Splanchnic Vein Thrombosis: Risk Factors, Clinical Presentation, Diagnosis, Treatment
Vasileios Perifanis, Nikolaos Neokleous*, Andreas Protopappas, Georgia Kaiafa and Christos Savopoulos
Department of Internal Medicine, AHEPA University Hospital, Aristotle University of Thessaloniki, Thessaloniki, Greece
Submission: January 31, 2018; Published: February 23, 2018
*Corresponding author: Nikolaos Neokleous, Department of Internal Medicine, AHEPA University Hospital, Aristotle University of Thessaloniki, Greece, Email: nneokleous@yahoo.gr
How to cite this article: Vasileios P, Nikolaos N, Andreas P, Georgia K, Christos S. Splanchnic Vein Thrombosis: Risk Factors, Clinical Presentation, Diagnosis, Treatment. Blood Res Transfus 006 ransfus J. 2018; 2(1) : 555577. DOI: 10.19080/OABTJ.2018.02.555578
Abstract
Splanchnic vein thrombosis is an uncommon manifestation of venous thromboembolism which encompasses one or more abdominal veins (portal, splenic, mesenteric and supra-hepatic veins). Splanchnic vein thrombosis may be related to variable underlying disorders, either local (abdominal cancer, liver cirrhosis, intra-abdominal inflammation or surgery) or systemic (hormonal treatment, thrombophilia conditions, myeloproliferative neoplasms, paroxysmal nocturnal haemoglobinuria or autoimmune diseases). Splanchnic vein thrombosis occurs with heterogeneous clinical presentations, extending from incidental finding to symptomatic thrombosis associated with gastrointestinal bleeding. Due to diagnostic and clinical complexity there is a clinical challenge for treatment decisions. In the absence of major contraindications, anticoagulant therapy is usually suggested for all patients presenting with acute symptomatic splanchnic vein thrombosis, but there is no recommendation about the use of anticoagulant drugs in chronic or incidentally detected episodes. There is no clear evidence on the acute and long-term management and the ratio between the risk and the benefit of using anticoagulant therapy in splanchnic vein thrombosis still needs to be well evaluated.
Keywords: Splanchnic vein thrombosis; Diagnosis; Risk factors; JAK2; Anticoagulation
Abbreviations: SVT: Splanchnic Vein Thrombosis; VTE: Venous Thromboembolism; BCS: Budd-Chiari Syndrome; PVT: Portal Vein Thrombosis; MVT: Mesenteric Vein Thrombosis; spVT: Splenic Vein Thrombosis; PNH: Paroxysmal Nocturnal Haemoglobinuria
Introduction
Splanchnic vein thrombosis (SVT) is generally defined as an uncommon manifestation of venous thromboembolism (VTE). Budd-Chiari syndrome (BCS), portal vein thrombosis (PVT), mesenteric vein thrombosis (MVT) and splenic vein thrombosis (spVT) are varied expressions of SVT with abnormal pathophysiology, clinical presentation and prognosis [1]. SVT is possibly under diagnosed, thus presenting heterogeneously and the asymptomatic incidental findings rate is unexpectedly significant. The epidemiology varies greatly depending on data sources. PVT is the most frequent manifestation in the spectrum of SVT [2]. On the contrary, BCS represents the least frequent variant with an estimated incidence of slightly less than 1 case per million people per year [3]. SVT is a potentially life-threatening disease. The clinical presentations are variable and may include the presence of extensive thrombosis and gastrointestinal bleeding. Abdominal pain is the most frequently reported symptom, with a prevalence varying from 40% in patients with PVT to more than 60% in patients with MVT [4]. Acute MVT is associated with intestinal infarction in almost one-third of patients and has a mortality rate of 20% at 30 days [5] . Other common clinical manifestations are gastrointestinal bleeding and ascites, found in one-fourth of patients, mainly PVT and BCS respectively, and caused by portal hypertension [4]. However, in a large cohort of 832 patients diagnosed with SVT 18% were asymptomatic [4].
Risk factors
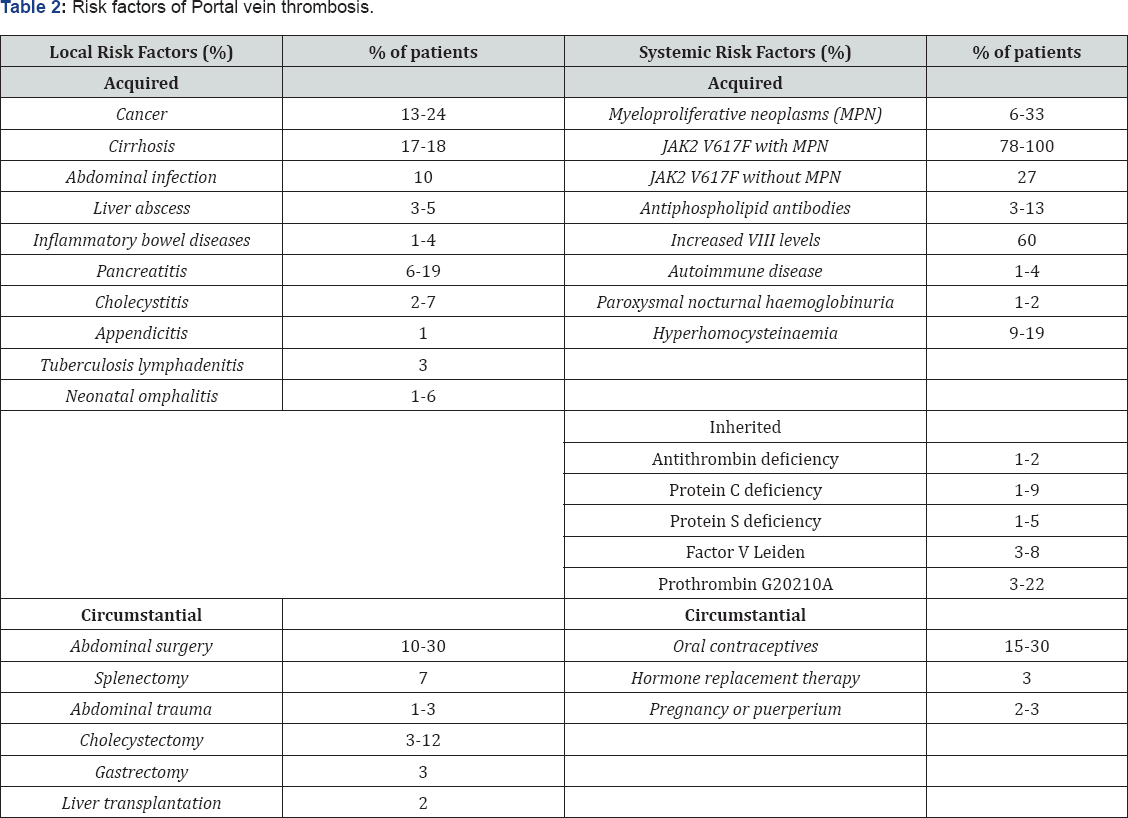
SVT may be connected with different underlying disorders, either local or systemic. The relative incidence of risk factors varies according to age, economic status, geographical area, and thrombosis location.
Budd-Chiari syndrome
The BCS is defined as any obstruction of the hepatic outflow. It can be occurred at any region along the circulation flow from the small hepatic veins to the junction of inferior vena cava to the right atrium. Hepatic veno-occlusive disease (sinusoidal obstruction syndrome) or cardiac disorders concomitant with right heart failure, which can lead to outflow obstruction are excluded from BCS [6]. BCS has a prevalence of 1.4 per million individuals in Western countries [3] and 2.4 per million in Japan [7]. BCS is considered primary when obstruction of the hepatic venous outflow tract is the result of an endoluminal venous lesion (thrombosis or web). In contrast, BCS is considered secondary when the obstruction is caused by the presence of extravascular material (tumour or parasitic mass) in the lumen or from extrinsic compression (abscesses, cysts, tumours). The prevalence of each underlying disease is varied world widely (Table 1). Common systemic risk factors for BCS are the use of oral contraceptives and pregnancy or puerperium; the former being more prevalent in the Western countries and the latter in the Eastern countries [8]. Unusual risk factors of the Mediterranean area include Behcet’s disease, inflammatory bowel diseases and parasites, while membranous webs of the inferior vena cava are typically found in Asian patients [8]. Among inherited thrombophilias, deficiencies of antithrombin, protein C and protein S are barely diagnosed, because of liver deficiency with impaired production of coagulation factors. Furthermore, Factor V Leiden mutation has shown stronger association with BCS [6] . In the last decades, myeloproliferative neoplasms (MPN) emerged as the major systemic cause of SVT, diagnosed in half of BCS patients [9-12]. Sozer et al. [13] and Rosti et al. [14] showed that the endothelium of splanchnic vessels expresses the JAK2 mutations and this condition could provoke a localised endothelial dysfunction that leads to thrombotic pathogenesis [13,14]. Even in the absence of known MPN, the presence of JAK2 V617F, a mutation of the tyrosine-kinase JAK2 that is strongly associated with the development of MPN [15], appears to have an important role also in the development of SVT [16]. However there are still difficulties in diagnosis of MPN among patients with SVT. This is mainly because splenomegaly is also equally sign of both conditions, the high haemoglobin of MPN usually is masked by the gastric haemorrhage and/or the hypersplenism, and the BCS patients have high levels of erythropoietin due to hepatic ischaemia. Bone biopsy is necessary to give solution and diagnosis; on the other hand red cell mass measurement is only needed when JAK2 mutations are existed.

Recently, an association between SVT and paroxysmal nocturnal haemoglobinuria (PNH) has also been reported. PNH is a rare acquired haematological disorder characterized by non- malignant clonal expansion of hematopoietic stem cells lacking the glycosylphosphatidylinositol-anchored proteins at the cell membrane, particularly complement inhibitors CD55 and CD59 [17]. Clinical manifestations include haemolytic anaemia, bone marrow failure and VTE. In these patients, VTE more commonly occurs in unusual sites, such as the splanchnic veins where PNH has been reported in up to 10% of BCS patients [18], and represents the major cause of morbidity and mortality in PNH patients [17]. IgG anticardiolipin antibodies have lately been associated with BCS.
Portal vein thrombosis
Portal vein thrombosis (PVT) is defined as extra-hepatic portal vein obstruction that may happen with or without the involvement of the intra-hepatic portal, splenic or superior mesenteric veins, formation of portal cavernoma and development of portal hypertension [6]. The annual incidence of PVT was less than four per million individuals [19], but a recent autopsy study estimates it at approximately three individuals per thousand [2]. A local triggering factor is found in 21-60% of those with PVT [20-22], essentially liver cirrhosis, hepatocarcinoma or other abdominal tumours, inflammatory diseases and abdominal surgery (Table 2). Recently, it was reviewed that PVT complicates hepatic cirrhosis in 10-20% of patients [23]. Interestingly, 5-8% of patients after splenectomy develop PVT, especially these with underlying myeloproliferative neoplasms or haemolytic anaemia [24]. Among the systemic risk factors myeloproliferative neoplasms are the primary cause of PVT, since they are found in one third of the patients 10-12,22,25]. A recent meta-analysis concerning prevalence of inherited factors in patients with PVT, showed a 4.5-fold and 2-fold increased risk of PVT for prothrombin G20210A and factor V Leiden, respectively [26].
Mesenteric vein thrombosis
Isolated Mesenteric vein thrombosis (MVT) without PVT or splenic vein thrombosis (SVT) is rare [27,28]. The annual incidence of superior mesenteric vein thrombosis is less than three per 100,000 individuals [5]. In 2005 a population based study showed thrombophilia markers in 67%, a local factor (surgery or inflammation) in 25%, cancer in 24%, and use of oral contraceptives in 6% of patients with MVT. Three years later an autopsy series showed the presence of abdominal cancer in 22% and liver cirrhosis in 17% of cases [5,27].
Splenic vein thrombosis
The most common cause of isolated thrombosis of the splenic vein is chronic pancreatitis caused by perivenous inflammation [29]. Splenic vein thrombosis (spVT) should be suspected in the following groups of patients:
i. Those with a history of pancreatitis and newly diagnosed GI bleeding,
ii. Patients with splenomegaly but no portal hypertension, cirrhosis, or hematologic disease, and
iii. Individuals with isolated gastric varices [30]. Although early reports found that the most common cause of spVT was pancreatic carcinoma, recent reviews have revealed acute or chronic pancreatitis to be the probable cause of spVT in the majority of cases [30,31]. The diffuse inflammation of pancreatitis is associated with the onset of spVT, although the diagnosis of spVT in these cases is not always made during the acute attack [29,30]. This suggests that recurrent indolent episodes of pancreatitis lead gradually to spVT. Other causes of spVT include adenopathy from metastatic carcinoma, lymphoma, ulcerative colitis and iatrogenic causes after surgery such as splenectomy, partial gastrectomy, and distal splenorenal shunt [30]. Independently on the cause the pathophysiology remains the same and includes diffuse local inflammation or obstruction due to extrinsic compression.
Clinical Presentation
Budd-Chiari syndrome
The majority of patients have hepatomegaly, splenomegaly, right upper abdominal quadrant pain, and ascites. In chronic disease forms, jaundice and elevation of the aminotransferase liver function tests could also be seen [9]. All the symptoms are based on the post-hepatic obstruction associated with BCS that leads to increased sinusoidal pressure, which may cause perisinusoidal necrosis and eventually liver failure. In 14-18% of cases, portal hypertension can develop when PVT is present [10].
Depending on the extension and rapidity of the outflow obstruction, as well as liver decompression via a collateral blood flow, BCS can be classified as fulminant, acute, subacute or chronic [32]. The chronic form of BCS is the most common form as it occurs in 60% of cases, usually with symptoms of portal hypertension and hepatic cirrhosis [33]. Acute and subacute BCS is reported in 20% of patients, and is associated with symptoms that last for a short duration, such as ascites and hepatic necrosis, without the formation of venous collaterals. Fulminant BCS is rare (5% of cases) and is associated with a quick onset, hepatocellular necrosis, and hepatic encephalopathy. The remaining 15% of BCS patients are asymptomatic due to collateral blood flow which can preserve the hepatic outflow [34]. However, the prevalence of asymptomatic patients recently was reported to be notably lower (3%) [10]. The overall survival of patients with BCS was 57-62% [35] in ten years follow- up. The PVT co-existence is worsening the prognosis [36]. Encephalopathy, ascites, prolongation of the prothrombin time, and elevated bilirubin were also independently associated with poor prognosis [36].
Portal vein thrombosis
Presentation of PVT can be acute or chronic. Acute thrombosis is characterised by abdominal pain, nausea, fever and diarrhoea, with no evidence of portal hypertension. In the absence of liver disease, liver function tests are normal due to increase arterial hepatic flow and the development of collateral veins. Intestinal ischaemia and bowel infarction can also be seen if MVT is present [37]. Nevertheless, PVT may also be asymptomatic, and incidentally diagnosed. The chronic form [20,21] is characterised by portal cavernoma, portal hypertension with hypersplenism and splenomegaly, ascites, gastrointestinal bleeding from oesophageal varices and collateral portosystemic circulation [38]. Other less frequent manifestations of a chronic PVT include portal cholangiopathy and hepatic encephalopathy [20]. The overall survival of patients with portal vein thrombosis varies between 54% and 81% after 10 years, depended on the presence of cancer, cirrhosis and MVT [25].
Mesenteric vein thrombosis
In the vast majority of patients, the onset of a MVT is characterised by acute abdominal pain. Other common symptoms include diarrhoea, nausea, vomiting, and lower gastrointestinal bleeding [39]. A MVT presentation can be acute, subacute, or chronic [39]. The one-third of patients with acute thrombosis is associated with a bowel infarction [5]. Patients with chronic MVT are presented without acute abdominal pain since they have developed venous collateral circulation [39]. A MTV is associated with PVT in 65% of patients [27]. The occlusion of both vessels is associated with a significant decrease in survival rate at 5 years when compared to a PVT alone (41 vs. 62%) [25], or to a MVT alone (20% vs 62%) [5].
Splenic vein thrombosis
Sinistral portal hypertension caused by spVT can cause massive gastrointestinal bleeding from oesophageal or gastric varices or hypertensive gastropathy. Unlike patients with generalized portal hypertension, most patients with SVT are asymptomatic and have normal hepatic function. Usually, due to pancreas involvement in the pathophysiology, the symptoms from the pancreatic impairment are leading [29].
Diagnosis
The occlusion of the hepatic veins, inferior vena cava, or both is the keystone of BCS diagnosis. Doppler ultrasound can detect almost 90% of the cases [40]. Typical ultrasound findings include liver lobe enlargement which compress inferior vena cava, liver inhomogeneity, intrahepatic collateral vessels and hypervascular nodules [40]. When definite diagnosis by ultrasound was dubious, then CT scan, MRI, hepatic venography and a liver biopsy are required. The presence of ascites, the patency of the vessels and the splanchnic hypertrophy can easily be evaluated by CT and MRI scans. Patchy liver is characteristic of acute BCS while multiple regenerative nodules are detected in chronic BCS. Venography allows for pressure measurements, while a liver biopsy can exclude other liver disease, such as veno- occlusive disease and cirrhosis of other aetiologies [32].
Overall sensitivity and specificity for detection of portal vein thrombosis are 89% and 92%. Colour flow Doppler sonograms can show flow around a thrombus that partially blocks the vein. CT scans usually show PVT as a non-enhancing filling defect within the lumen of the portal vein [41]. The hepatic segment supplied by the occluded portal venous branch can appear relatively hypodense. On gradient-echo MR images, PVT is usually seen as an area of diminished intravascular signal with intensity similar to that of adjacent stationary soft tissues.Angiography is used for preoperative vascular mapping in candidates for surgery [42].
CT and MRI scans should be considered the primary diagnostic modalities for evaluating patients with high clinical suspicion of nonsurgical mesenteric ischemia. On the other hand, ultrasound seems to be powerless against overlying bowel gas [42].
According splenic vein thrombosis, the most indicated imaging strategy seems to be CT scan and specially CT angiography. MRI and ultrasound have been also used with ambivalent sensitivity and specificity [42].
Prognosis and Recurrence
The prognosis of SVT depends on the extension of disease and on the presence of underlying disorders. A recent study examined 832 patients with SVT at any anatomic site and showed that the 10-year survival rate was 60%, and older age, active cancer, and MPN were independent predictors of mortality. In the same study the cumulative incidence of recurrence at 10 years was 24%, and the annual incidence of major bleeding events was 6.9/100 patient-years [4].
Other studies examined specific patients with BCS. One of them reported that overall survival at one, 5 and 10 years was 82%, 69%, and 62%, respectively. Encephalopathy, ascites, prolongation of the prothrombin time, and elevated bilirubin were independently associated with poor prognosis [43]. Another one showed that the survival rate was 87% at 1 year and 82% at 2 years [10]. The frequency of recurrent thrombosis or major bleeding was not reported in these studies.
A recent study examined the mortality rate of patients with PVT. It reported that the overall survival was 69% at 1 year and 54% at 5 years. The exclusion of patients with cancer or cirrhosis increased the rates to 92% and 76%, respectively [44]. Age, bilirubin, cirrhosis, and malignancy were associated with poor prognosis. In a study of 136 patients with non-malignant, no cirrhotic PVT, the frequency of thrombotic events was 5.5/100 patient-years, and the occurrence of any gastrointestinal bleeding was 12.5/100 patient-years. Large varices were strongly related with bleeding episodes, whereas the presence of an underlying prothrombotic state along with the absence of anticoagulant therapy were related with a higher risk for relapse [38]. In a prospective study of patients with PVT without cirrhosis or solid cancer, who were receiving anticoagulant therapy, the 1-year recanalization rate of the portal vein was achieved in 38% of patients, and all recanalizations occurred in the first 6 months of treatment [22]. In another study it was examined the recurrence rate to patients with PVT. The overall risk for recurrent thrombosis was 3% at 1 year, 8% at 5 years, and 24% at 10 years. The overall risk for gastrointestinal bleeding was 33% at 1 year, 43% at 5 years, and 46% at 10 years, with the majority of bleeds being variceal [45].
A few small cohort studies have stated survival in patients with MVT. In a study of 51 patients, 30-day mortality was 20% [5]. Intestinal infarction at the time of diagnosis was associated with poor prognosis, and cancer was also associated with mortality [5]. Another retrospective study of 68 patients reported approximately the same 30-day mortality rate and reported malignancy, advanced age, and symptom duration to be independent predictors of mortality [46].
Treatment
The management of SVT largely depends on the presence of predisposing conditions and the patient's comorbidities. The management of SVT requires a multidisciplinary approach that may include a gastroenterologist/hepatologist, a haematologist and/or a thrombosis expert, and an interventional radiologist and/or a surgeon.
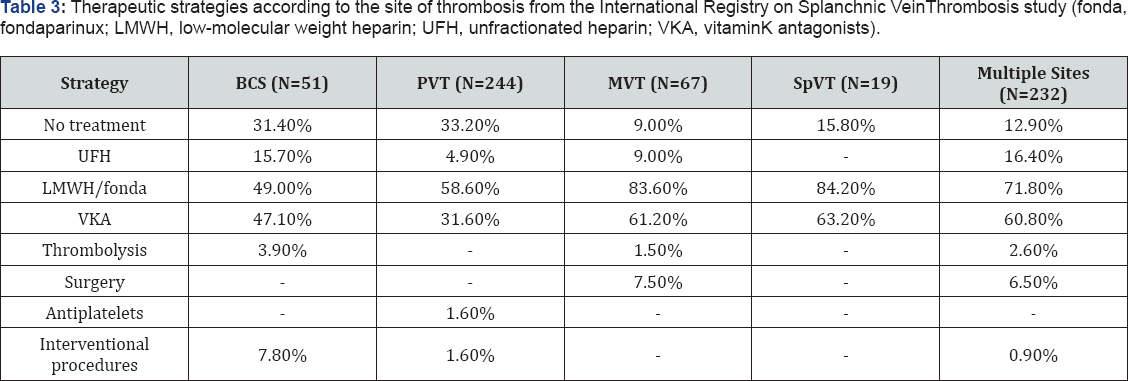
Ageno et al. [47] have recently completed an international registry defining treatment strategies and factors associated with therapeutic suggestions in patients with SVT (Table 3) [47].
Therapeutic decisions in BCS include antithrombotic (anticoagulants and diuretics) and surgical techniques (local factors and factors that control the intravascular pressure thrombolysis, percutaneous transluminal angioplasty, surgical or transjugular intrahepatic porto-systemic shunting and orthotopic liver transplantation) [32,48]. Beta blockers are also used to establish appropriate prophylaxis of bleeding along with endoscopic treatment of oesophageal and gastric varices [32,48]. Darwish-Murad et al. [10] showed that many patients were safely managed with medical treatments (86% anticoagulation and61% diuretics), but 51% required surgical operations, which are recommended in case of worsening despite medical treatment [10].
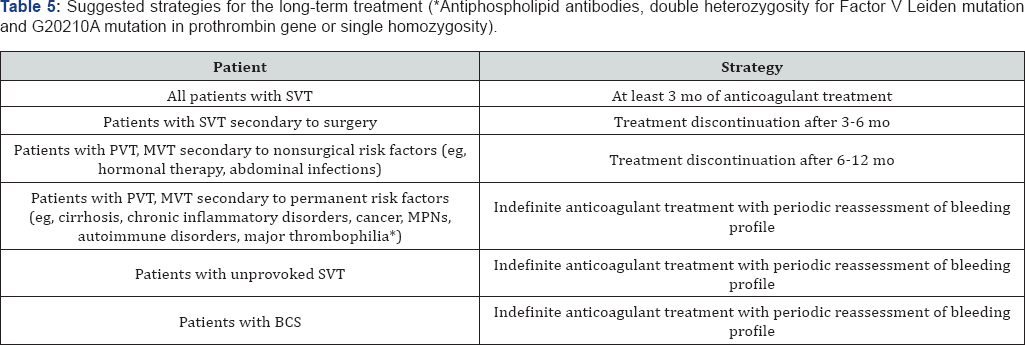
The use of anticoagulation in SVT still remains a controversial issue, due to the complex balance between the increased bleeding risk (oesophageal varices and thrombocytopenia) and the thrombotic predisposition (cirrhosis and malignancy). In the absence of major contraindications, anticoagulant therapy is generally recommended for all patients presenting with acute symptomatic SVT, starting with either low-molecular weight or unfractionated heparin and continuing with the vitamin K antagonists in most patients [49,50] (Table 4). It is generally recommended that anticoagulant treatment should be continued for at least 3 months, or indefinitely if underlying persistent prothrombotic factors are identified. There is no consensus about the use of anticoagulant drugs in chronic SVT, which presents with variceal bleeding and hypersplenism but without signs of recent occlusion. On the other hand, gastro-oesophageal haemorrhages are a common and feared complication of SVT. However, Thatipelli et al. [4] showed that warfarin therapy was independently associated [4] with bleeding. The optimal duration of oral anticoagulant therapy is challenging. The introduction in the 1980s of systematic use of VKA in patients with BCS has coincided with a better prognosis [32,35], although the benefit of oral anticoagulation on survival of the most severe patients is uncertain [43]. However, if a thrombophilic factor or JAK2 mutation is present, the anticoagulation therapy should be continued for life (Table 5) [51].

Thrombolysis remains an open issue. In patients with BCS, systemic thrombolytic therapy with tissue plasminogen activator is of little value, but catheter-directed thrombolysis appears to be effective in acute and partially occlusive thrombosis [52].
Also acute PVT and MVT require prompt anticoagulation. Basic management for PVT patients includes screening for oesophageal varices and treating complications of portal hypertension and portal cholangiopathy. Local thrombolysis may also be effective in patients with PVT and MVT [53,54]. Transjugular intrahepatic portosystemic shunt is minimally invasive and has been used also for patients with non- cavernomatous EHPVO [55]. The fear of bleeding complication exists here too specially in chronic cases. The decision is taken by balancing the pros and cons of using anticoagulation bearing in mind that bleeding could be fatal in many cases.
Conclusion
SVT is a challenging disease, with various and multifaceted clinical presentations, multifactorial pathophysiology, high rate of incidental findings and potentially life-threatening complications. Recent evidence recommends that, in the absence of major risk factors, screening for thrombophilic conditions, JAK2 mutation and PNH clone should always be performed. On the other hand liver biopsy seems to be individually used. Anticoagulant treatment is generally recommended for all patients presenting with acute symptomatic SVT, in the absence of major contraindications. Long-term management is still a controversial issue mainly due to the lack of high quality evidence on the acute SVT. Further research is needed to better evaluate the risk to benefit-ratio of anticoagulant therapy in SVT Likewise, the role of specific inhibitors on thrombotic episodes, otherwise used in cases with JAK2 mutations and PNH clone, has not yet been assessed.
References
- Riva N, Donadini M, Dentali F, Squizzato A, Ageno W (2012) "Clinical approach to splanchnic vein thrombosis: risk factors and treatment. Thromb Res 130: S1-3.
- Ögren M, Bergqvist D, Bjorck M, Acosta S, Eriksson H, et al. (2006) "Portal vein thrombosis: Prevalence, patient characteristics and lifetime risk: A population study based on 23 796 consecutive autopsies.” World J Gastroenterol 12: 2115-2119.
- Rajani R, Melin T, Bjornsson E, Broome U, Sangfelt P, et al. (2009) "Budd- Chiari syndrome in Sweden: epidemiology, clinical characteristics and survival-an 18-year experience.” Liver Int 29(2): 253-259.
- Thatipelli M, McBane R, Hodge D, Wysokinski W (2010) "Survival and Recurrence in Patients With Splanchnic Vein Thromboses.” Clin Gastroenterol Hepatol 8(2): 200-205.
- Acosta S, Alhadad A, Svensson P, Ekberg O (2008) "Epidemiology, risk and prognostic factors in mesenteric venous thrombosis.” Br J Surg 95(10): 1245-1251.
- De Stefano V, Martinelli I (2010) "Splanchnic vein thrombosis: clinical presentation, risk factors and treatment.” Intern Emerg Med 5(6): 487494.
- Valla D (2004) "Hepatic venous outflow tract obstruction etiopathogenesis: Asia versus the West,” J Gastroenterol Hepatol 19(7): 204-211.
- De Stefano V, Za T, Ciminello A, Betti S, et al. (2011) "Causes of adult splanchnic vein thrombosis in the mediterranean area,” Mediterr J Hematol Infect Dis 3(1): e2011063.
- Mentha G, Giostra E, Majno P, Bechstein W, Neuhaus P, et al. (2006) "Liver transplantation for Budd-Chiari syndrome: a European study on 248 patients from 51 centres.” J Hepatol 44(3): 520-528.
- Darvish SM, Plessier A, Hernandez-Guerra M, Fabris F, et al. (2009) "Etiology, management, and outcome of the Budd-Chiari syndrome.” Ann Intern Med 151(3): 167-175.
- Denninger M, Chait Y, Casadevall N, Hillaire S, Guillin MC, et al. (2000) "Cause of portal or hepatic venous thrombosis in adults: the role of multiple concurrent factors.” Hepatology 31: 587-591.
- Janssen H, Meinardi J, Vleggaar F, van Uum SH, Haagsma EB, et al. (2000) "Factor V Leiden mutation, prothrombin gene mutation, and deficiencies in coagulation inhibitors associated with Budd-Chiari syndrome and portal vein thrombosis: results of a case-control study.” Blood vol. 96: 2364-2368.
- Sozer S, Fiel M, Schiano T, Xu M, Mascarenhas J, et al. (2009) "The presence of JAK2V617F mutation in the liver endothelial cells of patients with Budd-Chiari syndrome.,” Blood 113(21): 5246-5249.
- Rosti V, Villani L, Riboni R, Poletto V, Bonetti E, et al. (2013) "Spleen endothelial cells from patients with myelofibrosis harbor the JAK2V617F mutation.” Blood 121(2): 360-368.
- Kralovics R, Passamonti F, Buser A, Teo S, Tiedt R, et al. (2005) "A gain- of-function mutation of JAK2 in myeloproliferative disorders.” N Engl J Med 352(17): 1779-1790.
- Colaizzo D, Amitrano L, Tiscia G, Scenna G, Grandone E, et al. (2007) "The JAK2 V617F mutation frequently occurs in patients with portal and mesenteric venous thrombosis.” J Thromb Haemost 5(1): 55-61.
- Parker C, Omine M, Richards S, Nishimura J, Bessler M, et al. (2005) "Diagnosis and management of paroxysmal nocturnal hemoglobinuria,” Blood 106(12): 3699-3709.
- Garcia-Págan J, Heydtmann M, Raffa S, Plessier A, Murad S, et al. (2008) "TIPS for Budd-Chiari syndrome: long-term results and prognostics factors in 124 patients.” Gastroenterology 135(3): 808-815.
- Almdal T, Sorensen T (1991) "Incidence of parenchymal liver diseases in Denmark. 1981 to 1985: analysis of hospitalization registry data. The Danish Association for the study of the Liver.” Hepatology 13(4): 650-655.
- Condat B, Valla D (2006) "Nonmalignant portal vein thrombosis in adults.” Nat Clin Pract Gastroenterol Hepatol 3: 505-315.
- Sogaard K, Astrup L, Vilstrup H (2007) "Portal vein thrombosis; risk factors, clinical presentation and treatment.” BMC Gastroenterol 7: 34-39.
- Plessier A, Darwish MS, Hernandez MG, and et al. (2010) 'Acute portal vein thrombosis unrelated to cirrhosis: a prospective multicenter follow-up study.” Hepatology 51(1): 210-218.
- Fimognari F, Violi F (2008) "Portal vein thrombosis in liver cirrhosis.” Intern Emerg Med 3: 213-218.
- Stamou K, Toutouzas K, Kekis P, Nakos S, Gafou A, et al. (2006) "Prospective study of the incidence and risk factors of postsplenectomy thrombosis of the portal, mesenteric, and splenic veins.” Arch Surg 141: 663-669.
- Janssen H, Wijnhoud A, Haagsma E, van Uum SH, van Nieuwkerk CM, et al. (2001) "Extrahepatic portal vein thrombosis: aetiology and determinants of survival.” Gut 49(5): 720-724.
- Dental IF, Galli M, Gianni M, Ageno Wet al. (2008) "Inherited thrombophilic abnormalities and risk of portal vein thrombosis. A meta-analysis.” Thromb Haemost 99(4): 675-682. v
- Acosta S, Ogren M, Sternby N, Bergqvist D, Bjorck M (2005) "Mesenteric venous thrombosis with transmural intestinal infarction: a population- based study.” J Vasc Surg 41(1): 59-63.
- Dentali F, Ageno W, Witt D, Malato A, Clark N, et al. (2009) "Natural history of mesenteric venous thrombosis in patients treated with vitamin K antagonists: a multi-centre, retrospective cohort study.” Thromb Haemost 102: 501-504.
- Nasiri S, Khurgami J, Donboli K (2007) "Splenic vein thrombosis in a patient with chronic pancreatitis.” The Internet Journal of Surgery 16(2): 1-3.
- Moosa A, Gadd M (1985) "Isolated splenic vein thrombosis.” World J Surg 9: 348-390.
- Bernades P, Baits A, Levy P, et al. (1992) "Splenic portal venous obstruction in chronic pancreatitis.” Dig. Sci 37: 340-346.
- Janssen H, Garcia-Pagan J, Elias E, Mentha G, Hadengue A, et al. (2003) "Budd-Chiari syndrome: a review by an expert panel.” J Hepatol 38: 364-371.
- Senzolo M, Cholongitas E, Patch D, Burroughs A (2005) "Update on the classification, assessment of prognosis and therapy of Budd-Chiari syndrome.” Nat Clin Pract Gastroenterol Hepatol 2: 182-190.
- Hadengue A, Poliquin M, Vilgrain V, Belghiti J, Degott C, et al. (1994) "The changing scene of hepatic vein thrombosis: recognition of asymptomatic cases.” Gastroenterology 106: 1042-1047.
- Zeitoun G, Escolano S, Hadengue A, Azar A, El Younsi M, et al. (1999) "Outcome of Budd-Chiari syndrome: a multivariate analysis of factors related to survival including surgical portosystemic shunting.” Hepatology 30(1): 84-89.
- Darvish MM, Valla D, de Groen P, Zeitoun G, Haagsma EB, et al. (2006) "Pathogenesis and treatment of Budd-Chiari syndrome combined with portal vein thrombosis.” Am J Gastroenterol 101(1): 83-90.
- Martinelli I, De Stefano D (2010) "Rare thromboses of cerebral, splanchnic and upper-extremity veins.” Thromb Haemost 103(6): 1136-1144.
- Condat B, Pessione F, Hillaire S, Denninger MH, Guillin MC, et al. (2001) "Current outcome of portal vein thrombosis in adults: risk and benefit of anticoagulant therapy.” Gastroenterology 120(2): 490-497.
- Kumar S, Sarr M, Kamath P (2001) "Mesenteric vein thrombosis.” N Engl J Med 345(23): 1683-1688.
- Kamath P (2006) "Budd-Chiari syndrome: radiologic findings.” Liver Transp 12(11 Suppl 2): S21-S22.
- Mathieu D, Vasile N, Grenier P (1985) "Portal thrombosis: dynamic CT features and course.” Radiology 154: 737-41.
- Parvey R, Raval B, Sandier C (1994) "Portal Vein Thrombosis: Imaging Findings.” AJR 162: 77-81.
- Darvish SM, Valla D, de Groen P, Zeitoun G, Hopmans JA, et al. (2004) "Determinants of survival and the effect of portosystemic shunting in patients with Budd-Chiari syndrome.” Hepatology 39: 500-508.
- Rajani R, Bjornsonn E, Bergquist A, Danielsson A, Gustavsson A, et al. (2010) "The epidemiology and clinical features of portal vein thrombosis: a multicentre study” Aliment Pharmacol Ther 95(10): 1154-1162.
- Spaander M, Hoekstra J, Hansen J, et al. (2013) "Anticoagulant therapy inpatients with non-cirrhotic portal vein thrombosis: effect on new thrombotic events and gastrointestinal bleeding.” J Thromb Haemost 11(3): 452-459.
- Hedayati N, Riha G, Kougias P, Huynh TT, Cheng C, et al. (2008) "Prognostic factors and treatment outcome in mesenteric vein thrombosis.” Vasc Endovascular Surg 42(3): 217-224.
- Ageno W, Riva N, Schulman S, Bang SM, Sartori MT, et al. (2014) "IRSVT study group. Antithrombotic treatment of splanchnic vein thrombosis: results of an international registry.” Semin Thromb Hemost 40(1): 99105.
- Senzolo M, Riggio O, Primignani M (2011) "Vascular disorders of the liver: recommendations from the Italian Association for the Study of the Liver (AISF) ad hoc committee.” Dig Liver Dis 43: 503-514.
- de Franchis R (2010) "Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension.” J Hepatol 53(4): 762768.
- Kearon C, Akl E, Comerota A, Prandoni P, Bounameaux H, et al. (2012) "Antithrombotic therapy for VTE disease: Antithrombotic Therapy and Prevention of Thrombosis, (9th edn). American College of Chest Physicians Evidence-Based Clinical Practice Guidelines, Chest 141: e419S-94S.
- Ageno W, Dentali F, Squizzato A (2014) "How I treat splanchnic vein thrombosis.” Blood 124(25): 3685-3691.
- Sharma S, Texeira A, Texeira P, Elias E, Wilde J, et al. (2004) "Pharmacological thrombolysis in Budd Chiari syndrome: a single centre experience and review of the literature.” J Hepatol 40: 172-180.
- Hollingshead M, Burke C, Mauro M, Weeks SM, Dixon RG, et al. (2005) "Transcatheter thrombolytic therapy for acute mesenteric and portal vein thrombosis.” J Vasc Interv Radiol 16(5): 651-661.
- Liu F, Wang M, Fan Q, et al. (2009) "Interventional treatment for symptomatic acute subacute portal and superior mesenteric vein thrombosis.” World J Gastroenterol 15(40): 5028-5034.
- Senzolo M, Patch D, Miotto D, Ferronato C, Cholongitas E, et al. (2008) "Interventional treatment should be incorporated in the algorithm for the management of patients with portal vein thrombosis.” Hepatology 48(4): 1352-1353.
- Ageno W, Squizzato A, Togna A, Magistrali F, Mangini M, et al. (2012) "Incidental diagnosis of a deep vein thrombosis in consecutive patients undergoing a computed tomography scan of the abdomen: a retrospective cohort study” J Thromb Haemost 10(1): 158-160.






























