Crotalus lepidus lepidus Venom Produces Hemolysis and Lipid Peroxidation in Human Erythrocytes In Vitro
Montañez Rodriguez Esau, Aguirre Joya Jorge Alejandro, Sierra-Rivera Crystel Aleyvick, Morlett Chavez Jesus, Muzquiz Ramos Elia Martha and Zugasti-Cruz Alejandro*
Department of Chemistry, Autonomous University of Coahuila, Mexico
Submission: January 30, 2016 ; Published: February 11, 2016
*Corresponding author: Alejandro Zugasti-Cruz, Laboratory of Immunology, Faculty of Chemistry, Autonomous University of Coahuila. Jose Cardenas Valdes Street, Saltillo, Coahuila, Mexico, Tel: +52 01 (844) 4390511; E-mail: alejandro_zugasti@uadec.edu.mx
How to cite this article: Montanez R E, Aguirre J J A, Sierra-Rivera C A, Morlett C J, Muzquiz R E M, et al. Crotalus lepidus lepidus Venom Produces Hemolysis and Lipid peroxidation in Human Erythrocytes in vitro. Open Access J of Tox. 2015; 1(1): 555554. DOI: 10.19080/OAJT.2016.01.555554
Abstract
The hemolytic and lipid peroxidative effects of crude venom from Crotalus lepidus lepidus, a rattle snake found in the Northeast of Mexico and southeast of the United States, were investigated in human erythrocytes in vitro. Significant concentration-dependent effects were found on both hemolysis (evaluated as release of hemoglobin) and lipid peroxidation (as a common index of oxidative damage to membrane lipids) in the red blood cells. These results suggest that hemolysis was produced with the involvement of oxidative stress as a potential mechanism of toxicity in the erythrocytes.
Keywords: Hemolysis; Lipid Peroxidation; Crotalus lepidus lepidus Venom
Abbreviations: TBA: Thiobarbituric Acid; TCA: Trichloroacetic Acid; MDA: Malondyaldehide; (C.l. lepidus): Crotalus Lepidus Lepidus
Introduction
Snake venoms are among the best pharmacologically characterized natural toxins, chiefly because of their deleterious effects on humans [1]. Snake venoms are toxic secretions, which are complex mixtures of molecules of different biochemical nature, with a predominance of proteins, many of which are endowed with enzymatic activities [2]. The most common snake venom enzymes include acetylcholinesterases, L-amino acid oxidases, serine proteinases, metalloproteinases and phospholipases A2 [2]. Some phospholipases A2 cause a wide variety of pharmacological effects such as pre-synaptic or post- synaptic neurotoxicity, miotoxicity, anticoagulant and hemolytic activities [3,4]. PLA2 catalyzes the specific hydrolysis of ester bonds at the C2 position of 1,2-diacyl-3-sn-glycerophospholipids with release of free fatty acids. Thus, phosholipases A2 are able to disrupt the phospholipid packings from several types of biological membranes, leading to cell lysis [5]. There is some evidence that animal venoms can induce oxidative stress. For example, El Asmar et al. and Dousset et al. [6,7] have reported that the increase in polyunsaturated fatty acids following envenomation by scorpion venom may lead to an increase in the rate of lipid peroxidation, which might be responsible for tissue damage. In the case of snakes, the venom of Echis piramidum causes lipid peroxidation in different organs of mice [8] and the venom of Bothrops induces renal tubular toxicity mediated in part by lipid peroxidation [9]. Currently, lipid peroxidation is one of the most important organic expressions of oxidative stress induced by the free radicals produced during various types of xenobiotic exposures or pathological conditions, animal venoms included. The potential consequences of the peroxidative process of membrane lipids include loss of polyunsaturated fatty acids, decreased lipid fluidity, altered membrane permeability, effects on membrane-associated enzymes, altered ion transport, release of material from subcellular compartments, and the generation of cytotoxic metabolites or lipid hydroperoxides [10]. Furthermore, vertebrate red blood cell membranes have a high content of unsaturated lipids as well as iron in hemoglobin, one of the most powerful catalysts capable of initiating lipid peroxidation [11]. In addition, erythrocytes are anucleated cells and therefore lack protein synthetic machinery, they cannot replace many cellular components, so oxidative damage may induce a permanent alteration in the red cells [11]. In this study, we evaluated hemolytic response and lipid peroxidation of the crude venom from the rattlesnake Crotalus lepidus lepidus on isolated human erythrocytes.
Materials and Methods
Chemicals
Sodium Citrate, Anhydrous Dextrose, Sodium Chloride, Citric Acid, Thiobarbituric Acid (TBA), Trichloroacetic Acid (TCA) were all purchased from J.T. Baker (Center Valley, PA). All other reagents were from Sigma (St. Louis, MO).
Preparation of venom extract
The venom of Crotalus lepidus lepidus was purchased from the laboratory of National Natural Toxins Research Center at Texas A&M University-Kingsville. Venom solution was prepared using 10 mg of lyophilized venom in 5.0 mL of Alsever's solution pH 6.4 (dextrose 0.116 M, NaCl 0.071 M, sodium citrate 0.027 M, and citric acid 0.002 M), then, centrifuged at 12,000 g for 10 min at 4°C. The pellet was discarded and the supernatant was aliquoted and stored at -20°C until use. Protein concentrations were determined in samples by the method of Bradford (1976) [12] with bovine serum albumin as a standard.
In vitro hemolysis assay
The present study was approved by the Ethics Committee on Animal Experimentation of the Faculty of Chemistry of the Autonomous University of Coahuila, Mexico. The hemolysis test was performed using human whole blood from healthy non-smoking donors with permission, following guidelines for studies using human samples. In briefly, blood was collected in heparinized-tubes, was centrifuged at 3000 rpm for 4 minutes at 4°C. The pellet was washed three times with cold Alsever's solution. The supernatant was then removed and 100 mL of the purified erythrocytes were diluted 1: 99 with Alsever's solution. Then, 150 mL of this suspension was suspended in Alsever's buffer and taken for the curve-response experiments (total volume 1500 ml). This suspension of red blood cells was always freshly prepared and used within 24h after collection. Crude venom concentrations were tested: 0.250, 0.500, 1.0 and 2.0 mg/mL. The tubes were gently mixed in a rotator shaker, and then incubated at 36.5°C ± 1°C within a shaking water bath, for 60 minutes. Alsever’s solution and deionized water were used as negative and positive controls, respectively. Each group contained three tubes. The samples were then centrifuged under 3000 rpm for 4 minutes to collect the supernatant. The absorbance (A) value of the hemoglobin released from the erythrocyte cells was measured spectrophotometrically at 415 nm (Thermo Spectronic Genesys 5). All trials were run three times. The absorbance obtained was correlated with a curve of human hemoglobin.
The experiments were run in triplicate and were repeated twice.
Determination of malondialdehyde (MDA)
Erythrocytes MDA concentration was determined by using the method described by Draper and Hadley (1990) based on TBA reactivity (TBARs) with some modifications. In this manner, 1.0 mL of supernatant of the erythrocytes suspension (it was prepared in the similar way as the hemolytic assay) was mixed with 45 mL of TCA (50%). The samples were centrifugated at 3000 rpm/10 min. Then, 115 mL of TBA (0.75 % in 0.1 M HCl) was added and put into a boiling water bath for 10 min. Tubes were centrifuged at 3,000 rpm for 10 min, and the absorbance was measured at 535 nm. Results were expressed as nmol TBARs/ mg protein. Concentration of MDA was calculated by a calibration curve using 1,1,3,3-tetraethoxypropane as a standard.
Phospholipase A2 activity assay
The method of Araujo and Radvanyi [13] was used with some modifications. Venom solution (10 mg in 10 ml of PBS pH 7.4) was added to the reaction medium (1.5 mL) containing 15 mol phosphatydylcholine, 18 mol Triton X-100, 5mol calcium chloride, 80 mol phenol red and 7.5 mmol Tris at pH 7.9. Subsequently, the solution was incubated for 5 min at room temperature and added to a cuvette. Changes in the absorbance at 558 nm were followed during 5-8 min. Decrease in absorbance of phenol red by the acidification of medium was proportional to the liberation of fatty acids. The specific activity was calculated in Units (mM) of fatty acid per min. Controls included samples incubated with Alsever solution instead of venom. This assay provides information about presence and activity of phospholipases A2 in the venom of C. I. lepidus.
Statistical Analysis
All data are expressed as mean SEM. All experiments were performed in triplicate and three independent experiments were carried out. For the statistical analysis of results, we employed one-way analysis of variance (ANOVA) followed by Dunnett's test for specific comparisons against control values. Values of p<0.05 was accepted as significant levels.
Results
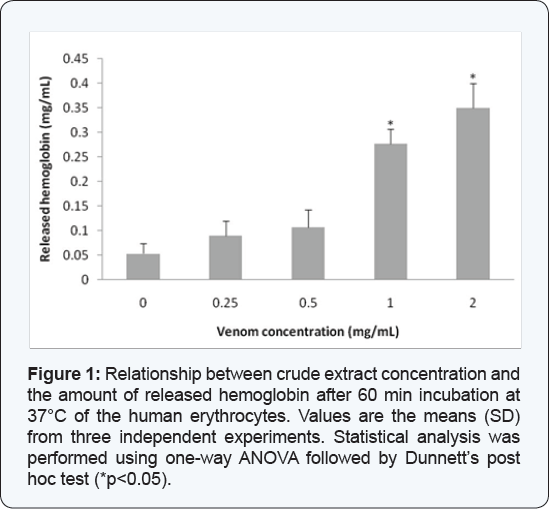
The crude extract of Crotalus lepidus lepidus added to the red blood cells resulted in a concentration-dependent hemolytic response, expressed as percentage of released hemoglobin, obtaining the following values: 0 mg/mL (0.05 mg Hb/mL), 0.25 mg/mL (0.09 mg Hb/mL), 0.5 mg/mL (0.106 mg Hb/mL), 1 mg/ mL (0.276 mg Hb/mL) and 2 mg/mL(0.35 mg Hb/mL) (Figure 1).
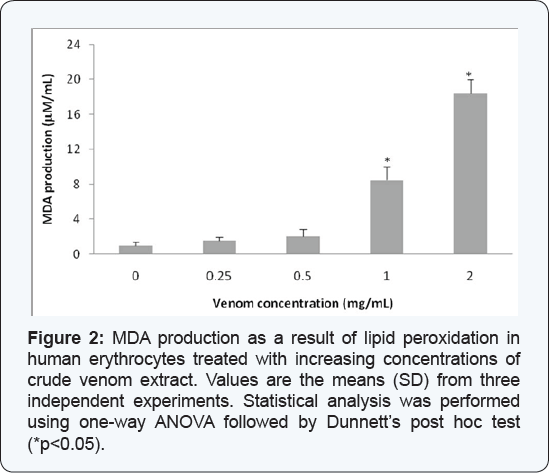
Lipid peroxidation in erythrocytes
Significant increases in the levels of peroxidized products (measured as MDA production), with respect to control group, were found in the blood cells exposed to concentrations of the crude venom of C. l. lepidus of 1.0 mg/mL (8.5 mM/mL) and 2.0 mg/mL (20.12 mM/mL) (Figure 2).
Phospholipase A2 activity
This assay was made to investigate presence and activity of phospholipases A2 in the venom of C. l. lepidus. The phospholipase A2 activity of the venom of C.l. lepidus (1mg/mL) was 0.041±0.0035 (mM/min). This concentration was chosen because it provoked the first increase in hemolysis and MDA production assays.
Discussion
The mottled rock rattlesnake (Crotalus lepidus lepidus) is one of four subspecies of C. lepidus. It is a relatively small rattle snake non aggressive although exhibits a wide range distribution includes southwest Texas, southeastern New Mexico and the Mexican Plateau to San Luis Potosi [14]. In venom of C.l. lepidus has been identified some partial sequences of metalloproteinases, galactose-specific lectins and phospholipases A2, as well produces hemorrhagic and lethal activities in mice [15]. The clinical effects of a case envenomation by C.l. lepidus include hemorrhagic effects, ecchymosis, soft tissue swelling, pain, thrombocytopenia and other hematological alterations [16]. Our results indicate that crude venom of C.l. lepidus in concentrations of 1.0 and 2.0 mg/mL is effective at producing toxicity in human erythrocytes in vitro, as indicated by hemolysis and lipid peroxidation. We employed the hemolytic response as a suitable tool to evidence cytotoxicity since some authors have reported that hemolysis represents a very sensitive test to assess and characterize phospholipases of snake venom [17,18] and the TBARs assays as a common index of lipid peroxidation [19,20] to assess the involvement of free radical formation during the cytotoxicity elicited by the crude venom of C.l. lepidus. In addition, we determined that C.l. lepidus venom possesses PLA2 activity, similar to the other Crotalus lepidus subspecies [15]. PLA2 enzymes are unique calcium-dependent hydrolytic enzymes on phospholipids, liberating free fatty acid and lysophospholipid [5,21]. Phospholipid hydrolysis by PLA2 enzyme also releases arachidonic acid whose metabolism results in the formation of potentially toxic reactive oxygen species and lipid peroxides. Red blood cells are particularly vulnerable to the attack by free radicals and therefore, they represent a suitable substrate to give evidence of oxidative stress [11,22]. Our results indicate that the venom of C.l. lepidus induces lipid peroxidation in human red blood cells. Erythrocytes lysis may be the end result of defects in the red blood cell membrane related to the peroxidative attack probably mediated by PLA2, suggesting that the peroxidative actions might, at least partially, contribute to the hemolytic effect. Interestingly, Norris [16] reported a clinical case of a bite in a person of 30 years by a captive specimen of C.l. lepidus who provokes a reduction in hematocrit and hemoglobin values in the patient. Erythrocytes are also susceptible to oxidative stress due to unsaturated membrane phospholipids, and the presence of hemoglobin and other hematin compounds may also augment the process of lipid peroxidation [23]. In addition, the erythrocytes have a high content of iron in hemoglobin, a powerful catalyst capable of initiating lipid peroxidation [11]. It's well known that lipid peroxidation produces oxidation of lipids, particularly polyunsaturated fatty acids and cholesterol [22,23]. In the lights of these results, the hemolytic and lipid peroxidation response to the crude venom was concentration- dependent, suggesting that part of the toxic action of the venom corresponds to the formation of free radicals mediated by PLA2, and hence the consequent changes in membrane structure after lipid peroxidation.
Conclusion
The findings of our study indicate that the crude venom from C.I. lepidus is able to induce hemolysis and lipid peroxidation in the isolated human red blood cells.
Acknowledgement
The financial grant was provided by the Secretaria de Educacion Publica, Subsecretaria de Mejoramiento del Profesorado, Programa de Mejoramiento del Profesorado. Mexico.
Conflict of Interest
Conflicts of Interest: There is no conflict.
References
- Marshall D. McCue (2005) Enzyme activities and biological functions of snake venoms. Applied Herpetology 2(2): 109-123.
- Kang TS, Georgieva D, Genov N, Murakami MT, Sinha M, et al. (2011) Enzymatic toxins from snake venom: structural characterization and mechanism of catalysis. FEBS J 278(23): 4544-4576.
- Prigent-Dachary J, Boffa MC, Boisseau MR, Dufourcq J (1980) Snake venom phospholipases A2. A fluorescence study of their binding to phospholipid vesicles correlation with their anticoagulant activities. J Biol Chem 255(16): 7734-7739.
- Six DA, Dennis EA (2000) The expanding super family of phospholipase A2 enzymes: classification and characterization. Biochim Biophys Acta 1488(1-2): 1-19.
- Dennis EA (1983) Phospholipases. In: Boyer (Eds.), The Enzymes. Academic Press, NY, USA, pp. 307-353.
- El-Asmar MF, Farag RM, Shoukry S, El Shimi IR (1979) Effect of scorpion (Leiurus quinquestriatus H. and E) venom on lipid peroxidation. Toxicon 17(3): 279-283.
- Dousset E, Carrega L, Steinberg JG, Clot-Faybesse O, Jouirou B (2005) Evidence that free radical generation occurs during scorpion envenomation. Comp Biochem Physiol C Toxicol Pharmacol 140(2): 221-226.
- Al Asmari A, Al Moutaery K, Manthari RA, Khan HA (2006) Time- course of lipid peroxidation in different organs of mice treated with Echispyramidum snake venom. J Biochem Mol Toxicol 20(2): 93-95.
- de Castro I, Burdmann EA, Seguro AC, Yu L (2004) Bothrops venom induces direct renal tubular injury: role for lipid peroxidation and prevention by antivenom. Toxicon 43(7): 833-839.
- Rice-Evans CA (1994) Formation of free radicals and mechanisms of action in normal biochemical processes and pathological states. In: Rice-Evans CA, Burdon RH (Eds.), Free Radical Damage and its Control. Elsevier Science BV, Amsterdam, USA, pp. 131-151.
- Chiu D, Lubin B, Shohet SB (1982) Peroxidative reactions in red cell biology. In: Pryor WA (Ed.), Free Radicals in Biology, Academic Press, New York, USA.
- Bradford MM (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein- dye binding. Anal Biochem 72: 248-254.
- Araujo AL, Radvanyi F (1987) Determination of phospholipase A2 activity by a colorimetric assay using a pH indicator. Toxicon 25(11): 1181-1188.
- Campbell JA, Lamar WW (2004) The Venomous Reptiles of the Western Hemisphere. Cornell University Press, Ithaca, NY, USA.
- Marti'nez-Romero G, Rucavado A, Lazcano D, Gutierrez JM, Borja M, et al. (2013) Comparison of venom composition and biological activities of the subspecies CrotaIus Iepidus Iepidus, CrotaIus Iepidus kIauberi and Crotalus lepidus morulus from Mexico. Toxicon 71: 84-95.
- Norris RL (2005) First report of a bite by the mottled rock rattlesnake (Crotalus lepidus lepidus). Toxicon 46(4): 414-417.
- Kini RM, Evans HJ (1987) Structure-function relationships of phospholipases. The anticoagulant region of phospholipases A2. J Biol Chem 262(30): 14402-14407.
- Al-Abdulla IH, SidkiAM, Landon J (1991) An indirect haemolytic assay for assessing anti venoms. Toxicon 29(8): 1043-1046.
- Sevanian A (1988) Lipid damage and repair, in oxidative damage and repair. In: Davies KJA (Ed.), Chemical, biological, and medical aspects. Pergamon Press, New York, USA, pp. 543-549.
- Draper HH, Hadley M (1990) MDA determination as an index of lipid peroxidation. Methods Enzymol 186: 421-430.
- Coles E, Mcilwain DL, Rapport MA (1974) The activity of pure phospholipase A2 from Crotalus atrox venom on myelin and on pure phospholipids. Biochim Biophys Acta 337(1): 68-78.
- Kupferberg, JP, Yokoyama S, Kezdy FJ (1981) The kinetics of the phospholipase A2-catalyzed hydrolysis of egg phosphatidylcholine in unilamellar vesicles. Product inhibition and its relief by serum albumin. J Biol Chem 256(12): 6274-6281.
- Niki E, Yoshida Y, Saito Y, Noguchi N (2005) Lipid peroxidation: Mechanisms, inhibition, and biological effects. Biochem Biophys Res Commun 338(1): 668-676.






























