Polycythemia Rubra vera-Clinical Profile & Predictors of Thrombotic Complications
Thomas Augustine1, Sasidharan PK1,2*and Muhammed Shaan1,2
1Department of Medicine, Government Medical College, Kozhikode, India
2Department of Hematology, Government Medical College, Kozhikode, India
Submission: March 23, 2017; Published: April 17, 2017
*Corresponding author: Sasidharan PK, Department of Medicine, Government Medical College, India, Email: sasidharanpk@gmail.com
How to cite this article: Thomas A, Sasidharan P, Muhammed S. Polycythemia Rubra Vera-Clinical Profile & Predictors of Thrombotic Complications . Open Acc Blood Res Trans J. 2017; 1(2) : 555556.
Abstract
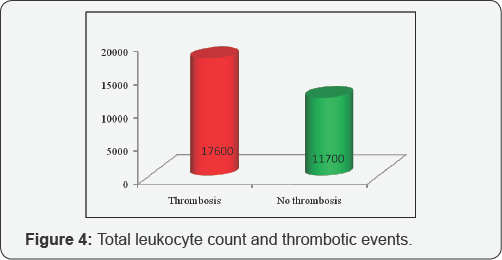
Polycythemia rubra vera (PRV) is a myeloproliferative disorder, with significant risk for thrombotic complications [1]. The study was an attempt to highlight the clinical and hematological profile of the disease in Indian population sampled from a tertiary care centre of Northern Kerala and to probe into the possible predictors of thrombotic events. Of the 37 cases, males constituted 73% and their mean age at diagnosis was 57 years. A few findings contradicted the clinical utility of 2008 WHO criteria for the diagnosis of PRV. 18.9% of the subjects had hemoglobin level less than the WHO cut off and splenomegaly was absent in another 18.9% of the cases, who were all JAK-2 positive and had symptoms of polycythemia. Thrombotic complications occurred in 35.1% of the subjects. Predictors of thrombosis included higher hemoglobin levels (19.4g% vs 18.6g%; P:0.026), high total leukocyte counts (17600/mm3 vs 11700/mm3; P:0.001) and elevated uric acid levels (8.31mg/dl vs 6.78 mg/dl; P:0.001). Subjects with more consumption of fruits, vegetables and pulses had a significantly lower risk of thrombotic events (P:0.01).
Keywords: Polycythemia rubra vera; Myeloproliferative disorder; Thrombotic events
Introduction
Polycythemia Rubra Vera (PRV) is one of the chronic myeloproliferative disorders (neoplasms), characterized by clonal proliferation of hematopoietic stem cells [2] with variable morphologic maturity and hematopoietic efficiency. The disease has a course with inter current thrombotic complications [1,3] and may end up in myelofibrosis or transformation to a haematological malignancy [4]. Major aim of the treatment is to prevent thrombotic complications and to retard the progression of the disease [5]. Identification of risk factors for thrombotic complications and prompt management can significantly reduce the morbidity and mortality associated with this disease.
The disease is diagnosed by demonstrating elevated red cell mass, after ruling out other secondary causes for erythrocytosis, with other supportive evidences which varies with different diagnostic criteria, which were modified based on new insight into the pathogenesis of this disease (for eg: Polycythemia Vera Study Group Criteria [6] and 2008 WHO criteria). But the latest diagnostic criteria like the 2008 WHO criteria includes tests such as endogenous erythroid colony (EEC) formation that are available only in few research laboratories and still not validated and hence most often we will have to rely on older clinical criteria for the diagnosis of this disease. Even though discovery of the JAK2 V617F mutation [6,7] in the pathogenesis of this disease has been elucidated recently, the fact that it is not the sole factor responsible for PRV marks its limitation.
Being a relatively rare disease, epidemiological studies highlighting the clinical and haematological profile of this disease in Indian scenario are very few. A variation in the pattern of disease in Indian population when compared to Western population can be expected, based on significant difference in normal haematological parameters between these groups due to the effect of ethnic, dietary, environmental and cultural factors. The present study was to get a complete clinical profile of the patients with polycythemia rubra vera in this part of the country and to look into possible predictors of thrombotic events.
Materials and Methods
The study was conducted during a period of one year (1/1/2013 to 31/12/2013) in the Department of Medicine and Hematology unit of Government Medical College, Kozhikode. It was a descriptive observational study and the subjects included were patients with polycythemia rubra vera already on follow up as well as newly diagnosed cases during the study period.The disease was diagnosed using the clinical diagnostic criteria for Polycythemia rubra vera [8].
a. Presence of high hematocrit values (more than 55% in males and more than 50% in females at the time of diagnosis) along with splenomegaly which cannot be attributed to any other cause or thrombocytosis and leukocytosis.
b. A secondary cause of elevated hematocrit is not evident in the clinical setting (like COPD, congenital cyanotic heart disease, capillary leak syndrome, renal artery stenosis, hepatocellular carcinoma and renal cell carcinoma) which are rued out with suitable investigations (like chest X - ray, 2- D Echocardiography, arterial blood gas analysis, USG abdomen with Doppler).
c. In cases where categorization was difficult between primary and secondary erythrocytosis, serum erythropoietin and JAK-2 mutation analysis were done.
All the study subjects were assessed by complete clinical evaluation followed by investigations to study the hematological profile of the disease and to exclude secondary causes of erythrocytosis. In those under follow up previous treatment records were also looked into to study the hematological parameters at the time of diagnosis. Patients with thrombotic events were then compared with those without them, with respect to various clinical and hematological parameters as well as their diet, lifestyle and environmental factors to look for the predictors of thrombotic episodes. Dietary information of the study subjects were estimated using a semi quantitative food frequency questionnaire derived from dietary risk assessment score used for non-communicable disease survey format (Phase I) by IDSP (Integrated Disease Surveillance Project). The data was entered using Microsoft Excel and analyzed using SPSS software.
Results
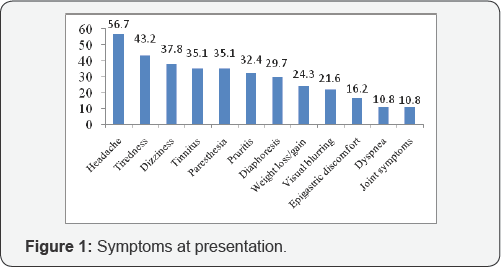
Of the 37 patients, there were 13 newly diagnosed cases of polycythemia rubra vera during the study period and others were diagnosed cases under follow up. The subjects were of age group between 45 years and 72 years and mean age at presentation was 57 years. 27(73%) of them were males. First clinical presentation was a thrombotic event in 32.4% of cases, symptoms of polycythemia in 54% and was an incidental detection in 11%. Headache (57%), dizziness (43%), tiredness (38%) and tinnitus (35%) were the most common symptoms (Figure 1) while conjunctival injection (84%) and splenomegaly (81%) were the common signs (Figure 2).
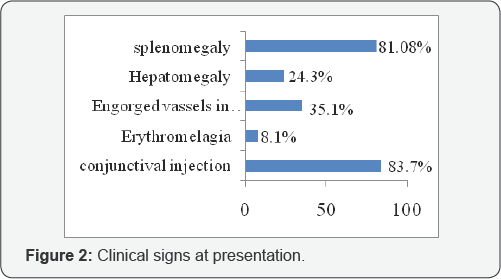
The characteristic symptom of pruritis was observed only in 32% of cases and only 22% had typical aquagenic pruritis. Erythromelalgia was observed only in 2(5.4%) patients. Splenomegaly was absent in seven cases (18.91%) of JAK-2 positive subjects (Figure 2) and majority (51.35%) of patients had only mild splenomegaly, massive splenomegaly was seen only in one case.
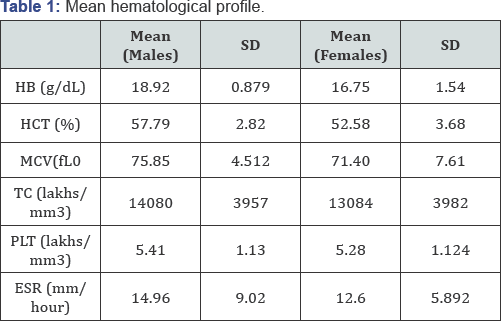
Mean hemoglobin level was 18.92g% in males and 16.75g% in females. Other hematological parameters in males and females respectively were: MCV (78.85fl, 71.4fl); total leukocyte count (14080/mm3, 13084/mm3); platelet count (5.41lakhs/mm3, 5.28lakhs/mm3) and ESR (14.9mm/hr, 12.6mm/hr) (Table 1).
Hemoglobin levels were less than the cutoff level for the diagnosis of polycythemia (18.5g% in males and 16.5g% in females) according to 2008 WHO criteria in 7 (18.91%) of our diagnosed cases of polycythemia rubra vera and these patients still had signs and symptoms described for polycythemia. The most common findings in peripheral smear were erythrocytosis with microcytic hypochromic RBC's, leukocytosis and thrombocytosis.
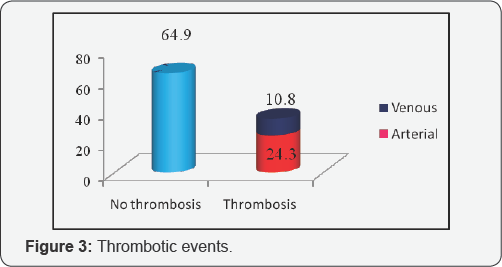
Thrombotic event occurred in 35.1% of study subjects, of which 24.3% were arterial thrombosis (Figure 3). Of the 10.8% cases of venous thrombosis, there was a single case of Budd Chiari syndrome, which was the only case of mortality during the study period.
There were significantly higher levels of hemoglobin (19.37g% vs 18.61g%; P=0.026), total leukocyte count (17,600/ mm3 vs 11700/mm3; P=0.001) (Figure 4) and uric acid (8.31mg/ dl vs 6.78mg/dl; P=0.001) in those with thrombotic events when compared to patients without thrombotic event, even though no such difference could be obtained in terms of platelet count (5.28lakhs/mm3 vs 5.43lakhs/mm3; P=0.705).
Consumption of fruits, vegetables, pulses were low in patients with thrombotic events when compared to those without thrombotic events (P=0.01), as assessed using modified ICMR food frequency questionnaire.
Discussion
There is a much higher male preponderance (male: female ratio of 2.7: 1) when compared to previous studies (ratio ranges between 1.2:1-2.2:1) [1]. This could be the situation in the population here or due to smaller number of subjects included in the study.
In the present study, 37.8% were of the age group more than 60 years and another 35.1% in the age group between 50 and 60 years. This was comparable with previous studies where the peak incidence of the disease was between 60 to 80 years.
Symptoms of polycythemia were the initial presentation in 54.1% of study subjects. Of all those who were symptomatic (including those with thrombotic event as initial presentation), most common symptoms were headache (57.7%), tiredness (43.2%), dizziness (37.8%), tinnitus and paresthesia (both 35.1%). This was consistent with the findings of the original polycythemia study group [1] where the common symptoms described where headache (48%), weakness (47%), dizziness (43%) and diaphoresis (33%).
Even though aquagenic pruritis has been described in the literature as a common (nearly 50%) [9,10] and characteristic symptom of polycythemia vera, we could observe pruritis only in 32.4% of cases and pruritis after water bath only in 21.6% of cases. And most of them had platelet count more than 4lakhs/ mm3. But none of them had pruritus as the sole presenting complaint which lead to the diagnosis of polycythemia Vera. Three patients with features of erythromelalgia had a platelet count more than 7lakh/mm3, though it was observed at platelet counts even more than 4 lakhs/mm3 according to previous studies [11,12]. Splenomegaly was found to be absent in 19%, which was as high as 30% in some of the previous studies. Hence splenomegaly is only a supportive criterion for the diagnosis of polycythemia vera and its absence does not rule out the diagnosis. Mean hemoglobin, hematocrit and other cell counts were more in males as compared to females this difference could be due to the normal physiological and hormonal influences. Mean corpuscular volume was also lower than normal in either sexes which can be explained by the depletion of marrow iron stores that occurs due to erythrocytosis. Mean ESR was 15 mm in first hour in males and 13mm in females and but in previous studies very low ESR values have been reported, and this difference could be due to lower mean hematocrit levels in our study population. Seven subjects had hemoglobin levels less than the WHO cutoff values. This may be due to the fact that hemoglobin levels in general population in our country is less than that of the western population, probably due to high prevalence of nutritional deficiencies. So a significant rise in hemoglobin levels from the baseline in patients with polycythemia vera may not amount to the cutoff levels set by the international standards. Clinical implication of this is that, suspicion of the clinical diagnosis of polycythemia vera may get delayed and patients may get subjected to unnecessary investigations for the evaluation of splenomegaly or symptoms of polycythemia. These subjects were included in the present study after confirming the diagnosis of polycythemia vera by doing JAK-2 mutation analysis. Interestingly all these patients had symptoms described for polycythemia even at lower hemoglobin levels.
Thrombotic events accounted for more than 30% of morbidity according to the observations by the Polycythemia Vera Study Group (PVSG), more than 50% of which were arterial thrombosis and its incidence increases with age and with other risk factors for thrombosis [1]. In the present study, thrombotic events occurred in 35 % of study subjects, of which 24% were arterial and 11% were venous thrombosis. Though more than 80% of fatal thrombotic events were arterial as per PVSG observations, the only case of mortality during the present study was due to a venous thrombosis which was Budd Chiari syndrome. Those with thrombotic event had higher hemoglobin, hematocrit, total leukocyte count as compared to those without thrombotic events and was statistically significant. On comparing with previous studies, hemoglobin, hematocrit and age at diagnosis were the risk factors consistently associated with thrombotic events, due to increased blood viscosity [3]. Leukocytosis has been suggested as an adverse risk factor for thrombotic event in some previous studies [13,14] probably due to its activation and interaction with platelets and endothelial cells.
There was no association between platelet counts and thrombotic events in the present study. Platelet count as a risk factor for thrombosis has been demonstrated only in very few studies [15], but the degree of thrombocytosis was not consistently correlated with the thrombotic risk. Indeed there is an increased risk of bleeding at very high platelet counts due to acquired Von-Willibrand factor deficiency, especially when platelet counts are more than 10lakhs/mm , but none of our patients had such very high platelet counts and bleeding complications were not noticed. Patients with thrombotic events had significantly higher uric acid levels when compared to subjects without thrombotic event. But this association could be an indirect evidence of high cell turn over secondary to high cell counts involving all myeloid lineages. None of the previous studies have demonstrated such an association. Even though uric acid levels were high, none of the patients in the present study had gouty arthritis or tophaceous deposits, but there were non-specific joint symptoms in a few patients.
Study subjects with adequate consumption of fruits, vegetables and pulses had a lower incidence of thrombotic events. The reasons for this association are unclear. However, some studies have proven that low fiber intake is associated with higher activity of plasminogen activator inhibitor-1 (PAI-1), the body's main inhibitor of fibrinolysis [16]. And these findings though simple are important and often neglected, but can be put into clinical practice very easily and may bring positive outcomes on health and treatment expenses.
References
- GruppoItaliano Studio Policitemia (1995) Polycythemia vera: the natural history of 1213 patients followed for 20 years. Ann Intern Med 123(9): 656-664.
- Adamson JW, Fialkow PJ, Murphy S, Prchal JF, Steinmann L, et al. (1976) Polycythemia vera: stem-cell and probable clonal origin of the disease. N Engl J Med 295(17): 913-
- Lawrence JH, Berlin NI, Huff RL (1953) The nature and treatment of polycythemia; studies on 263 patients. Medicine (Baltimore) 32(3): 323-388.
- Marchioli R, Finazzi G, Landolfi R, Kutti J, Gisslinger H, et al. (2005) Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J ClinOncol 23(10): 2224-2232.
- Finazzi G, Barbui T (2007) How I treat patients with polycythemia vera. Blood 109(12): 5104-5111.
- Berlin NI (1975) Diagnosis and classification of the polycythemias. SeminHematol 12(4): 339-351.
- James C, Ugo V, Le Couedic JP, Staerk J, Delhommeau F, et al. (2005) A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemiavera. Nature 434(7037): 1144-1188.
- Sasidharan PK (2008) Polycythemia how and how much to investigate? Medicine Update. Associations of Physicians of India 18(46): 358-367.
- Steinman HK, Kobza-Black A, Lotti TM, Brunetti L, Panconesi E, et al. (1987) Polycythaemia rubravera and water-induced pruritus: blood histamine levels and cutaneous fibrinolytic activity before and after water challenge. Br J Dermatol 116(3): 329-333.
- Jackson N, Burt D, Crocker J, Boughton B (1987) Skin mast cells in polycythaemiavera: relationship to the pathogenesis and treatment of pruritus. Br J Dermatol 116(1): 21-29.
- van Genderen PJJ, Michiels JJ (1994) Erythromelalgic, thrombotic and haemorrhagic manifestations of thrombocythaemia. Presse Med 23(Suppl 1): 73-77.
- Landolfi R, Ciabattoni G, Patrignani P, Castellana MA, Pogliani E, et al. (1992) Increased thromboxane biosynthesis in patients with polycythemia vera: evidence for aspirin-suppressible platelet activation in-vivo. Blood 80(8): 1965-1971.
- Gangat N, Wolanskyj AP, Schwager SM, Hanson CA, Tefferi A (2009) Leukocytosis at diagnosis and the risk of subsequent thrombosis in patients with low-risk essential thrombocythemia and polycythemia vera. Cancer 115(24): 5740-5745.
- Gangat N, Strand J, Li CY, Wu W, Pardanani A, et al. (2007) Leucocytosis in polycythaemiavera predicts both inferior survival and leukaemic transformation. Br J Haematol 138(3): 354-358.
- Ohyashiki K, Akahane D, Gotoh A, Ito Y, Tauchi T, et al. (2007) Uncontrolled thrombocytosis in polycythemia vera is a risk for thrombosis, regardless of JAK2 (V617F) mutational status. Leukemia 21(12): 2544-2545.
- Boman K, Hellsten G, Bruce A, Hallmans G, Nilsson TK (1994) Endurance physical activity, diet, and fibrinolysis. Atherosclerosis 106(1): 65-74.






























