage bentonite-HACC has over the other adsorbents (Table 1) has to do with the recovery of the metal adsorbed. Huang et al. [12] reported that treatment of the bentonite with 0.01 ethylene diamine tetra-acetic acid resulted in the regeneration of the adsorbent and subsequent reuse [12].
Experiments carried out with the thiourea-modified natural zeolite showed that at 120mg/L of Cd2+ initial concentration for 48 hours desorption did not occur [3].
The potential to remove heavy metals from aqueous solutions these natural adsorbents have is significant in terms of the relatively low cost of operation, sorption capacity and environment safety. This alternative technology can definitively sum up to the efforts for a cleaner and healthier environment.
Tendency in sorption operations
Biosorption, the process to take up pollutants with plant debris, biomass or organic waste, has become very popular and extended lately [8, 21]. The trend is to recycle organic matter, plant and animal remains such as stems, roots, peels, bones, etc., to remove heavy metals and other pollutants form aqueous solutions.
Asia and Europe reported different synthetic material to clean up contaminated water that show bigger and regular pores [22]. Some of these synthetic materials have been proved efficient in removing radioactive pollutants after loaded with Ag or Cu, and for targeted drug delivery [23,24].
Conclusion
Heavy metals such as cadmium, lead, zinc and chromium are targeted as hazardous to human health, therefore a lot of research has been developed to propose alternative technology to remove them from water. Sorption is a relatively easy and cheap operation to take up heavy metals from aqueous solutions.
Some natural adsorbents have been proved successfully to remove cadmium, lead, zinc and chromium, their sorption capacity has improved after modification with organic molecules such as thiourea, chitosan, and surfactants.
A well-identified toxicity is related to cadmium, lead, zinc and chromium exposure. Some of the toxicology involves respiratory stress, but it can be as bad as cancer. However, zinc and chromium are considered oligo-elements, necessary in the human diet.
Biosorption and synthetic adsorbents are gaining more application over sorption operations with natural modified adsorbents due to their regular pore distribution and high specialized performance in Environmental Sciences and Medicine.
Conflict of Interest
The authors declare there is not any conflict of interest that impedes to submit this article.
References
- Cortéz-Martínez R, Marcos Solache-Ríos, Verónica Martínez-Miranda, Ruth Alfaro-Cuevas (2009) Removal of Cadmium by Natural and Surfactant-modified Mexican Zeolitic Rocks in Fixed Bed Columns. Water Air Soil Pollut 196: 199-210.
- Marmiroli M, Pagano L, Savo Sardaro ML, Villani M, Marmiroli N (2014) Genome-Wide Approach in Arabidopsis thaliana to Assess the Toxicity of Cadmium Sulfide Quantum Dots. Environ Sci Tech 48(10): 59025909.
- Barragán P, Macedo M MG, Olguín MT (2017) Cadmium sorption by sodium and thiourea-modified zeolite-rich tuffs. J Environ Sci (China) 52: 39-48.
- Anju M, Banerjee DK (2010) Associations of cadmium, zinc, and lead in soils from a lead and zinc mining area as studied by a single and sequential extractions. Environ Monit Assess 176(1-4): 67-85.
- Volesky B (2001) Detoxification of metal-bearing effluents: biosorption for the next century. Hydrometallurgy 59: 203-216.
- Mathews CK, VH (1998) Bioquimica (2nd edn). Madrid: McGraw-Hill.
- Seung SH, Kim M, Lee SM, Lee JP, Kim S, et al. (2013) Cadmium expossure induces hematuria in Korean adults. Environmental Research 124: 23-27.
- Manasi RV, Rajesh Vidya, Santhana Krishna Kumar A, Rajesh N (2014) Biosorption of Cadmium using a novel bacterium isolated from an electronic industry effluent. Chemical Engineering Journal 235: 176185.
- Gochfeld M, LR (2011) Chemical Hazards. In W. D. Levy B.S, Occupational and Environmental Health. Oxford, UK.
- Plum LM, Rink L, Haase H (2010) The essential toxin: Impact of Zinc on Human Health. Int J Environ Res Public Health 7(4): 1342-1365.
- Wilbur S, AH (2012) Health Effects -Toxicological Profile for Chromium- NCB. Agency for Toxic Substances and Disease Registry, USA.
- Huang R, WB, Bingchao Yang, Zengqiang Zhang (2011) Equilibrium, kinetic and thermodynamic studies of adsorption of Cd (II) from aqueous solution onto HACC-bentonite. Desalination 280(1): 297-304.
- Salgado Gomez N, Macedo Miranda MG, Olguin MT (2014) Chromium (VI) adsorption from sodium chromate and potassium dichromate aqueous systems by hexadecyl trimethyl ammonium-modified zeolite rich tuff. Applied Clay Sci 95: 197-204.
- LC Koffi Akissi1, K Adouby, B Yao, D Boa (2013) Batch and Column Studies for Cadmium (II) removal using sawdust from Triplochiton scleroxylon. Intl J Engineering Sci and Tech 5(1): 31-40.
- PV Haseenaa, KS Padmavathya, P RohitKrishnana, G Madhu (2016) Adsorption of Ammonium Nitrogen from Aqueous Systems using chitosan bentonite film composite. Procedia Technology 24: 733-740.
- Ozge Can, Devrim Balkose, Semra Ulku (2010) Batch and column studies on heavy metal removal using a local zeolitic tuff. Desalination 259(1-3): 17-21.
- Thuy CN, PL (2015). Simultaneous adsorption of Cd, Cr, Cu, Pb, and Zn by an iron-coated Australian zeolite in batch and fixed-bed column studies. Chemical Engineering Journal 270: 393-404.
- Khachatryan SV (2014) Heavy metal adsorption by Armenian natural zeolite from natural aqueous solutions. Proceedings of the Yerevan State University (2): 31-35.
- Atar Necip, Olgun A, Wang Shaobin (2012) Adsorption of Cadmium (II) and Zinc (II) on boron enrichment process waste in aqueous solutions: Batched and fixed-bed system studies. Chemical Engineering Journal, Curtin University 192: 1-7.
- Valenzuela Calahorro C (1995) General Chemistry, Introduction to the theoretical Chemistry. Salamanca: Agapea Salamanca University.
- Kyong-Soo Hong, Hak Myoung Lee, Jong Seong Bae, Myoung Gyu Ha, Jong Sung Jin, et al. (2011) Removal of Heavy Metal Ions by using Calcium Carbonate extracted from starfish treated by Protease and Amylase. Journal of Analytical Sci Tech 2(2): 75-82.
- Van Aelst J, PA (2015) Efficient design of hierarchical structure in highly dealuminated USY zeolites without extraction of framework atoms. 2nd Euro-Asia Zeolite Conference (p. 57) Nice, Caen, France.
- Chebbi M, AB (2015) Exchanged-zeolites for the trapping of iodine species in the context of nuclear accident. 2nd Euro-Asia Zeolite Conference (pp. 122) Nice, Caen, France.
- Schmidt A, WV (2015) Multifunctional Mesoporous Nanoparticles for targeted drug delivery. 2nd Euro-Asia Zeolite Conference (p. 4) Nice, Caen, France.






























