Formulation and In-vitro Characterization of Aceclofenac Emulgel Prepared using Carbopol 934 and Sodium CMC
Bipindra Pandey1ϯ, Laxman Subedi1, 2ϯ and Rishiram Baral1, 3*
1School of Health and Allied Sciences, Faculty of Health Science, Pokhara University, Nepal
2Department of Biomedicine, Health & Life Convergence Sciences, Biomedical and Healthcare Research Institute, Mokpo National University, South Korea
3Research Institute of Pharmaceutical Sciences, College of Pharmacy, Kyungpook National University, Daegu, South Korea
ϯThese authors contributed equally to this work.
Submission: June 08, 2022; Published: July 20, 2022
*Corresponding author: Rishiram Baral, School of Health and Allied Sciences, Faculty of Health Science, Pokhara University, Pokhara, Nepal, Research Institute of Pharmaceutical Sciences, College of Pharmacy, Kyungpook National University, South Korea
How to cite this article: Bipindra P, Laxman S, Rishiram B. Formulation and In-vitro Characterization of Aceclofenac Emulgel Prepared using Carbopol 934 and Sodium CMC. Nov Appro Drug Des Dev. 2022; 6(3): 555688. DOI: 10.19080/NAPDD.2022.06.555688
Abstract
Emulgel is a recent novel drug targeting technique highly studied for control release of both emulsion and gel at a time. Delivery of the hydrophobic drug to its target site is mostly pronounced with the emulgel formulations. In this study, an attempt was made to formulate and evaluate an emulgel of aceclofenac by varying the concentration of both gelling bases and the oil phase of an emulsion. Two types of gelling agents namely carbopol 934 and sodium CMC were used followed by sesame oil and light liquid paraffin (LLP) as two different oils. Overall twelve formulations were formulated, and their quality parameters were evaluated. The aqueous phase and oil phase was prepared and mixed together to form an emulsion. The gel and the emulsion were mixed together for the preparation of an emulgel. The prepared emulgel was then evaluated for physical properties, pH, spreadability, viscosity, method validation, drug content, and drug release. All emulgel showed acceptable physical properties concerning color, homogeneity, consistency, and pH value. The F1 formulation showed better result whose drug content was found to be 97.84 ±0.439 showing drug release rate as 30% (using carbopol 934) and 23.18% (using Sodium CMC) at 5 hours. Among all emulgel formulations, carbopol 934 formulation with sesame oil as an oil phase showed superior drug release compared to other.
Keywords: Emugel; Aceclofenac; Gelling agent; Carbopol 934; Sodium CMC; Sesame oil; Light liquid paraffin
Abbreviations: LLP: Light Liquid Paraffin; TDDS: Topical Drug Delivery System; NSAID: Non-Steriodal Anti-Inflammatory Drug; COX: Cyclo-Oxygenase; FTIR: Fourier-Transform Infrared
Introduction
The Advancement in pharmaceutical technologies has encouraged to formulate alternative drug targeting approach which efficiently and effectively reaches the site of action. The treatment of illness is accomplished by administering a drug by different routes in the human body that are oral, sublingual, parental, topical, rectal etc. Topical drug administration is usually preferred when other route fail to address the site specification [1,2]. Topical drug administration is one of the simplest and easiest routes for localized drug delivery. The positive aspects of topical drug delivery system (TDDS) is it aids to overcome first pass effect, avoid gastric irritation, and prevent metabolic degradation associated with oral administration [3]. Gel formulation provides a faster drug release than other topically application procedures such as ointment and creams. In spite of the many advantages of gel, a major limitation is the delivery of hydrophobic drugs which could be overcome by an emulsion-based approach refer to as emulgel. When gel and emulsion are used in combination, emulgel is formed. In emulgel, polymers can function as emulsifiers and thickeners. The gelling capacity of these compounds allow the formulation of the stable emulsion by decreasing surface and interfacial tension and increasing the viscosity of the aqueous phase. The hydrophobic therapeutic moiety can be incorporated successfully, provides the degree of elegance, can be easily washed off [4].
Carbopol 934, hydroxypropyl methylcellulose, Sodium carboxymethylcellulose (Sodium CMC) are widely used gelling agents in emulgel formulation. Followingly, propylene glycol, light liquid paraffins, sesame oils etc. are the oil phase used in the formulation design. Change in the amount and type of these gelling agents as well as oil phase alter the pharmacokinetic parameters of the emulgel formulation to great extent [3]. Two phases of emulsion such as both oil in water emulsion and water in oil emulsion can be used as vehicle to deliver the drug inside skin [5]. Water phase containing gelling agent converts an emulsion into an emulgel. Oil in water system is used for encapsulating lipophilic drugs whereas water/oil system is used for hydrophilic drug [6]. Basic properties of the emulgel are thixotropic, greaseless, easily spreadable, easy removal, water soluble, non-staining, pleasing appearance, and long shelf-life. They are used to deliver various drugs across skin to enhanced drug absorption due to emulsionbased approach.
Aceclofenac (ACF), chemically known as 2- [(2, 6-dichlorophenyl) amino] phenylacetoxyacetic acid is categorized as non-steriodal anti-inflammatory drug (NSAID) which effectively inhibits the cyclo-oxygenase (COX) enzyme. Inhibition of COX results in the decrease synthesis of prostaglandins which are inflammatory mediators that cause pain, swelling, inflammation and fever. High permeability of ACF through synovial joints assist in the treatment of joint pain caused by loss of articular cartilage, tenderness, stiffness, crepitus, and local inflammation in patients with osteoarthritis and related conditions. Aceclofenac can also be used in other painful conditions such as dental and gynecological conditions [4,7]. Currently available dosage form of ACF are crowned with suborders side effects though they have significant patient satisfaction. The chemically modified ACF, known as diclofenac is still not able to subside all the side effects of ACF though it enhance the gastrointestinal tolerability of the drug [8]. Therefore, new approach of targeting ACF to the specific site of action is the optimal requirement.
The rationale of this study is to increase the penetration of ACF into the skin using emulgel technology. We know that the most widely cited side-effect of NSAIDs include, gastrointestinal ulcer, accompanied by anaemia due to bleeding, which is also true for aceclofenac, on the administration of conventional dosage form. In order to avoid such shortcomings topical administration serve as a promising approach. In this study, we are trying to formulate a new formulation of ACF in emulgel for a topical application which is expected to improve patient compliance and skin penetration, in comparison with conventional dosage form. Emulgel is used to counteract the adverse effects of drug compounds that make the formulation more tolerable.
Materials and Methods
Chemical and Reagents
Aceclofenac (ACF) was provided by Florid Pharmaceuticals Pvt. Ltd, Lalitpur Nepal as bounteous gift. Sodium CMC and carbopol 934 used as Gelling agent, light liquid paraffin and sesame oil as oil phase, tween 80 as an emulsifier, methylparaben and propylparaben as a preservative were purchased from Hi Media Laboratories Pvt. Ltd. Dehydrogenase alcohol as co-solvent, propylene glycol as a penetration enhancer, BHT as an antioxidant, triethanolamine as PH adjuster and phosphate buffer (PH 7.4) were purchased from Tokyo Chemical Industries. All chemical and reagents used were of analytical reagent grades.
Formulation Matrix: *Sufficient quantity was added to maintain the pH of an emulgel.
Each formulation consist of 100 g emulgel (Table 1 & 2).
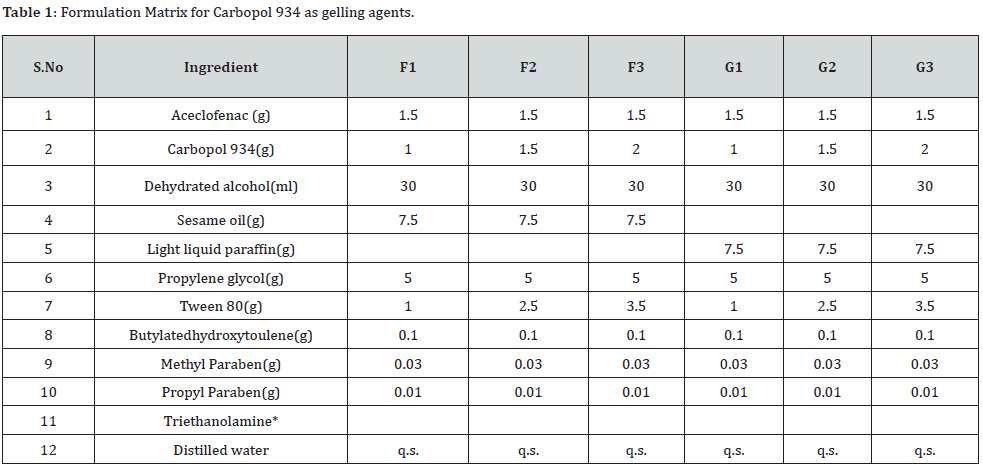
*Sufficient quantity was added to maintain the pH of an emulgel.
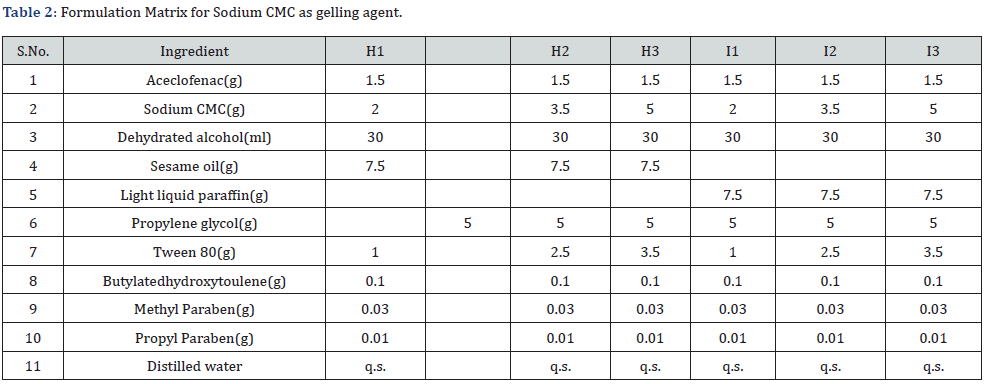
Each formulation consist of 100 g emulgel
Identification of raw material (aceclofenac)
The melting point of raw aceclofenac was observed by using melting point apparatus as per IP 2010. The 20ppm solution of ACF dissolved in methanol was observed by UV-visible spectrophotometer in absorbance range of 220 nm to 373 nm. The maximum absorbance was then noted. Concentration range of ACF was prepared by the serial dilution technique.
Preparation of Aqueous Phase:
The aqueous phase of the emulsion was prepared by dissolving Tween 80 in distilled water. Methyl Paraben and Propyl Paraben was dissolved in propylene glycol whereas the drug was dissolved in ethanol and both solutions are mixed with the aqueous phase.
Preparation of Oil Phase
The required amount of oil was weighed. Both the oil phase and aqueous phase were heated to 75⁰C separately. Then the oil phase is added to the aqueous phase with continuous stirring until cooled to room temperature.
Preparation of Gel: The gel bases were prepared by dispersing different concentrations of polymers in distilled water separately with constant stirring at a moderate speed using a mechanical shaker. The pH of all formulations was adjusted to5.5- 6.5 using triethanolamine (TEA).
Preparation of Emulgel: The obtained emulsion was mixed with the gel with gentle stirring to obtain the emulgel.
Physical characterization and In-vitro Pharmacokinetics study
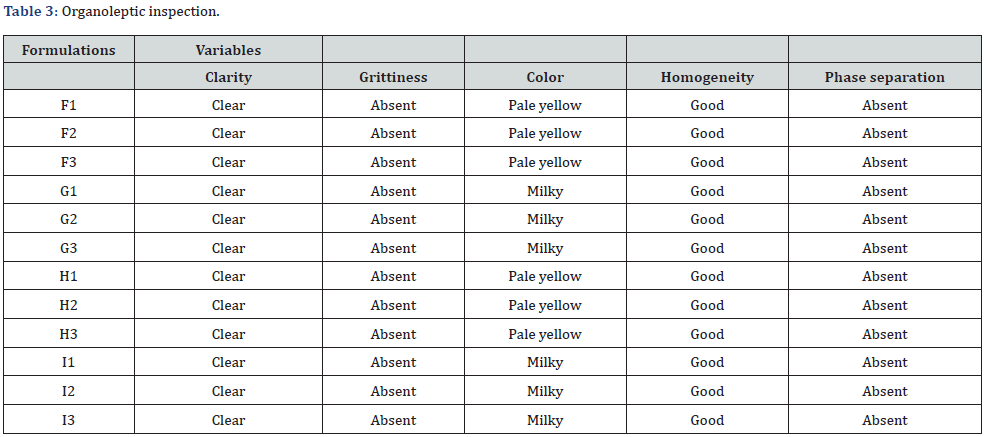
Organoleptic Inspection: The emulgel was observed for clarity, color, grittiness, phase separation, homogeneity (Table 3).
pH measurement: One gram of emulgel was dissolved in 100ml of distilled water and stored for 2 hours. Finally, pH was measured using a digital pH meter previously calibrated using standard hydrogen chloride of pH solution 4.0 and phosphate buffer of pH 7.01. Experiment was performed in triplicate.
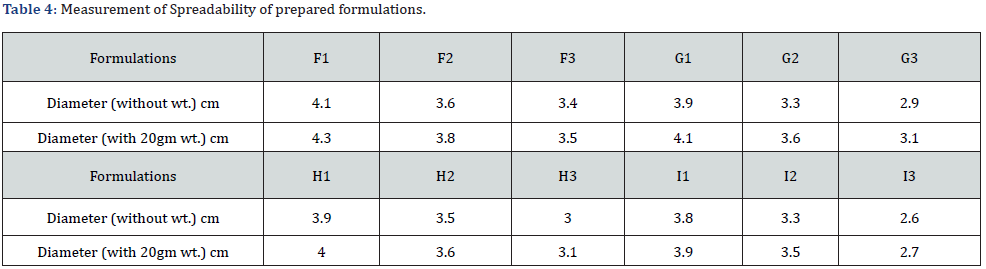
Spreadability: 300 mg of emulgel was weighed and placed on a petri-dish and another glass plate (47.59 g) was dropped from a certain distance. The diameter (cm) of spread, emulgel was then measured (Table 4).
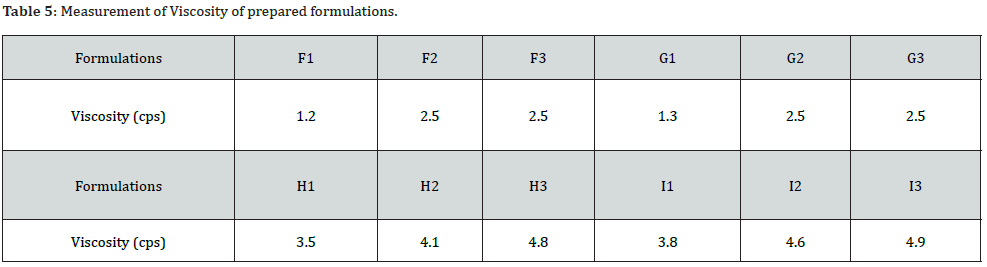
Viscosity: One gram of emulgel was dissolved in 100ml distilled water and filled in Ostwald’s viscometer. The solution was allowed to pass from mark A to mark B. Then the time (t) required to reach from A to B was noted using stopwatch.
Analytical method validation
Linearity: A stock solution of ACF was prepared in PBS (pH=7.4) of concentration 500 ppm and diluted to prepare the solutions of 15, 20, 25, 30 and 35 ppm concentration. 30ppm solution w was observed in UV spectrophotometer in spectrum mode to determine the absorption maximum. The absorbance of all the remaining working solutions was then observed at the absorption maximum. The standard plot of concentration versus absorbance was then obtained.
Accuracy and precision
Among twelve formulations, six samples (of 1g) were taken from each of them and then diluted to prepare 150 ppm solution which was then diluted to 30 ppm solution. The solution was then observed in a UV. Accuracy and precision were then determined from the obtained data.
Drug Content
1gm aceclofenac topical emulgel (equivalent to 15mg aceclofenac) was weighed and dissolved in 100 ml phosphate buffer (pH 7.4). The solution was filtered and diluted to 30 ppm. The volumetric flask was kept for 2 hours and shaken well for proper mixing. Then absorbance was measured spectrometrically at 273 nm.
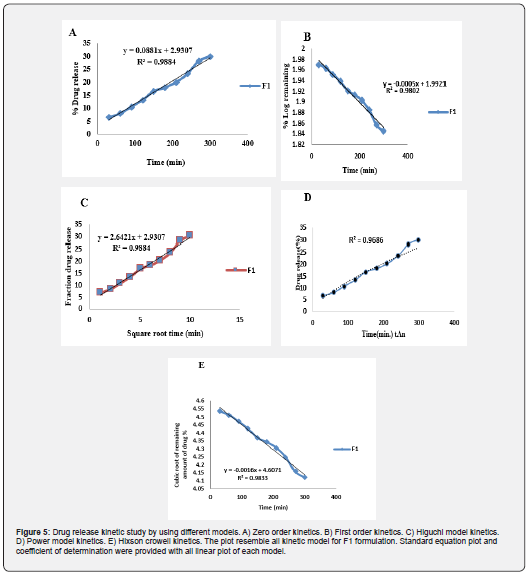
Drug Release Kinetic Study
To analyze the mechanism of drug release from the topical emulgel, the release data were fitted to the following equations:
Zero order equation: 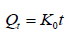
Where, Q is the percent of drug released at time t, and K0 is the zero order release rate.
First - order equation: 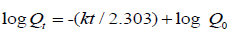
Where, Q0 is the initial amount of drug and Qt is the remaining amount of drug after time t and k is the first order release rate.
Higuchi’s equation: 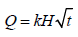
Where, Q is the percent of drug release at time t, and kH is the Higuchi constant.
Hixoncrowell model: 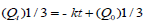
Where, Q0 is the initial amount of drug and Qt is the remaining amount of drug after time t and k is the Hixoncrowell release rate.
Power law: 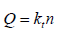
Where, Q is the percent of drug released at time t, and k is the release rate and n is the release exponent.
Results and Discussion
Identification
CThe melting point of raw ACF was observed at 151⁰C within specification of 149 -153 ⁰C according to IP. As per IP 2010, the maximum absorbance was absorbed at 275 nm for the 20ppm solution of ACF prepared in methanol which coincide with our result.
Organoleptic Inspection
The general evaluation for emulgel was performed by organoleptic inspection. All the formulations of emulgel were found to be spotless. Similarly, the color of emulgel differed according to the use of oil phase such as, Carbopol 934 (F1-F3) and Sodium CMC (H1-H3) with sesame oil containing preparation were pale yellowish in color while carbopol 934 (G1-G3) and sodium CMC (I1-I3) with light liquid paraffin emulgel were milky white. Moreover, formulations were free from grittiness which was admitted by inspecting emulgels from top, middle and bottom sides. Meanwhile, the preparation was smooth with good homogeneity. Furthermore, the phase separation of emulgel was evaluated visually throughout the study period in room temperature and no deformities were observed.
Evaluation of Physicochemical Properties
Measurement of pH
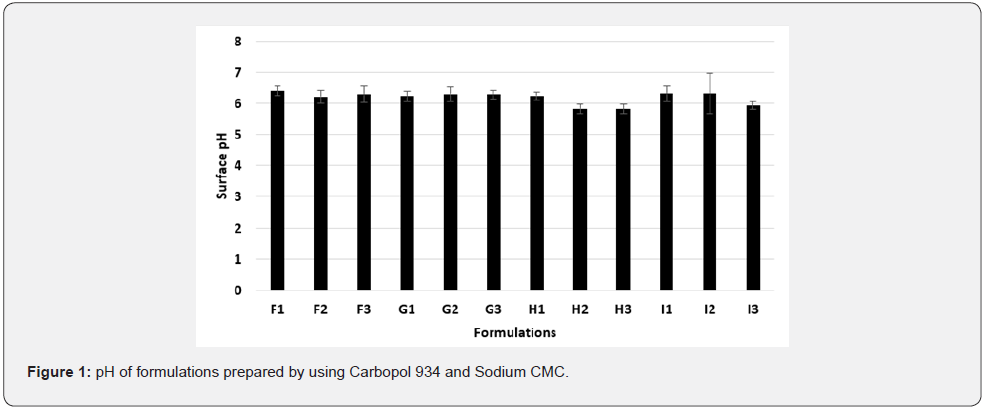
The pH is one of the crucial factor that are measured for semisolid preparation, in order to determine the acidity or alkalinity of the product. The pH of most of the formulations were within the range of 5.5 to 6.5, which is considered acceptable to avoid the risk of irritation upon application to the skin as per IP (Figure 1).
Spreadability
From the result it was depicted that the spreadability of the formulations (F1-F3, G1-G3) with carbopol 934 as a gelling agent were higher as compare in the formulation (H1-H3, I1-I3) with sodium CMC as a gelling agent. In both formulations (carbopol 934 and sodium CMC), there was a decrease in spreadability with the increase in the concentration of the gelling agent. This showed the inverse relationship between the concentration of the gelling agent and the spreadability. Moreover, spreadability was found to be better in a formulation that used sesame oil as an oil phase than in formulations where liquid paraffin was used as an oil phase. It was observed that with the increase in the viscosity of emulgel the spreadability decreases.
Rheological properties
The viscosity of all the formulation was measured by Ostwald’s Viscometer. The viscosity of F1 (carbopol 1%) was found to be lower i.e.1.24cps which may be due to the use of a lower concentration of carbopol. Meanwhile, the formulation F3 (carbopol 2%) showed viscosity of 2.48cps. Similarly, the viscosity of H1 (Sodium CMC 2%) was found lower i.e. 3.45cps and H3(Sodium CMC 5%) to be 4.75 cps. The result indicated that the viscosity of the emulgel depend on the concentration of gelling agent used (Table 5).
Analytical method validation
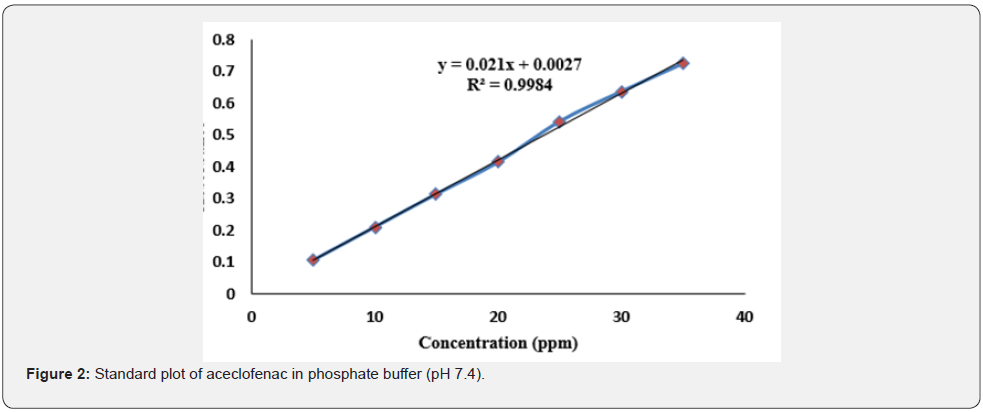
Standard curve of ACF was plotted using Phosphate buffer with seven different concentrations. Linear plot was obtained with acceptable coefficient of determination (Figure 2).
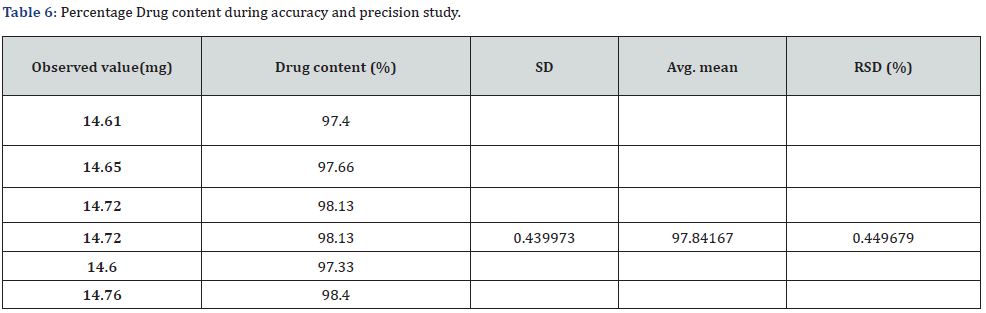
Accuracy and Precision
The drug content study for accuracy and precision showed that the observed values were near the true value. Therefore, the method which was flowed in our study was accurate. Moreover, the RSD was less than 2% depicting the precision of our study (Table 6).
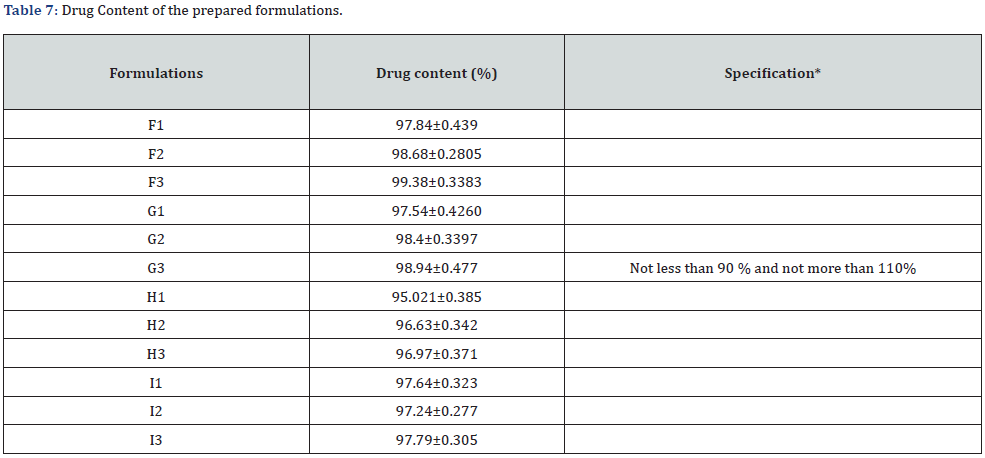
Drug Content
The results of the study revealed that the drug content of the prepared formulations lie within the acceptable range of 90 to 110 % (Table 7).
In Vitro Release Study (Using Diffusion Cell)
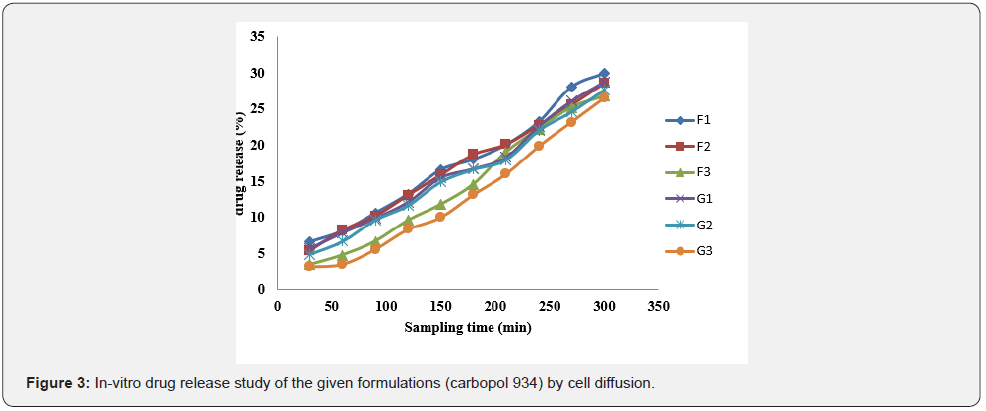
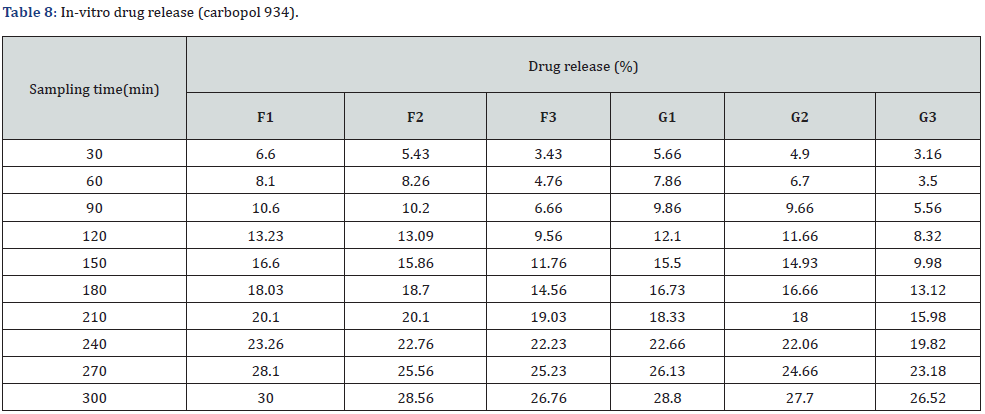
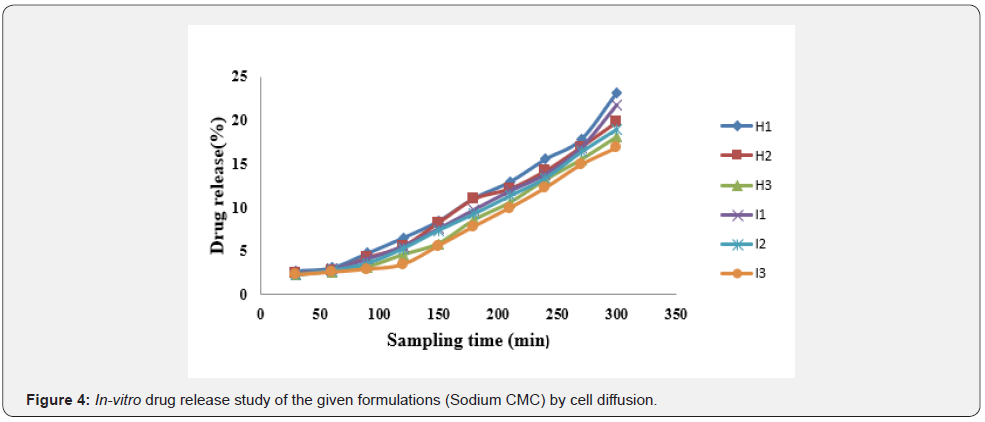
In vitro drug release study of emulsified gel was carried out for 5 hours using diffusion cell and percentage of drug release are shown in Table 8 & Table 9 (Figures 3-5).
In-vitro drug release study showed that % drug release was found to be highest in the formulation F1 i.e. 30% where carbopol 934 (1%). Among G1-G3 formulation G1 (carbopol 934 1%) had greater drug release i.e. 28.8%. Similarly, H1 (sodium CMC 2%) had a greater drug release among formulations H1-H3. And I1 (sodium CMC 2% had a greater drug release among formulations I1-I3. The study was carried out for 5 hours sampling for every 30 minutes (Table 10-12).
Dry bulb temperature= 190C, Wet bulb temperature=16oC
Data are expressed as mean ± standard deviation (n=6)
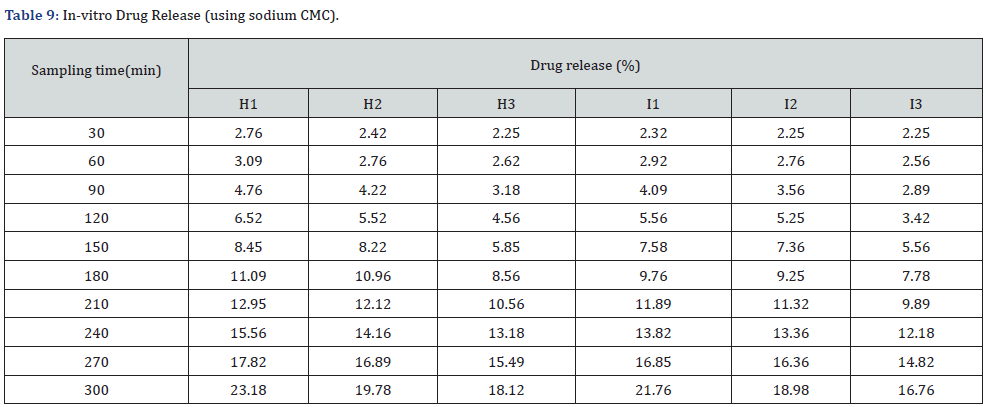
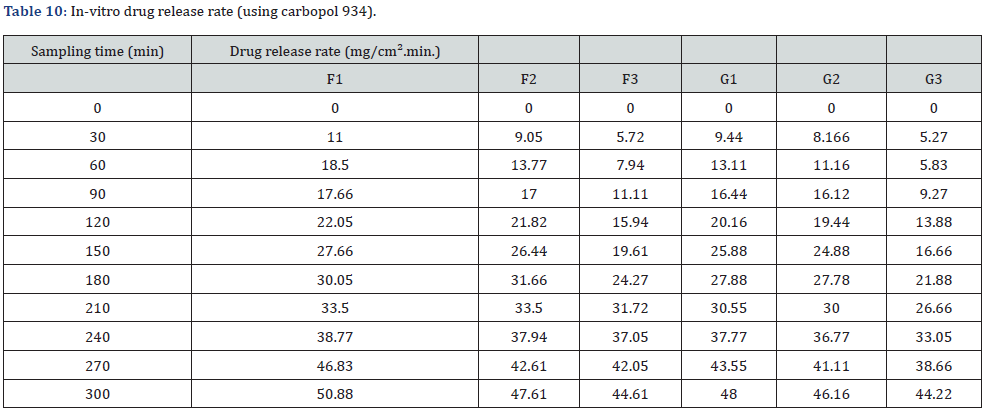
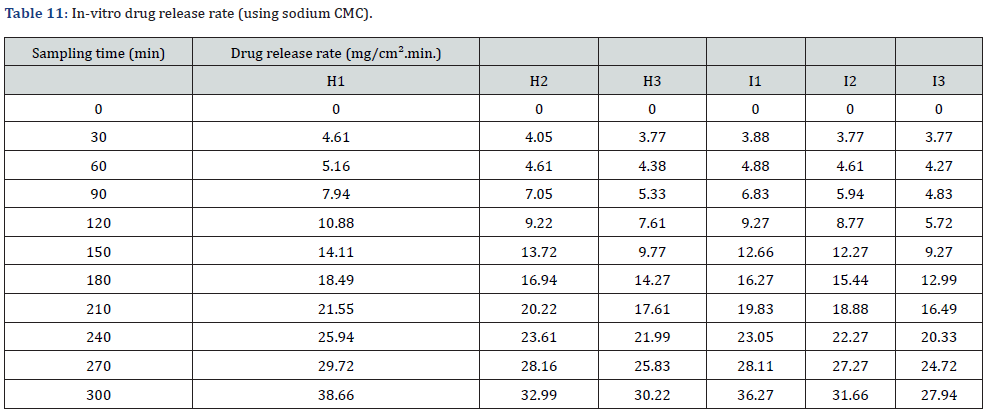
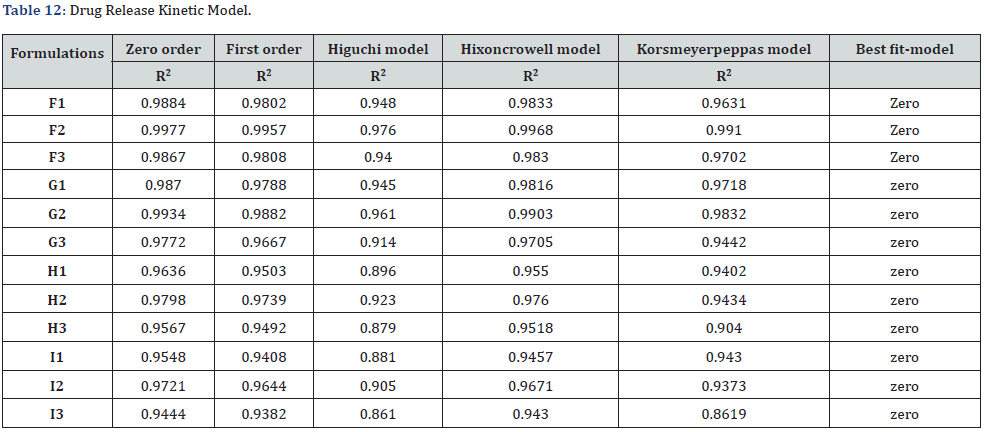
The best fitted model for drug release for all of the formulations was found to be zero order.
Discussion
ACF belongs to class II of BCS having the properties of low solubility and high permeability with hydrophobic nature [9]. ACF is a phenylacetic acid derivatives categorized as anti-inflammatory and analgesic. The well known therapeutic indication of ACF are osteoarthritis, rheumatoid arthritis, post-traumatic pain, ankylosing spondylitis, etc. ACF is immediately and effectively soaked after oral administration but have comparatively shorter half-life of 4-4.3 h [10]. Furthermore, side effects and complications associated with ACF are gastrointestinal disturbances, peptic ulceration, gastrointestinal bleeding, abdominal pain, diarrhea, vomiting, skin rash, dizziness etc. [11]. Therefore, sustained, and controlled release formulations are utmost important to reduce the dosing frequency with improved bioavailability as well as with improve safety and efficacy of the medication [12].
Improving the specific site of action of active pharmaceutical ingredients without compromising their pharmaceutical ability is another utmost requirement in medical and pharmaceutical fields [13]. Targeting of lipophilic molecule to the specific site of action is a challenging and tedious task due to low aqueous solubility resulting in poor bioaccessibility and bioavailability [14]. To overcome this aspects, one of the most commonly entertained method is embedding the lipophilic compounds in oil-in-water (O/W) emulgel formulation [15]. Therefore, this study was designed for the formulation of emulgel dosage form expected to be useful for topical application for hydrophobic drug ACF. Furthermore, we aimed to increase the stability of emulsion and increase penetration by altering the concentration of gelling agent Carbopol 934 and Sodium CMC.
To determine the compatibility of two gelling agents Carbopol 934 and Sodium CMC, several literature review was done. In a study by Nayak et al. 2021, formulation, development, and physicochemical characterization of diclofenac topical emulgel was performed. The study used Carbopol 934, hydroxypropyl methyl cellulose and Sodium CMC as gelling agent. The study showed that the drug release profile in the formulation with Carbopol 934 and Sodium CMC lies within the acceptable range and drug stability and bioavailability in the presence of these gelling agent is satisfactory [16]. Another study was performed by Pakhare et al. 2017 to explore the potential of emulgel in enhancing the topical delivery of diclofenac potassium. In this study, compatibility of diclofenac with Carbopol 934 and Sodium CMC was studied using Fourier-transform infrared (FTIR) and Differential scanning calorimetry (DSC). The FTIR and DSC study proved that the drug and excipients are compatible with each other [17]. By taking these results into consideration we have chosen Carbopol 934 and Sodium CMC for our study.
The result from the study showed that all the formulations are clear without spots, grittiness free, have pale yellow and milky color, good homogeneity and have no phase separation. These all meet the criteria of IP specification indicating that the formulation prepared have acceptable organoleptic properties. Spreadability is another criteria for an emulgel to meet the ideal requirement. Spreadability denote the extent to which drug is readily spreads on the skin surface when applied [18]. The result from our study showed that the spreadability was found to be better in a formulation that used sesame oil as an oil phase than in formulations where liquid paraffin was used. It was observed that with the increase in the viscosity of emulgel the spreadability decreases.
Viscosity is an important physical property in a topical formulation, which affects the rate of drug release. In general, an increase in the viscosity there is a consequent decrease in the rate of drug release. Similarly, viscosity is a parameter that can affect the other mechanical and physical properties like spreadability and consistency of product. Furthermore, it is also related to the release of emulgel from the container as well as the application on the target site [19]. The result of our study indicated that the viscosity of the emulgel depend on the concentration of gelling agent used as the F1 with least Carbopol showed less viscous properties while the I3 with high concentration of gelling agent seems to be highly viscous.
Moreover, in-vitro release study is a critical criteria to assess the safety, efficacy and quality of nanoparticle based drug delivery system but the compendial standard are still not available making the direct comparision difficult [20]. However, three widely used method to determine the in vitro release of modified formulations like emulgel are continuous flow, dialysis membrane and sample and separate methods [21]. In this study we adapted dialysis membrane technique and result showed that with an increase in the concentration of the gelling agent the drug release was decreased. This is because of the increase in viscosity on increasing the concentration of gelling agent. Hence the formulation using carbopol 934 had superior drug release than the formulation using sodium CMC. Also, the formulation using carbopol 934 (sesame oil) has the highest drug release i.e. F1 formulation.
Conclusion
Gel provides an aqueous environment to the drug. It favor its dissolution and provides a quicker release of the drug as compared to other topical delivery systems. Due to the limitation of gel to deliver hydrophobic drugs, an emulsion-based approach is selected to overcome it. Emulsion based gel provides a suitable medium for the delivery of such hydrophobic drugs where drugs can be incorporated into its oily phase and delivered to the site of action. In the present study, attempt has been made to know the effect of alteration in concentration of the polymers (carbopol 934 and sodium CMC) in organoleptic and in-vitro drug release properties of emulgel. Also, the properties of emulgel were evaluated by altering the concentration in oil phase (sesame oil and LLP). From the result, it is seen that Carbopol 934 showed improved matrix than sodium CMC. Similarly, it was concluded that sesame oil-based emulgel has optimal drug release properties in comparison to light liquid paraffin. Entire study concludes that release kinetic of the emulgel formulations are dependent on the concentration and type of gelling agents used as well as the nature of the oil phase selected. Further cell-based and in-vivo experiments are desired to extraplot the possibility of formulation of ACF as emulgel with the concentrations of gelling agents and oil phase depicted from our study.
Acknowledgment
This work was supported by Asian College for Advance Studies, Lalitpur, Nepal. We would like to express our gratitude to Florid Pharmaceuticals Pvt. Ltd., Lalitpur, Nepal for providing Aceclofenac active ingredient as bounteous gift. The authors are immensely thankful to all the faculties of Department of Pharmacy in Asian College for Advance Studies.
Authors Contribution
Conceptualization, Bipindra Pandey, Rishiram Baral; methodology, Bipindra Pandey, Rishiram Baral, Laxman Subedi; investigation, Bipindra Pandey, Rishiram Baral, Laxman Subedi; writing-original draft preparation, Rishiram Baral, Bipindra Pandey, writing-review and editing, Rishiram Baral; supervision and project administration, Bipindra Pandey, Rishiram Baral.
References
1. Thomas J, Kuppuswamy S, Sahib AA, Benedict A, George E, et al. (2017) A review on emulgel as a current trend in topical drug delivery system. International Journal of pharmacy pharmaceutical science 9(3): 273-281.
2. Subedi L, Pandey P, Shim JH, Kim KT, Cho SS, et al. (2022) Preparation of topical bimatoprost with enhanced skin infiltration and in vivo hair regrowth efficacy in androgenic alopecia. Drug Deliv 29(1): 328-341.
- Acharya P, Baral R, Manandhar S, Shahi P, Gurung B et al, (2021) Formulation and In-vitro Characterization of Fluoxetine Hydrochloride Loaded Fast Dissolving Oral Film Using HPMC 15CPS and HPMC K4M. J Pharma Drug Develop 8(1): 101.
- Ashara Kalpesh C, Jignesh S, Mendapara Vishal P, Mori Nitin M. (2014) British Biomedical Bulletin.
- Ojha A, Ojha M, Madhav NS (2017) Recent advancement in emulgel: A novel approach for topical drug delivery. Int. J. Adv. Pharm 06(01): 17-23.
- Haneefa KM, Easo S, Hafsa P, Mohanta GP, Nayar C (2013) Emulgel: An advanced review. Journal of pharmaceutical sciences research 5(12): 254-258.
- Goci E, Haloci E, Xhulaj S, Malaj L (2014) Formulation and in vitro evaluation of diclofenac sodium gel. Int J Pharm Pharm Sci 6(6): 259-261.
- Manjanna K, Rajesh K, Shivakumar B (2013) Formulation and optimization of natural polysaccharide hydrogel microbeads of aceclofenac sodium for oral controlled drug delivery. American Journal of Medical Sciences Medicine 1(1): 5-17.
- Kaur L, Bala R, Kanojia N, Nagpal M, Dhingra GA, (2014) Formulation development and optimization of fast dissolving tablets of aceclofenac using natural superdisintegrant. International Scholarly Research Notices.
- Raza K, Kumar M, Kumar P, Malik R, Sharma G, et al. (2014) Topical delivery of aceclofenac: challenges and promises of novel drug delivery systems. BioMed research international.
- Iolascon G, Gimenez S, Mogyorosi D (2021) A Review of Aceclofenac: Analgesic and Anti-Inflammatory Effects on Musculoskeletal Disorders. J Pain Res 30(14): 3651-3663.
- Solanki SS, Dahima R (2011) Formulation and evaluation of aceclofenac mouth-dissolving tablet J Adv Pharm Technol Res 2(2): 128-131.
- Falsini S, Clemente I, Papini A, Tani C, Schiff S, et al. (2019) When Sustainable nanochemistry meets agriculture: Lignin nanocapsules for bioactive compound delivery to plantlets. ACS Sustainable Chem. Eng 7(24): 19935-19942.
- Ravanfar R, Abbaspourrad A (2019) l-Histidine Crystals as Efficient Vehicles to Deliver Hydrophobic Molecules. ACS applied materials interfaces 11(42): 39376-39384.
- Mohamed MI (2004) Optimization of chlorphenesin emulgel formulation. The AAPS journal 6(3): 81-87.
- Nayak A, Mandal SK, Ramadan MA, Rath SK (2021) Formulation, Development and Physicochemical Characterization of Diclofenac Topical Emulgel. Egyptian Journal of Chemistry 64(3): 1563-1573.
- Pakhare AV, Deshmane SV, Deshmane SS, Biyani KR (2017) Design and development of emulgel preparation containing diclofenac potassium. Asian Journal of Pharmaceutics 11(04).
- Singla V, Saini S, Joshi B, Rana A (2012) Emulgel: A new platform for topical drug delivery. International Journal of Pharma Bio Sciences 3(1): 485-498.
- Subedi L, Song SY, Jha SK, Lee SH, Pangeni R, et al. (2021) Preparation of topical itraconazole with enhanced skin/nail permeability and in vivo antifungal efficacy against superficial mycosis. Pharmaceutics 13 (5): 622.
- Weng J, Tong HH, Chow SF (2020) In vitro release study of the polymeric drug nanoparticles: development and validation of a novel method. Pharmaceutics 12(8): 732.
- Dsouza S (2014) A review of in vitro drug release test methods for nano-sized dosage forms. Advances in Pharmaceutics 2014: 12.






























