Correlation of Acute Chest Syndrome with e-NOS, ARG1 and GSNOR Gene Polymorphisms in Omani Sickle Cell Patients
Aiman Al Wahaibi1, Anil Pathare2, Shoaib Al Zadjali3 and Salam Alkindi2,3*
1Department of Medicine, Oman Medical Specialty Board, Oman
2Department of Haematology, Sultan Qaboos University Hospital, Oman
3Hege of Medicine and Health Sciences, Oman
Submission: January 08, 2018 Published: February 02, 2018
*Corresponding author: Salam Alkindi, College of Medicine & Health Sciences, Consultant Haematologist, Department of Haematology, Sultan Qaboos University Hospital, Muscat Oman, Tel: +(968) 24144915; Fax: +(968) 24144887; Email: sskindi@jrahoo.com
How to cite this article: Aiman Al Wahaibi, Anil Pathare, Shoaib Al Zadjali, Salam Alkindi. Correlation of Acute Chest Syndrome with e-NOS, ARG1 and GSNOR Gene Polymorphisms in Omani Sickle Cell Patients. Blood Res Transfus J. 2018; 1(5): 555573. DOI: 10.19080/OABTJ.2018.01.555573
Abstract
Introduction: Acute chest syndrome (ACS), is the most common pulmonary complication of sickle cell disease (SCD) and reduced nitric oxide (NO) is considered to play a significant role. NO induces vasodilatation and helps in the recruitment of neutrophils. However, the endothelial nitric oxide synthase (NOS3) gene polymorphisms T-786C significantly reduces eNOS gene promoter activity, whereas the E298D changes an amino acid in the enzyme’s oxygenase domain. The expression of eNOS is also related to the number of 27bp repeat VNTRs in intron 4, with genotype 4bb showing a decrease in eNOS expression whereas, homozygosity for the minor allele of GSNOR SNP rs28730619 is associated with increased risk of asthma.
Objectives: Endothelial nitric oxide synthase (NOS3) gene polymorphisms (T-786C, E298D and Intron 4 VNTR) as well as ARG1 and GSNOR gene polymorphisms were evaluated in homozygous SCD patients from Oman with the occurrence of ACS and painful vaso-occlusive crises.
Method: Genomic DNA was isolated using the standard techniques and stored at -200C pending analysis. DNA was studied for polymorphisms of HBB gene (β6Glu>Val), NOS3 gene polymorphisms (T-786C, E298D and Intron 4 VNTR) as well as for ARG1 and GSNOR gene polymorphisms by direct sequencing of the relevant genomic segment amplified by polymerase chain reaction on an ABI PRISM 3100 genetic analyzer using appropriate primers described in literature.
Observation: We observed that only the eNOS promoter C-786 allele showed a statistically significant association (P=0.016, Chi-Square test) in ACS cases, especially so with the female gender. There was no correlation observed with eNOS polymorphisms E298D and Intron 4VNTR, ARG1 and GSNOR gene studied in this cohort of SCD patients with ACS.
Conclusion: This study reveals that eNOS promoter C-786 variant, which reduces eNOS gene activity was observed as a genetic risk factor for ACS in adult female SCD patients, and may be explained by the fact that eNOS is known to be regulated by estrogens.
Keywords: Acute chest syndrome; eNOS; GSNOR; ARG1; SCD
Abbreviations: ACS: Acute Chest Syndrome; SCD: Sickle Cell Disease; NO: Nitric Oxide; NOS3: Nitric Oxide Synthase; VOC: Vaso-Occlusive Crises; eNOS: Endothelial NOS; nNOS: Neuronal NOS; iNOS: Inducible NOS; SQUH: Sultan Qaboos University Hospital
Introduction
Sickle cell disease (SCD) is one of the most common inherited diseases in the world. Acute chest syndrome (ACS) and painful vaso-occlusive crises (VOC) are the most common causes of hospitalization in sickle cell anemia. ACS occurs in 15-43% of patients with SCD [1-2]. It is the second most important causes of premature death (25%) [3].
Nitric oxide (NO), is produced by three isoforms of NO synthase. Endothelial NOS (eNOS) is mostly expressed in endothelial cell, whereas, neuronal NOS (nNOS) is constitutively expressed in central and peripheral neurons, while inducible NOS (iNOS) can be expressed in many cell types in response to lipopolysaccharide, cytokines, or other agents. Several studies have shown that depletion of endothelial NO is involved in the pathogenesis of VOC and ACS in SCD patients [4-6]. Hence, the identification of genetic modifiers of ACS would help in designing treatment strategies.
There have been several reports on e-NOS promoter mutations. It has been shown that the T-786C polymorphism significantly reduces eNOS gene promoter activity, whereas, the E298D changes an amino acid in the enzyme’s oxygenase domain [7-8]. The expression of eNOS is also related to a variable number of tandem (27-base pair) repeats (VNTR) in intron 4, with genotype 4bb repeats, showing a decrease in eNOS expression [9] whereas, homozygosity for the minor allele of GSNOR SNP rs28730619 is associated with increased risk of asthma [10]. Additionally, NO levels are also affected by ARG1 and GSNOR gene polymorphisms. The aim of this study was to examine these polymorphisms (T-786C, E298D and Intron 4 VNTR) as well ARG1 and GSNOR gene polymorphisms in Omani SCD patients as potential modifiers for ACS acting by reducing NO.
Materials and Methods
A total of 88 Omani SCD patients were enrolled from Department of Haematology at Sultan Qaboos University Hospital (SQUH). Patients were enrolled after an informed consent during their regular SCD clinic visits. Inclusion criteria were: SS genotype and Omani ethnicity. Clinical data during ACS episodes were obtained by retrospective chart review. The study was conducted following approval by the hospital medical research and ethics committee. The diagnosis of ACS was defined by following criteria: presence of new pulmonary infiltrate(s) on chest X-ray, history of cough and dyspnea and presence of acute chest pain and hypoxemia. Exclusion criteria were respiratory distress syndrome or blood transfusion within the past 30 days. Genomic DNA was isolated from peripheral blood using the standard techniques and stored at -20 °C pending analysis.
DNA sequence polymorphisms for HBB gene(β6Glu>Val), NOS3 gene polymorphisms (T-786C, E298D and Intron 4 VNTR) as well as ARG1 and GSNOR gene polymorphisms were studied by direct sequencing of the relevant genomic segment amplified by polymerase chain reaction on an ABI PRISM 3100 genetic analyzer using appropriate primers described in literature.
Statistical methods
Allele and genotype frequencies were compared by standard contingency table analysis using two-tail Fisher's exact test probabilities. Multiple logistic regression analysis utilizing Stata software ver. 11.1 (Statacorp, College Station, TX, USA). Statistical significance was defined as a p-value <0.05.
Results
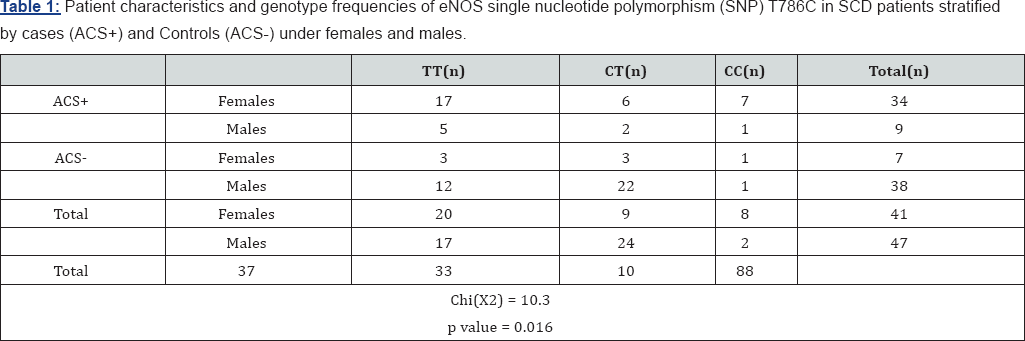
The mean (±SD) age of our patient cohort (n=88) was 29.8 ± 9.4 years (range: 9-55). Forty-seven (53.4%) of them were males. This cohort comprised 45 SCD patients with ACS complication along with 43 SCD patients who did not have ACS (controls). The mean (±SD) age in the SCD ACS cohort was 29.1 ± 7.6 years (range: 12-46), whereas in the SCD patients without ACS, it was 30.5+10.7 years (range: 9-55). Amongst the 5 different studied polymorphisms: only genotype distributions for eNOS3 T-786C alleles were significantly different between SCD patients with (4.7%) or without (21%) ACS (p=0.001, Chi-Square test). Furthermore, although our results suggest that the eNOS3 C-786 allele decreases the risk of ACS in Omani SCD patients, it was enriched in female patients with ACS (21.7%) with significance p value of 0.016 (Table 1). Unfortunately, we did not find any association with E298D and Intron 4VNTR, ARG1 and GSNOR gene polymorphisms that were studied in this cohort of SCD patients with ACS (p>0.05, Chi square test).
Discussion
ACS is a lung injury syndrome precipitated by infection, embolization of bone marrow fat, and intravascular sequestration of red cells. All of these triggers are related to VOC [11]. Although several mechanistic pathways are involved, NO deficiency is postulated to play a significant part in the pathogenesis because it has a major role in lung physiology and airway disease [12]. During ACS, bioavailability of NO is reduced and is regulated by NOS which helps in the synthesis of NO from L-arginine.
It is important to understand the significance of poor representation of the eNOS3 C-786 allele in homozygous state in SCD patients with ACS. It is thus postulated that these results imply involvement of different mechanistic pathway, the consequences of which vary accordingly to the events triggering ACS [13]. eNOS promoter C-786 variant which reduces eNOS gene activity was observed as a genetic risk factor for ACS in adult female sickle cell anemia patients, and that could be explained with the fact that eNOS is known to be regulated by estrogens.It is understood that because T-786C site is in close proximity with a predicted estrogen responsive element, a DNA binding protein, RPA1, has been shown to reduce C-786 eNOS promoter activity [5,14]. That could explain why male patients did not show any association with this eNOS polymorphism. However, in addition to estrogen, eNOS is also regulated or altered by drugs, cigarette smoking, and by many other disease states, including hypercholesterolemia, diabetes and hypertension.
Furthermore, we did not find any correlation with the other eNOS polymorphisms namely, E298D and Intron 4VNTR, ARG1 and GSNOR gene polymorphisms that were studied in this cohort of SCD patients with ACS. Thus, it is proposed that a larger study of these SNPs must be conducted to examine its interactions with known ACS risk factors. Furthermore, studies are also warranted to measure NO bioavailability in known T-786C SCD patients.
Conclusion
The study documents that eNOS promoter C-786 variant is indeed a genetic predisposing risk factor for ACS in female SCD patients, and substantiates a similar observation that has been reported earlier.
Acknowledgement
We sincerely thank the Hospital Director and the administration for allowing the use of clinical and laboratory material for the purpose of this study.
References
- Graido-Gonzalez E, Doherty JC, Bergreen EW, Organ G, et al. (1998) Plasma endothelin-1, cytokine, and prostaglandin E2 levels in sickle cell disease and acute vasoocclusive sickle crisis. Blood 92(7): 25512555.
- Hammerman SI, Kourembanas S, Conca TJ, Tucci M, Brauer M, et al. (1997) Endothelin-1 production during the acute chest syndrome in sickle cell disease. Am J Respir Crit Care Med 156(1): 280-255.
- Bunn HF (1997) Pathogenesis and treatment of sickle cell disease. N Engl J Med 337(11): 762-769.
- Vichinsky EP, Styles LA, Colangelo LH, Wright EC, Castro O, et al. (1997) Acute chest syndrome in sickle cell disease: clinical presentation and course. Cooperative Study of Sickle Cell Disease. Blood 89(5): 17871792.
- Sharan K, Surrey S, Ballas S, Borowski M, Devoto M, et al. (2004) Association of T-786C eNOS gene polymorphism with increased susceptibility to acute chest syndrome in females with sickle cell disease. Br J Haematol 124(2): 240-243.
- Yousry SM, Ellithy HN, Shahin GH (2016) Endothelial nitric oxide synthase gene polymorphisms and the risk of vasculopathy in sickle cell disease. Hematology 21(6): 359-367.
- Nakayama M, Yasue H, Yoshimura M, Shimasaki Y, Kugiyama K, et al.(1999) T-786- C mutation in the 5'-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation, 99(22): 2864-2870.
- Yoshimura M, Yasue H, Nakayama M, Shimasaki Y, Sumida H, Sugiyama S, et al. (1998) A missense Glu298Asp variant in the endothelial nitric oxide synthase gene is associated with coronary spasm in the Japanese. Human Genetics 103(10): 65-69.
- Tsukada T, Yokoyama K, Arai T, Takemoto F, Hara S, et al. (1998) Evidence of association of the eNOS gene polymorphism with plasma NO metabolite levels in humans. Biochem Biophys Res Commun 245(1): 190-193.
- Boyd JH, Moinuddin A, Strunk RC, Debaun MR (2004) Asthma and acute chest in sickle-cell disease. Pediatr Pulmonol 38(3): 229-232.
- Mark T, Vichinsky E (2008) Pulmonary Complications of Sickle Cell Disease. N Engl J Med 359(21): 2254-2265.
- Gladwin MT, Schechter AN, Shelhamer JH, Ognibene FP (1999) The Acute Chest Syndrome in Sickle Cell Disease Possible Role of Nitric Oxide in Its Pathophysiology and Treatment. Am J Respir Crit Care Med 159(5 pT 1): 1368-1376.
- Chaar V, Tarer V, Etienne-Julan M, Diara JP, Elion J, et al. (2006) ET-1 and ecNOS gene polymorphisms and susceptibility to acute chest syndrome and painful vaso-occlusive crises in children with sickle cell anemia. Haematologica 91(9): 1277-1278.
- Miyamoto Y, Saito Y, Nakayama M, Shimasaki Y, Yoshimura T, et al.(2000)Replication protein A1 reduces transcription of the endothelial nitric oxide synthase gene containing a -786T-->C mutation associated with coronary spastic angina. Hum Mol Genet 9(18): 2629-2637.






























