Standardization of Centrifugation Parameters in the Preparation Canine Platelet Rich Plasma using Remi-K70 Blood Bag Centrifuge at TANUVAS Animal Blood Bank
Baranidharan GR* and Abhishek Kalundia
Department of Veterinary Clinical Medicine, TANUVAS Animal Blood Bank, Madras Veterinary College, Tamil Nadu Veterinary and Animal Sciences University, India
Submission: January 03, 2018; Published: March 01, 2018
*Corresponding author: Baranidharan GR, Department of Veterinary Clinical Medicine, TANUVAS Animal Blood Bank, Madras Veterinary College, Tamil Nadu Veterinary and Animal Sciences University, India, Email: Baranidharan GR* and Abhishek Kalundia
How to cite this article: Baranidharan GR, Abhishek Kalundia. Standardization of Centrifugation Parameters in the Preparation Canine Platelet Rich Plasma using Remi-K70 Blood Bag Centrifuge at TANUVAS Animal Blood Bank. Blood Res Transfus J. 2018; 2(1) : 555580. DOI: 10.19080/OABTJ.2018.01.555580.
Introduction
Bleeding disorders concurrent with haemaprotozoan diseases are often encountered in canine practices in India and hence forth the canine blood component transfusion is slowly gaining momentum for countering the haematological emergencies Bleeding disorders in dogs can either be due to defects in primary hemostasis (Platelets) or secondary hemostasis (Clotting factors present in the plasma). Canine Platelet-Rich Plasma (PRP) is a blood product prepared form dog whole blood containing both platelets and plasma, indicated for bleeding emergencies, severe thrombocytopenia, thrombopathia complicated with clotting factor deficiencies or defects [1,2]. Platelets, in immune-mediated thrombocytopenia are destroyed rapidly in the liver and spleen, where platelet-rich plasma can provide an immediate short term local hemostatic effect at sites of bleeding thereby reducing mortalities [3,4].
Though standardization has been has been established in human transfusion medicine globally [5-11] and veterinary transfusion medicine in the western countries, no much work has been done till date in India in the standardization of preparation of canine PRP. The centrifugation speeds and other parameters are very machine dependent and the study was carried out using Remi K70 blood bag refrigerated centrifuge.
In dogs, the normal platelet concentration is 2-5 lakh platelets per microliter [12] and below 1 lakh is considered as mild thrombocytopenia (TCP), 25,000 to 50,000cells/cm, moderate TCP and below 25,000cells/cm severe TCP. The dogs showing bleeding tendencies like petechiation, echymosis, purpura, epistaxis, haematuria, haemoglobinuria, melena, pulmonary haemorhage need emergency PRP transfusion. The PRP concentration of platelets is 3-8 times more. To obtain platelet-rich plasma, the donor’s blood is mixed with an anticoagulant and processed either manually by spinning it in a centrifuge to separate its components (centrifugation) or through an automated system. This processed fraction of the blood is termed "platelet-rich plasma" [13]. The goal is to achieve the highest concentrations of platelets and remove the other components of the blood such as the red and white blood cells.
Aim
The aim of this study was to standardize the centrifuge speeds, acceleration, deceleration, temperature control and duration involved in the PRP preparation, using Remi-K70 blood bag centrifuge. Feldman and Sink (2008) reviewed the centrifugation techniques and suggested that 2000g for 3 min at 22 - 25 °C would be ideal for PRP separation.
Materials and Methods
Platelet Rich Plasma is made from one unit of Fresh Whole Blood using 350ml triple or quadruple blood bags i.e., one collection bag containing Citrate Phosphate Dextrose Adenine (CPDA) 49ml, one transfer bag containing Saline Adenine Glucose Mannitol (SAGM) 73ml, one Tri-Octyl-Tri-Mellitate (TOTM) bag for platelet collection and storage.
Collection
350ml of whole blood was collected from the dog blood donors in triple or quadruple blood bags and was processed immediately to harvest viable platelets.
Sealing
The blood collection line was sealed using Terumo Penpol blood bag sealerTM. Multiple seals were made with each segment containing the similar blood bag number to enable blood sample testing. Each segment was folded end to end and gathered with a rubber band to prevent getting entangled during the blood processing procedure. All the blood bags including the satellite bags were labeled with the component name, collection date, donor number and expiration date, their respective blood group i.e., Dog Erythrocytic Antigen (DEA) 1.1 negative or positive using a permanent marker. The entire unit of blood and attached satellite bags were weighed accurately with the Terumo ComposcaleTM for balancing the centrifuge.
Centrifugation
After sealing, the Blood bag units with the labels facing out were placed in Remi Blood bag centrifugeTM buckets carefully. The whole blood unit was centrifuged using a light spin in a centrifuge at 2000g for 10 minutes at 22 °C.
The centrifuge was allowed to stop spinning without operator intervention because any acute stop of the rotor, including brake use, would disturb the red cell/plasma line thereby contaminating the plasma with red cells.
Platelet Rich Plasma (PRP) Expression
The unit of whole blood was removed from the centrifuge without agitation so as not to disturb red cells and plasma and placed on a plasma extractor (Plasma Expressor, Terumo®).
o The hinged plate is attached to the stand and was released to apply pressure to the unit of whole blood in order to express the plasma into a satellite bag.
o An empty satellite bag or the desired satellite bag was placed on a balance. The weight was tarred to zero. The Platelet Rich Plasma will be expressed into the empty satellite bag.
o The seal at the port of the parent blood bag containing the three layers (plasma, platelets and the RBCs) of the collected blood, was broken manually. Following which, the upper layers containing the plasma and the platelets started flowing into the above mentioned satellite bag gradually. The harvested Platelet Rich Plasma was light yellow in color and did not contain visible red cell contamination.
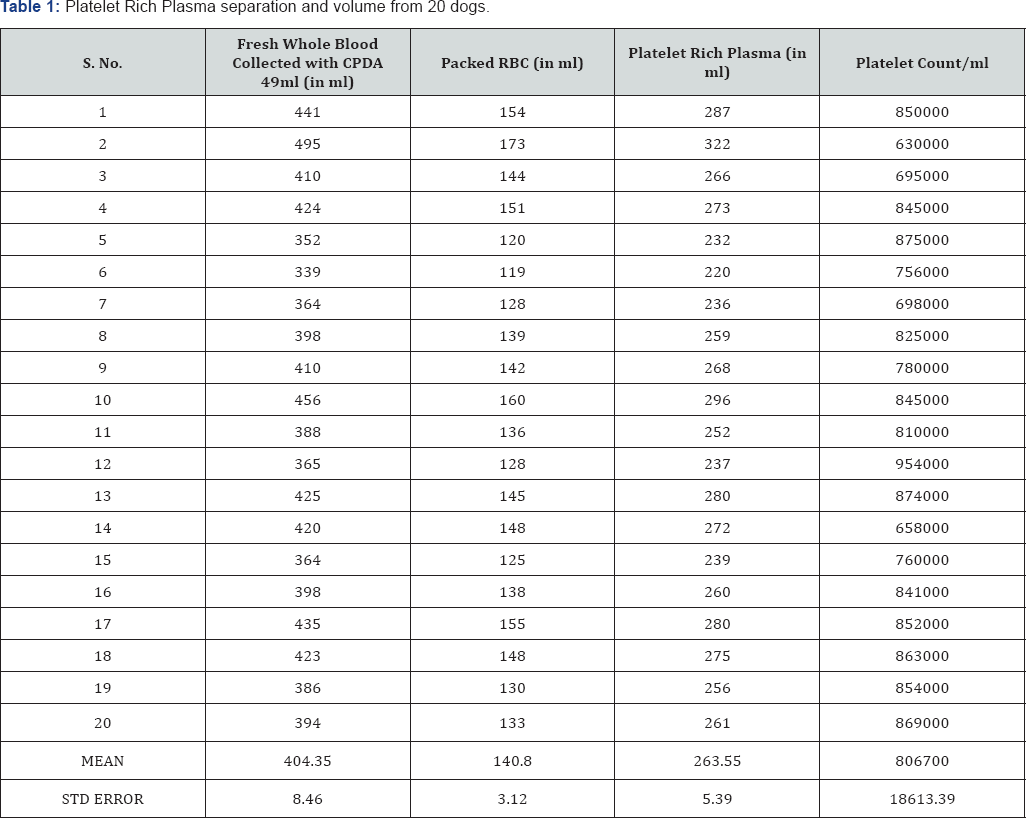
o Once harvested, the line was clamped immediately and carefully using a clamp or a hemostat artery forceps (one click) and sealed (Table 1).
Labeling and Storage
o The volume of both packed RBC (pRBC) and PRP were determined in milliliters using Compo Scale, TERUMO and recorded.
o In order to preserve platelet viability, Platelet Rich Plasma was allowed to rest at room temperature maximum for 1 - 2 hours and transfused as soon as possible thereafter.
Discussion
PRP therapy is one of the mainstay in most of the cases wherein the dog/puppy is suffering with severe hypovolaemic shock due to various medical emergencies like haemoprotozoan diseases, Canine parvo virus infection, Immune mediated thrombocytopenia, non-overt Disseminated Intravascular coagulopathy and cancers. In this study, it was prepared by a process known as differential centrifugation. In differential centrifugation, acceleration force is adjusted to sediment certain cellular constituents based on different specific gravity and there are many ways of preparing PRP (PRP method or by the buffy-coat method). It was seen that at 22 °C for 10min and at 2200RPM it is possible to successfully harvest an average of eighty thousand platelet count per unit which was in accordance with the findings of Schneider, 1995. This harvest (PRP) can be given @ 10ml/kg B.wt. in dogs [11]. Platelet-rich plasma (PRP) has been utilized in surgery for 2 decades in human medicine and urgical cases. PRP contains growth factors and bioactive proteins that influence the healing of tendon, ligament, muscle, and bone. In veterinary science, there has been a recent interest in the use of PRP for the treatment of skin related injuries and osteoarthritis in horses and dogs [4, 14-21].
Conclusion
The incidences of bleeding tendencies in dogs with haemoprotozoan diseases have increased lately resulting in progressive demand for blood and blood components in India. This article will enlighten the standardization of therapeutic canine Platelet Rich Plasma preparation and the centrifugation speeds have been enlisted which would prove very useful for tertiary veterinary hospitals and institutes which would establish animal blood banks in the near future.
Acknowledgement
The authors are thankful to the Vice Chancellor, Director of Clinics, TANUVAS, Chennai 51, Dean, Madras Veterinary College, Chennai for their immense support for the establishment of TANUVAS Animal Blood Bank, Chennai.
References
- Abrams-Ogg C, Kruth S, Carter R, Valli V, Kamel-Reid S, Dube I (1993) Preparation and transfusion of canine platelet concentrates. Am J Vet Res 54(4): 635-642.
- Allyson K, Abrams-Ogg A, Johnstone I (1997) Room temperature storage and cryopreservation of canine platelet concentrates. Am J Vet Res 58(11): 1338-1347.
- Adler SC, Kent KJ (2002) Enhancing healing with growth Facial factors. Plast Surg Clin North Am 10(2): 129-146.
- Kaufman P (1992) Supplies for Blood Transfusions in Dogs and Cats. Probl Vet Med 4(4): 582-593.
- Berg U, Bang P (2004) Exercise and circulating insulin-like growth factor-1. Horm Res 62(1): 50-58.
- Knighton DR, Ciresi KF, Fiegel VD, Austin LL, Butler EL (1986) Classification and treatment of chronic nonhealing wounds: successful treatment with autologous platelet-derived wound healing factors (PDWHF). Ann Surg 204(3): 322-330.
- Lee C, Henry J (1979) Immunology, Blood Banking and Hemotherapy. In: Henry J (Eds.) Clinical Diagnosis and Management by Laboratory Methods (16th edn). Philadelphia: WB Saunders; pp.1460.
- Murray MM, Spindler KP, Devin C, Snyder BS, Muller J, et al. (2006) Use of a collagen-platelet rich plasma scaffold to stimulate healing of a central defect in the canine ACL. Journal of orthopaedic research 24(4): 820-830.
- Venglen-Tyler V (1999) Blood Component Preparation, Storage, Shipping and Transportation. In: Technical Manual. Technical Manual (13th edn), American Association of Blood Banks, Bethesda, pp. 161192.
- Venglen-Tyler V (1999) Blood Collection, Storage and Component Preparation. Technical Manual (13th edn), American Association of Blood Banks, Bethesda, pp. 711-737.
- Zimmerman K (2010) Drug-Induced Thrombocytopenias. In: Feldman B, Zinkl J, et al. (6th edn) Schalm’s Veterinary Hematology, Willey- Blackwell Publication, pp. 1132.
- Jain N (1993) The Platelets. Essentials of Veterinary Hematology. Philadelphia: Lea and Febiger, pp. 105-132.
- Nolte I, Mischke R (1995) Investigations of platelet aggregation and platelet counts from stored canine whole blood. Res Vet Sci 58: 190192.
- Feldman BF, Sink CA (2008) Practical Transfusion Medicine. IN: Feldman BF, Sink CA (Eds.). Publisher: Teton NewMedia, Jackson, WY, USA.
- Conley C (1961) Blood Platelets and Platelet Transfusions. Arch Intern Med 107: 635-638.
- Feldman B, Kristensen A (1995) Modern Veterinary Blood Banking Practices and Their Applications in Companion Animal Practice. In: Kristensen A, Feldman B (Eds.), The Veterinary Clinics of North America Small Animal Practice Canine and Feline Transfusion Medicine. WB Saunders, Philadelphia, pp.1231-1243.
- Kristensen A, Feldman B (1995) General Principles of Small Animal Blood Component Administration. In: Kristensen A, Feldman B (Eds.), The Veterinary Clinics of North America Small Animal Practice Canine and Feline Transfusion Medicine. WB Saunders, Philadelphia, pp. 1277-1290.
- Messora R, Nagata MJ, Furlaneto APC, Dornelles ACM, Bomfim SRM, et al. (2011) A standardized research protocol for platelet-rich plasma (PRP) preparation in rats. RSBO 8(3): 299-304.
- Schneider A (1995) Blood Components Collection, Processing and Storage. In: Kristensen A, Feldman B (Eds.), The Veterinary Clinics of North America Small Animal Practice Canine and Feline Transfusion Medicine, WB Saunders, Philadelphia, pp.1245-1261.
- Schneider A (2000) Principles of Blood Collection and Processing In: Feldman B, Zinkl J et al. (Eds.), Schalm's Veterinary Hematology (5th edn), Lippincott Williams and Wilkins, Philadelphia, pp. 827-832.
- Spindler KP, Murray MM, Devin C, Nanney LB, Davidson JM (2006) The central ACL defect as a model for failure of intra-articular healing. Journal of orthopaedic research 24(3): 401-406.






























