- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
The Importance of Dose-Time-Response Relationships for Hazard Identification and Limitation of Animal Experiments
Henk A Tennekes*
Experimental Toxicology Services (ETS) Nederland BV, Netherlands
Submission: August 08, 2017; Published: August 22, 2017
Corresponding author: Henk A Tennekes, Experimental Toxicology Services (ETS) Nederland BV, Frankensteeg 4, 7201KN Zutphen, Netherlands, Tel: +31(0)575 547717; Email: info@toxicology.nl
How to cite this article:Henk A Tennekes. The Importance of Dose-Time-Response Relationships for Hazard Identification and Limitation of Animal Experiments. Open Acc J of Toxicol. 2017;1(5): 555572. DOI: 10.19080/OAJT.2017.01.555572
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
Introduction
Historian Heiko Stoff has recently sketched a fascinating controversy in the 1950's on chemical risk assessment [1]. Two renowned scientists in the Farbstoffkommission (Dye Committee) of the Deutsche Forschungsgemeinschaft (DFG, German Research Community), pharmacologist Hermann Druckrey and biochemist (and Nobel Prize winner) Adolf Butenandt, were advocates of a preventive risk approach. This approach was largely determined by a groundbreaking study conducted by Druckrey during the war years with the carcinogenic dye 4-dimethylaminoazobenzene (also known as "butter yellow") [2], and Druckrey's un-intentional cooperation with the electrophysicist Karl Küpfmüller in an American detention camp in Hammelburg, Bavaria [3].
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
The History of the Druckrey-Küpfmüller Equation
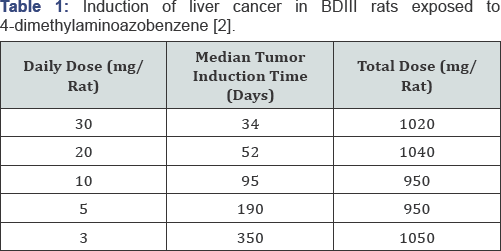
Druckrey demonstrated in 1943 that the carcinogenic action of "butter yellow" was determined by the total dose, and completely independent of the daily dose (Table 1). The product of daily dose D and exposure time (up to liver cancer manifestation) T was always the same:
- D. T = constant (1)
and found to be about 1000mg (1 gram) in the case of butter yellow (Table 1).
This dose-effect relationship is known as Haber's rule (or law) [4], named after the German chemist Fritz Haber, who played a key role in chemical warfare in the Great War. Haber's rule originally described the relationship between gas concentration c and time to death t. The smaller the c.t product, the higher the toxicity. Druckrey's observation that Haber's rule also described the dose-response relationship of a carcinogenic substance was remarkable in view of long latency periods. Druckrey concluded from this study that the harmful effects of a carcinogen were cumulative and that thresholds of toxicity for carcinogens do not exist.
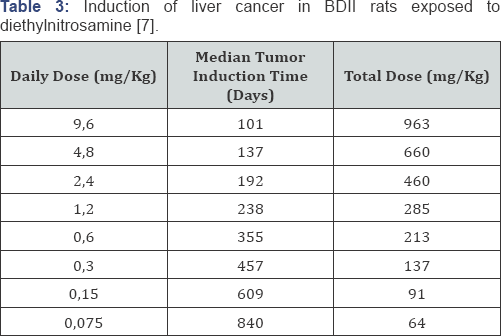
Druckrey and Kupfmuller also explained this dose-response relationship theoretically with a mathematical analysis of receptor kinetics, as shown in Table 2 [5]. It was assumed that the carcinogenic effect of butter yellow was due to irreversible interactions with a specific receptor. We now know that the receptor is DNA, and that cancer is the result of cumulative damage to DNA, but that was not known in those years. However, Druckrey and Kupfmuller also postulated that when the effect of receptor binding is irreversible as well, effects would be amplified over time (Table 2). In 1956, when Peter Magee and John Barnes linked the carcinogenicity of dimethylnitrosamine to alkylation of nucleic acids [6], Druckrey took the initiative to investigate the dose-time relationship of diethylnitrosamine (DENA) in rats to verify possible reinforcement of effects by exposure time. After all, irreversible DNA alkylation results in irreversible mutations, and nitrosamines therefore appeared to be perfect model substances. This study confirmed potentiation of carcinogenicity by time [7] which could be expressed as follows:
D. T n = constant (2)
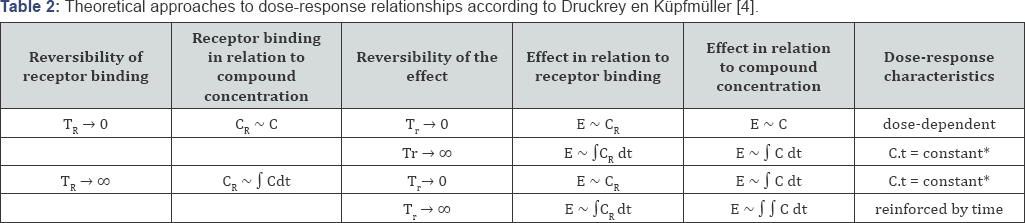
TR is the time constant for the reversibility of receptor binding.
Tr is the time constant for the reversibility of the effect.
C is the concentration of the compound at the site of interaction with the receptor.
CR is the concentration of bound receptors.
E is the effect.
* if C remains constant.
with a value ofthe time exponent n of 2.3. This dose-response relationship is now known as the Druckrey-Kupfmuller equation. The equation explains the harmful effects of low exposure levels of a poison during prolonged exposure (Table 3). The lower the daily dose, the lower the total dose required for the damaging effect, even though the adverse effect occurs only after a long exposure period.
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
Risk Prevention is Displaced by Risk Management
It is not surprising therefore that Druckrey and his influential friend Butenandt became major advocates of risk prevention. Only substances with a reversible mechanism of action and dose-dependent toxicology (Table 2) were acceptable in their eyes because safe exposure concentrations below a threshold of toxicity could be defined. By contrast, substances with an irreversible mechanism of action which followed equations (1) and (2), had no threshold and exposure should, where possible, be avoided.
However, in the 1960's, this approach was displaced by the ADI (acceptable daily intake) concept, which defines an acceptable level of exposure for a substance, independent of the mechanism of action. The ADI concept was mainly propagated by French professor René Truhaut [8] and received a lot of support from the chemical industry, because it was seen as a manageable concept for product development. This had major consequences for toxicological research. The primary objective of animal experiments was no longer clarification of the dose-response relationship and the mechanism of action, but determination of the dose that did not cause any harmful effect, in comparison to control animals, the so-called No-Observed-Adverse-Effect Level (NOAEL). The NOAEL is then divided by a safety factor, usually 100, to take account of possible differences in sensitivity between experimental animals and humans, and individual variation in sensitivity between humans. That exposure level (NOAEL: 100) is then considered as the ADI, the permissible daily human exposure. An exception was made for substances with mutagenic (DNA-damaging) properties, which were not allowed unless used to treat life-threatening diseases.
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
The Druckrey-Küpfmüller Equation is Generally Applicable
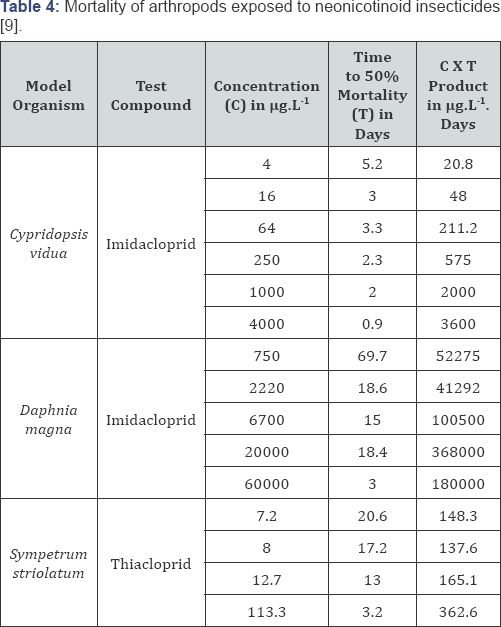
In 2009, Francisco Sanchez-Bayo showed that the toxicity of the neonicotinoid insecticides imidacloprid and thiacloprid for arthropods was not only dependent on the dose but also on the duration of exposure [9]. Henk Tennekes, who was mentored by Hermann Druckrey in his time at the German Cancer Center in Heidelberg, recognized the dose-response relationships as Druckrey-Küpfmüller equations [10]. The lower the exposure concentration, the lower the total dose required for the damaging effect (Table 4). The subsequent collaboration between Tennekes and Sanchez-Bayo provided additional examples of substances with time-cumulative toxicity (Cartap, Diphacinone, organic mercury) [11,12]. All of these substances cause irreversible receptor binding and irreversible effects, and there are no indications for a threshold [13], so that the ADI for these substances underestimate the actual risks. In any case, it became clear that the theories of Druckrey and Küpfmüller are generally applicable and that equations (1) and (2) are of importance to the risk analysis of chemicals.
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
The Risks of an Irreversible Mechanism of Action are Underestimated
The widely used neonicotinoid insecticide imidacloprid binds virtually irreversibly to nicotinic acetylcholine receptors in the central nervous system of insects and causes irreparable damage to nerve cells [14,15]. The substance is very slowly decomposed in the soil (half-life 200 days), and may leach into the groundwater, or run-off to surface water [16]. In water, the substance can only be degraded by ultraviolet light (photolysis). In many areas of intensive agriculture, surface water is contaminated with imidacloprid [16]. As a result, nontarget insects are exposed to an extremely toxic substance for a long time, which can lead to massive insect mortality and a break in the food chain [16]. Studies of the Universities of Utrecht and Nijmegen showed that the pollution of surface water with imidacloprid quantitatively correlated with decline of invertebrates and insects-dependent bird species [17,18]. Research in the National Park Dwingelderveld, The Netherlands, and in a nature reserve in Krefeld, Germany, showed that, since the introduction of imidacloprid in the mid-1990's, at least three-quarters of the ground beetles and flying insects have disappeared [16,19]. The risks of imidacloprid have been completely underestimated, with catastrophic consequences for insects and insectivores.
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
Review of Risk Analysis is Urgently Required
Hermann Druckrey and Adolf Butenandt seem to have defined the correct approach to risk analysis of chemicals in the 1950's, as the new insights of recent years show. The ADI concept of René Truhaut is in any case unacceptable for substances with action mechanisms described by Haber’s rule or the Druckrey- Küpfmüller equation. Dose-response relationships are of much greater importance than a NOAEL in an experimental experiment because irreversible effects can be identified. In addition, dose- response relationships can make accurate estimates of the risks in the real world. This may make risk management more restrictive, but at least a lot safer. And there is another important perspective. Dose-response relationships can make a significant contribution to a strong reduction in the use of laboratory animals (Table 4).
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
The development of a new pesticide now costs almost $300 million [20], and expenditure for international market authorization procedures have risen exponentially in recent years. This development reflects the increasing concern about harmful effects of pesticides on farmland biodiversity. Imidacloprid is a case in point. The general applicability of the theories of Druckrey and Küpfmüller means that analysis of dose- response relationships can identify and eliminate hazardous substances at an early stage of product development [21]. Dose- response studies can be performed in a short period of time, for example with Daphnia magna (water fleas). In doing so, product development is shifted to substances with dose-dependent toxicology (Table 2). Long-term experimental experiments to determine a NOAEL are no longer necessary, as exposure time has no effect. In combination with a multitude of available in vitro studies (http://www.oecd.org/env/ehs/testing/adopted- testguidelines-toxicity-testing-3r-relevance.htm), this strategy can make a major contribution to the implementation of the 3R Principles (Replacement, Reduction and Refinement) that Russel & Burch first described in 1959 to limit the use of experimental animals [22].
- Editorial
- Introduction
- The History of the Druckrey-Küpfmüller Equation
- Risk Prevention is Displaced by Risk Management
- The Druckrey-Küpfmüller Equation is Generally Applicable
- The Risks of an Irreversible Mechanism of Action are Underestimated
- Review of Risk Analysis is Urgently Required
- Analysis of Dose: Response Relationships Can Make Many Animal Studies Superfluou
- Reference
Reference
- Stoff VH (2014) Zur Kritik der Chemisierung und Technisierung der Umwelt. Risiko-und Präventionspolitik von Lebensmittelzusatzstoffen in den 1950er Jahren. Technikgeschichte Bd 81(H3): 253-273.
- Druckrey H (1943) Quantitative Grundlagen der Krebserzeugung. Klinische Wochenschriften 22(34-35): 532-534.
- Wunderlich V (2005) On the Origins of the Druckrey-Küpfmüller Papers (1948-1949): Dose-Effect Relations in Carcinogenic Substances. Medizinhist J 40(3-4): 369-397.
- Haber F (1924) Zur Geschichte des Gaskrieges. Funf Vortrage aus den Jahren 1920-1923, pp. 76-92.
- Druckrey H, Küpfmüller K (1949) Dosis und Wirkung. Beitrage zur theoretischen pharmakologie, Editio Cantor GmbH, Freiburg imBreisgau, Germany
- Magee PN, Barnes JM (1956) The production of malignant primary hepatic tumours in the rat by feeding dimethylnitrosamine. Br J Cancer 10: 114-22.
- Druckrey H, Schildbach A, Schmaehl D, Preussmann R, Ivankovic S (1963) Quantitative Analysis of the Carcinogenic Effect of Diethylnitrosamine. Arzneimittelforschung 13: 841-851.
- Truhaut R (1991) The concept of the acceptable daily intake: an historical review. Food Addit Contam 8(2): 151-162.
- Sanchez-Bayo F (2009) From simple toxicological models to prediction of toxic effects in time. Ecotoxicology 18(3): 343-354.
- Tennekes HA (2010) The significance of the Druckrey-Kupfmuller equation for risk assessment-the toxicity of neonicotinoid insecticides to arthropods is reinforced by exposure time. Toxicology 276(1): 1-4.
- Tennekes HA, Sanchez-Bayo F (2013) The molecular basis of simple relationships between exposure concentration and toxic effects with time. Toxicology 309: 39-51.
- Pletz J, Sanchez-Bayo F, Tennekes HA (2016) Dose-response analysis indicating time-dependent neurotoxicity caused by organic and inorganic mercury- Implications for toxic effects in the developing brain. Toxicology 347-349: 1-5.
- Tennekes HA (2016) A Critical Appraisal of the Threshold of Toxicity Model for Non-Carcinogens. J Environ Anal Toxicol 6: 5.
- Abbink J (1991) The Biochemistry of Imidacloprid. PflanzenschutzNachrichten Bayer, Serial ID- ISSN: 0340-1723, Germany
- Mehlhorn H, Mencke N, Hansen O (1999) Effects of imidacloprid on adult and larval stages of the flea Cfenoaephalides felis after in vivo and in vitro application: a light-and electron microscopy study. Parasitol Res. 85(8-9): 625-637.
- Tennekes HA (2010) The Systemic Insecticides: A Disaster in the Making. Zutphen, ETS Nederland BV, Netherlands, p. 72.
- Van Dijk TC, Staalduinen VMA, Van der Sluijs JP (2013) MacroInvertebrate Decline in Surface Water Polluted with Imidacloprid. PLoS ONE 8(5): e62374.
- Hallmann CA, Foppen RP, van Turnhout CA, de Kroon H, Jongejans E (2014) Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511(7509): 341-343.
- Sorg M, Schwan H, Stenmans W, Muller A (2013) Ermittlung der Biomassen flugaktiver Insekten im Naturschutzgebiet Orbroicher Bruch mit Malaise Fallen in den Jahren 1989 und 2013. Mitteilungen aus dem Entomologischen Verein Krefeld 1: 1-5.
- Anonymous (2016) The Cost of New Agrochemical Product Discovery Development and Registration in 1995, 2000, 2005-8 and 2010 to 2014. R&D expenditure in 2014 and expectations for 2019. Phillips McDougall Consultancy Study for CropLife International, CropLife America and the European Crop Protection Association, USA.
- Tennekes HA (2017) Dose: Time-to-effect analyses can identify hazardous chemicals at an early stage of product development. Environ Risk Assess Remediat 1(2): 16-21.
- Russell WMS, Burch RL (1959) The principles of humane experimental technique. Universities Federation for Animal Welfare, Hertfordshire, United Kingdom.






























