Successful Treatment of Disseminated Fungal Infections in three Pediatric Patients with Acute Lymphoblastic Leukemia
Funda Tekkeşin1*, Zühre Kaya1, EmineBetul Derinkuyu2, Alp Özgün Börcek3, BurcuCeylanCura Yayla4, Hasan Tezer4, Ayşe Kalkanci5, Ülker KoÇak1, idil Yenicesu1 and Alper Ceylan6
1Department of Pediatric Hematology, Gazi University, Turkey
2Department of Pediatric Radiology, Gazi University, Turkey
3Department of Neurosurgery, Gazi University, Turkey
4Department of Pediatric Infectious Diseases, Gazi University, Turkey
5Department of Microbiology, Gazi University, Turkey
6Department of Otorhinolaryngology, Gazi University, Turkey
Submission: January 19, 2018 Published: February 13, 2018
*Corresponding author: Funda Tekkeşin, Department of Pediatrics, Unit of Pediatric Hematology, Gazi University School of Medicine, Beşevler, Ankara, Turkey 06500, Tel: + 90 533 269 44 70; Email: tekkesinfunda@gmail.com
How to cite this article: Funda T, Zuhre K, EmineBetul D, Alp O B, BurcuCeylanCura Y. Successful Treatment of Disseminated Fungal Infections in three Pediatric Patients with Acute Lymphoblastic Leukemia. Blood Res Transfus 004 ransfus J. 2018; 2(1) : 555576. DOI: 10.19080/OABTJ.2018.02.555576
Abstract
We present the cases of three children with acute lymphoblastic leukemia (ALL) who developed disseminated invasive fungal infections (IFIs) (Aspergillusspp. n=1, Candida spp. n=2). All were successfully treated with antifungal drugs and ancillary treatments; however, in two cases, leukemic cells were not eradicated by chemotherapy before transplantation. It has been noted that, despite the success of novel antifungal drugs in patients with ALL, long-lasting IFIs during chemotherapy may negatively affect leukemia prognosis. We emphasize the need for guidelines to provide assistance in preventing, evaluating and treating IFIs in this patient group.
Keywords: Disseminated fungal infections; Acute leukemia; Children
Abbreviations : IFI: Invasive Fungal Infection; ALL: Acute Lymphoblastic Leukemia; HSCT: Hematopoietic Stem Cell Transplantation; CNS: Central Nervous System; BSAs: Broad-Spectrum Antibiotics; MRI: Magnetic Resonance Imaging; HRCT: High-Resolution Computerized Tomography; LAMB: Liposomal Amphotericin B; VRC: Voriconazole; G-CSF: Granulocyte-Colony Stimulating Factor; IRIS: Immune Reconstitution Inflammatory Syndrome
Introduction
Invasive fungal infection (IFI) is a major cause of morbidity and mortality in children with leukemia. Aspergillus spp. and Candida spp. are the main pathogens in most of these cases [1]. These infections pose major threats for immunocompromised patients, especially those with acute myeloid leukemia and highrisk acute lymphoblastic leukemia (ALL) who are undergoing chemotherapy or hematopoietic stem cell transplantation (HSCT) [2].
Fungal infections cause various clinical presentations in children with leukemia, ranging from asymptomatic to life-threatening disseminated disease. The most frequently reported locations of IFI are the liver and lung, whereas the central nervous system (CNS), kidney, epiglottis and muscle are less common sites [3-10]. Early detection and treatment of disseminated IFIs and underlying hematological problems are critically important for patient survival. Here we describe the clinical courses and successful treatment of three children with ALL who had disseminated IFIs.
Case 1: A 14-year-old boy with t(9;22)-positive high-risk ALL and asthma presented initially with persistent cough. At the end of the first induction treatment of the ALL-BFM 95 protocol under fluconazole prophylaxis, he developed febrile neutropenia, headache and visual hallucinations. The patient was treated with broad-spectrum antibiotics (BSAs) and neutrophil recovery was observed; however, his neurological symptoms worsened. Cranial magnetic resonance imaging (MRI) revealed two nodular lesions 3cm and 5cm diameter in his frontal and temporal lobes, respectively. These were considered to be consistent with possible lymphocytic sarcoma. The larger cranial lesion was removed by neurosurgeons and tested positive for Aspergillus spp. on real-time PCR (TibMolbiol, Berlin, Germany) , galactomannan antigen assay, and tissue culture, thus confirming IFI according to the definition criteria set by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group [11]. High-resolution computerized tomography (HRCT) and abdominal ultrasonography also showed multiple nodular lesions in the lungs (Figure 1) and liver. As well, peripheral blood and central catheter fungal cultures were positive for Aspergillus spp.
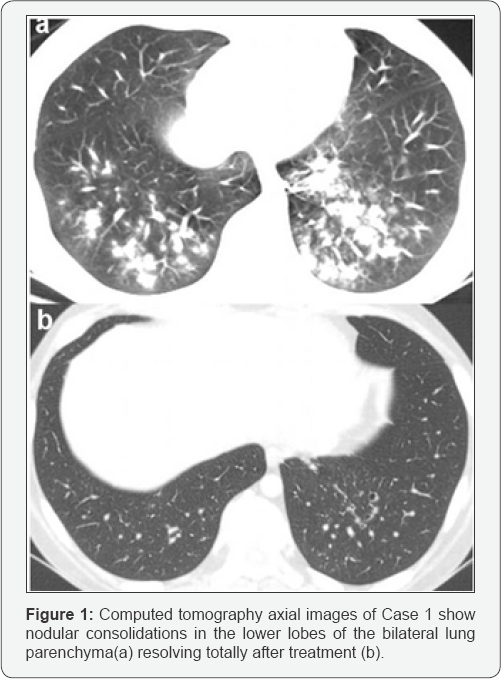
The patient was treated with a combination of liposomal amphotericin B (LAMB) and voriconazole (VRC) for 2 months. After intensive chemotherapy with the ALL-BFM 95 protocol, he entered complete remission. However, when the second originally detected lesion (presumed to be fungal) in his brain increased in size during chemotherapy, this treatment was discontinued and the lesion was removed by surgery. After 3 months of VRC treatment alone, the patient's pulmonary and liver lesions showed prominent regression and he underwent successful allogeneic (allo)-HSCT from his sibling while still receiving VRC. He achieved complete remission for 2 years.
Case 2: An 11-year-old boy with ALL relapse was scheduled to undergo allo-HSCT. While he was on the FLAG-IDA protocol and 5 mg/kg fluconazole prophylaxis, he developed prolonged neutropenic fever, altered mental status, and widespread maculopapular eruptions across his face and body. He was treated with BSAs while also receiving empirical LAMB 3mg/ kg, but his respiratory pattern deteriorated. Candida albicans was cultured from all the skin lesions, and from his peripheral blood, central catheter and cerebrospinal fluid, thus confirming IFI. As well, real-time PCR (TibMolbiol, Berlin, Germany) was positive for Candida spp. The patient's central catheter could not be removed because of vascular access problems. Early tracheostomy was performed to address sudden inspiratory stridor after laryngoscopic examination suggested fungal infection of the epiglottitis. Granulocyte transfusion and granulocyte-colony stimulating factor (G-CSF) treatment were administered for 3 weeks until neutrophil recovery was observed. Caspofungin was added as combination treatment with LAMB in the first 3 months. After this period, HRCT showed nodular lesions consistent with pulmonary aspergillosis, and VRC and LAMB combination was used for salvage over the next 3 months. The patient was discharged from hospital without IFI and free of leukemia. He remained well during next 2 years, but then developed isolated bone marrow relapse and died of persistent leukemia.
Case 3: An 8-year-old girl presented to the outpatient clinic with fever and cough, and was treated with an oral antibiotic. In the subsequent 2 weeks, she was diagnosed with mediumrisk precursor B ALL. Chemotherapy was initiated according to the ALL BFM-95 protocol. The patient developed mucositis and severe neutropenic fever at the end of the induction treatment, and BSAs were started on Day 1. However, her fever did not respond to this therapy, and empirical antifungal treatment with caspofungin was added seven days later. Peripheral blood and central catheter fungal cultures grew Candida tropicalis. The patient developed hematuria and difficulty walking on Day 10. Radiological imaging showed nodular lesions in the spleen and renal abscess, all suggesting fungal infection. Pelvic MRI revealed multifocal lesions in the gluteal muscles bilaterally and in the left proximal muscles, suggesting abscesses consistent with Candida pyomyositis.Caspofungin combined with LAMB was continued for at least 5 days, but LAMB was switched to VRC on Day 15 because of persistent fever. Although the patient’s neutrophil count gradually increased over the next 3 days, her condition suddenly deteriorated with dyspnea, tachypnea and hypoxia as well as increased C-reactive protein level, which together suggested immune reconstitution inflammatory syndrome (IRIS). The patient was treated with steroid 2mg/kg for 2 weeks, and clinical and laboratory assessments revealed dramatic response. The combined antifungal treatment was continued for 28 days, and she received VRC treatment during chemotherapy. The patient entered complete remission on day 33. At the time of writing, she remained well and had had no signs of fungal disease recurrence on radiological imaging during 10 months of follow-up.
Discussion
Limited data are available about the frequency, clinical features, and optimal treatment schedule for disseminated IFIs and their prognosis in children with ALL [1,2,12]. A previous comprehensive study of 2,021 children who were treated with the ALL-BFM protocol suggested that, in nine cases, prolonged neutropenia, long-lasting steroid use, and presence of organ dysfunction were important risk factors for developing IFI [1]. Aspergillus spp. was the most common cause of IFI in that study, and accounted for 21% of infection-related deaths [1]. Another investigation from Turkey revealed 10.2% incidence of IFI in pediatric ALL patients receiving fluconazole prophylaxis [13]. Steroid medications was found to be significantly associated with development of IFI in 81.8% of patients with ALL and, Candida spp. were the most common cause of IFI in that study [13]. Our results are consistent with these data. Comprehensive studies have recommended primary antifungal prophylaxis for patients with high-risk ALL, whereas smaller series or case reports have noted that assessment of IFIs in patients with non-high risk ALL is greatly restricted by the secondary prophylactic and empirical use of several antifungal drugs [1,2,14].
Candida spp. frequently colonize the liver and spleen, whereas Aspergillus spp. tend to localize in the sinuses and lungs [13]. Invasive fungal infections may also occur in other organs, including the kidneys, CNS, epiglottis, skin and muscle [3-10]. Simultaneous occurrence in these organs, as observed in our cases, has been previously reported in smaller series and case reports [3-12,15]. Cerebral involvement usually occurs secondary to hematogenous dissemination of an IFI throughout the systemic circulation, and is associated with high mortality [3-5]. Treatment of IFI in the CNS remains challenging due to low penetration by novel antifungal drugs that do not readily cross the blood-brain barrier. Although it is well established that VRC passes into the CSF, appropriate levels can only be reached by monitoring drug concentration levels [16-18]. Since it is not technically possible to measure blood levels of VRC at our clinic, we used VRC in combination with LAMB to address CNS involvement. To date, no standard antifungal drug combinations have been reported for treating IFI. Based on the limited number of reported cases, we suggest that antifungal drug combinations may be used as salvage treatment for patients with disseminated IFIs. Alternatively, as was the situation in our Patient 1, surgery might be considered as a life-saving option for select patients with well-defined fungal lesions, and can be supported further by G-CSF or granulocyte infusion for neutropenia [3-5].
A literature search for Candida infection of the epiglottitis and disseminated skin lesions in children with ALL revealed only one reported pediatric patient with simultaneous locations of IFI and ALL, as we observed in our Patient 2 [6]. Concomitant Varicella and Candida infections were important causes of acute inspiratory stridor and skin eruptions in that case. The clinical picture was also similar to that observed in our second case; however, fungal cultures of our patient's skin biopsy and blood and CNS revealed C.albicans and he had multiple papular lesions scattered over his head, neck and trunk, which is a rare presentation of Candida. It is important to emphasize that diagnosis of a patient with IFI in such locations may be delayed because many physicians are not familiar with this clinical picture. Despite the broad spectrum of new antifungal drugs available, there have been challenges reaching adequate concentrations in various tissues, including the CNS, skin and epiglottis [16-18]. We successfully treated disseminated IFI in children with ALL using a combination of LAMB and caspofungin or VRC.
Concomitant kidney and muscle IFI associated with ALL has been described in only one child with ALL to date [8]. Patients with fungal myositis have been diagnosed either on postmortem examination or during induction for adult leukemia [8-10]. In parallel with these findings, kidney and muscle involvement of C. tropicalis were detected at the end of induction treatment for leukemia in our Patient 3. In this case, we administered antifungal drug combinations for 4 weeks, after which clinical and laboratory assessments revealed a dramatic response. Induction chemotherapy with VRC treatment was continued without complications. However, her condition then suddenly deteriorated, with dyspnea, tachypnea and hypoxia, increased neutrophil count and elevated C-reactive protein level, all suggesting IRIS syndrome during disseminated candidemia. Life-threatening pulmonary symptoms are uncommon in Candida infection but they may be observed during periods of IFI, presumably as a part of a hyper inflammatory syndrome. [19,20]. The literature contains a few cases of IRIS syndrome secondary to Candida infection caused by exaggerated inflammatory response during resolution of neutropenia [19,20]. The appropriate diagnosis and identification of IFI in patients with acute leukemia are of great clinical importance because IRIS syndrome is reversible with steroid administration, as we observed with our Patient 3.
Our experience reinforce the finding in previous reports that long term medical treatment of disseminated IFI with combined antifungal treatment can be lifesaving in cases with leukemia.
References
- Grigull L, Beier R, Schrauder A, Kirschner P, Loening L, et al. (2003) Invasive fungal infections are responsible for one-fifth of the infectious deaths in children with ALL. Mycoses 46(11-12): 441-446.
- Groll AH, Castagnola E, Cesaro S, Dalle JH, Engelhard D, et al. (2014) Fourth European Conference on infections in leukemia (ECIL- 4):guidelines for diagnosis, prevention, and treatment of invasive fungal diseases in paediatric patients with cancer or allogeneic haematopoetic stem cell transplantation. Lancet Oncol 15(8): 327-340.
- Carter J, Lenahan J, Ishak GE, Geyer JR, Pollard J, et al. (2015) Medical management of invasive fungal infections of the central nervous system in pediatric cancer patients. Pediatr Blood Cancer 62(6): 1095-1098.
- Dotis J, Iosifidis E, Roilides E (2007) Central nervous system aspergillosis in children: a systematic review of reported cases. Int J Infect Dis 11(5): 381-393.
- Middelhof CA, Loudon WG, Muhonen MD, Xavier C, Greene CS (2005) Improved survival in central nervous system aspergillosis:a series of immunocompromised children with leukemia undergoing stereotactic resection of aspergillomas. Report of four cases. J Neurosurg 103(4 Suppl): 374-388.
- Chiou CC, Seibel NL, Derito FA, Bulas D, Walsh TJ, et al. (2006) Concomitant Candida epiglottitis and disseminated Varicella zoster virus infection associated with acute lymphoblastic leukemia. J PediatrHematol Oncol 28: 757-759.
- Schwartz DM, Morgan ER (2008) Multimodality imaging of Candida tropicalis myositis. Pediatr Radiol 38(4): 473-476.
- Kressel B, Szewczyk C, Tuazon CU (1978) Early clinical recognition of disseminated candidiasis by muscle and skin biopsy. Arch Intern Med 138(3): 429-433.
- Fornadley JA, Parker GS, Rickman LS, Paparello S (1990) Candida myositis manifesting as a discrete neck mass. Otolaryngol Head Neck Surg 102(1): 74-76.
- Lin XJ, Yao RX, He MQ, Zhu BL, Guo WJ (2013) Treatment of fungal myositis with intra-lesional and intravenous itroconazole: a case report. J Med Case Rep 7: 132.
- Ascioglu S, Rex JH, de Pauw B, Bennett JE, Bille J, et al. (2002) Invasive Fungal Infections Cooperative Group of the European Organization for Research and Treatment of Cancer; Mycoses Study Group of the National Institute of Allergy and Infectious Diseases. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: An international consensus. Clin Infect Dis 34(1): 7-14.
- Morgan JE, Hassan H, Cockle JV, Lethaby C, James B, et al. (2017) Critical review of current clinical practice guidelines for antifungal therapy in paediatrichaematology and oncology. Support Care Cancer 25(1): 221-228.
- Kaya Z, Gursel T, Kocak U, Aral YZ, Kalkanci A, et al. (2009) Invasive fungal infections in pediatric leukemia patients receiving fluconazole prophylaxis. Pediatr Blood Cancer 52(4): 470-475.
- Rüping MJ, Vehreschild JJ, Cornely OA (2008) Antifungal treatment strategies in high risk patients. Mycoses 51(Suppl 2): 46-51.
- Zaoutis TE, Greves HM, Lautenbach E, Bilker WB, Coffin SE (2004) Risk factors for disseminated candidiasis in children with candidemia. Pediatr Infect Dis J 23(7): 635-641.
- Grigull L, Beilken A, Schmid H, Kirschner P, Sykora KW, et al. (2006) Secondary prophylaxis of invasive fungal infections with combination antifungal therapy and G-CSF mobilized granulocyte transfusions in three children with hematological malignancies. Support Care Cancer 14(7): 783-786.
- Barrios NJ, Echevarria ME, Velez R (2005) Succesful treatment of disseminated aspergillosis in a leukemic child. PR Health Sci J 24(2): 157-160.
- Schwartz S, Reisman A, Troke PF (2011) The efficacy of voriconazole in the treatment of 192 fungal central nervous system infections: a retrospective analysis. Infection 39: 201-210.
- Chaussade H, Bastides F, Lissandre S, Blouin P, Bailly E, et al. (2012) Usefulness of corticosteroid therapy during chronic disseminated candidiasis:Case report and literature review. J Antimicrob Chemother 67(6): 1493-1495.
- Zajac-Spyhala O, Ukielska B, Jonczyk-Potoczna K, et al. (2016) Chronic disseminated candidiasis complicated by immune reconstitution inflammatory syndrome in child with acute lymphoblastic leukemia. Case Rep Hematol.






























