In Silico Molecular Docking Study on Selective Cyclooxygenase-2 Inhibitor Drugs For SARS-Cov-2 Active Main Protease
Aslınur Doğan1, Fatma Şengül2, Nebih Lolak1 and Suleyman Akocak1*
1Adıyaman University, Faculty of Pharmacy, Department of Pharmaceutical Chemistry, 02040, Adiyaman, Turkey
2Adıyaman University, Faculty of Pharmacy, Department of Biochemistry, 02040 Adıyaman, Turkey
Submission: February 7, 2022; Published: March 09, 2022
*Corresponding author: Suleyman Akocak, Department of Pharmaceutical Chemistry, Adıyaman University, Faculty of Pharmacy, Adiyaman, Turkey
How to cite this article: Aslınur D, Fatma Ş, Nebih L, Suleyman A. In Silico Molecular Docking Study on Selective Cyclooxygenase-2 Inhibitor Drugs For SARS-Cov-2 Active Main Protease. Nov Appro Drug Des Dev. 2022; 6(3): 555686. DOI: 10.19080/NAPDD.2022.06.555686
Abstract
The coronavirus (COVID-19) pandemic became one of the most important disease problem across the globe for last few years since there is no recommended efficacious drugs in the market. So, there is an urgent need for efficient drugs to treat this disease in the near future. In the present study, molecular docking analyses of selective cyclooxygenase-2 inhibitor drugs (Celecoxib, Rofecoxib, Valdecoxib, Lumiracoxib, Parecoxib, Etoricoxib, and Firocoxib) were performed against the therapeutic target proteins of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) main protease (Mpro) enzyme into the catalytic active site. On the other hand, these drugs were compared with standard drugs such as Favipiravir, Chloroquine and Hydroxychloroquine to understand the binding sites and find the best poses. The results revealed that all the selective cyclooxygenase-2 inhibitor drugs (except Lumiracoxib) showed a better binding affinity against SARS-CoV-2 Mpro enzyme than the standard drugs. Among them, Etoricoxib (-9.40 kcal/mol) have shown the best binding affinity. As a result, this study shows that these selective cyclooxygenase-2 inhibitor drugs might be interesting lead compounds to discover more potent SARS-CoV-2 Mpro inhibitors and find to cure severe COVID-19 disease with better drugs.
Keywords: Molecular docking; Coronavirus; COVID-19; Cyclooxygenase-2 inhibitor; In silico
Introduction
An outbreak was reported by the World Health Organization (WHO) in Wuhan, China in December 2019. This epidemic was named Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) on January 30, 2020 [1,2]. In the second week of March 2020, the epidemic was declared global pandemic and within a few months the number of cases increased to 4 million and the death rate increased significantly [3]. The viral agent causing the outbreak belongs to the betacoronavirus family [1,2]. The disease caused by the SARS-CoV-2 factor is highly contagious [4]. This illness is basically a type of viral infection that spreads rapidly through respiratory droplet and direct contact. This infection has many symptoms, such as fever, cough, shortness of breath and gastrointestinal diseases [3,5-7]. The COVID-19 epidemic, in addition to threatening human health, has also had negative effects in areas such as the global economy around the world [3].
Cyclooxygenases are enzymes that allow free fatty acids to convert into cyclic endoperoxides. Arachidonic acid and some other fatty acids are exposed to the action of this enzyme and forming various prostaglandins [8,9]. Studies have shown that there are two different isoforms of the enzyme [8]. The first isoform, known as COX-1, is the structural form and is continuously present in the region in which it is produced. The COX-2 isoform is the inducible form [10,11]. This enzyme isoform is induced, especially in cases that cause inflammation. As a result of increased expression of the enzyme COX-2, abundant prostanoids are formed. In the presence of systemic infection, this rate increases even more. It has been found that this isoform increases in various pathologies, such as certain types of cancer and diseases of the central nervous system [8].
Microorganisms stimulate processes associated with the immune system and inflammatory events in the tissues they attack. Elimination the inflammatory condition that occurs is very important for the treatment of diseases caused by infection. In this context, polyunsaturated fatty acids (PUFA) and their metabolites play a very important role. In studies, lipid derivatives have been found to kill various microorganisms [12]. In general, PUFA kill microbes by their direct effect on microbial cell membranes. Arachidonic, eicosapentaenoic and docosahexaenoic acids act as endogenous antibacterial and antifungals. These lipid molecules also have antiviral, antiparazit and immunomodulatory effects. Cytokines involved in cell defense induce the release of PUFA from the cell membrane. These lipid molecules provide the formation of lipoxins and resolvins that have antimicrobial effects [13]. A study has shown that COX inhibition protects the virus from spreading from cell to cell by a mechanism that inhibits cytomegalovirus maturation [14]. COX-2 inhibitors such as etoricoxib or celecoxib are drugs that contribute to a decrease in mortality in severe influenza. COX-2 inhibitors are thought to be secure in the treatment of COVID-19 and may reduce disease progression in groups of high risky elderly patients with pneumonia due to their treatment of inflammation [15].
It has also been shown in previous studies that the severity and course of the inflammatory process differ decidedly between male and female [16]. Simona Pace et al. found that isolated lipopolysaccharide causes more PGE2 production in males and this may be due to increased COX-2 expression [17]. It is also thought that PGE2 levels, which are an important lipid agent and enhance more in men, may be a factor that explains the more severe disease formation condition of COVID-19 in men [16]. The parallelism of the increase in PGE2 and disease rates suggests that COX-2 inhibitors may be effective in treatment.
The aim of this study is to evaluate the place of COX-2 enzyme inhibition in COVID-19 treatment as in silico. In addition to the effect of these drugs (Scheme 1) on suppressing inflammation and reducing the severity of the disease, the ability to bind to the SARSCoV- 2 factor will be evaluated. This attachment is very important in terms of preventing the viral factor from entering the cell and preventing the effects of the disease on the body.
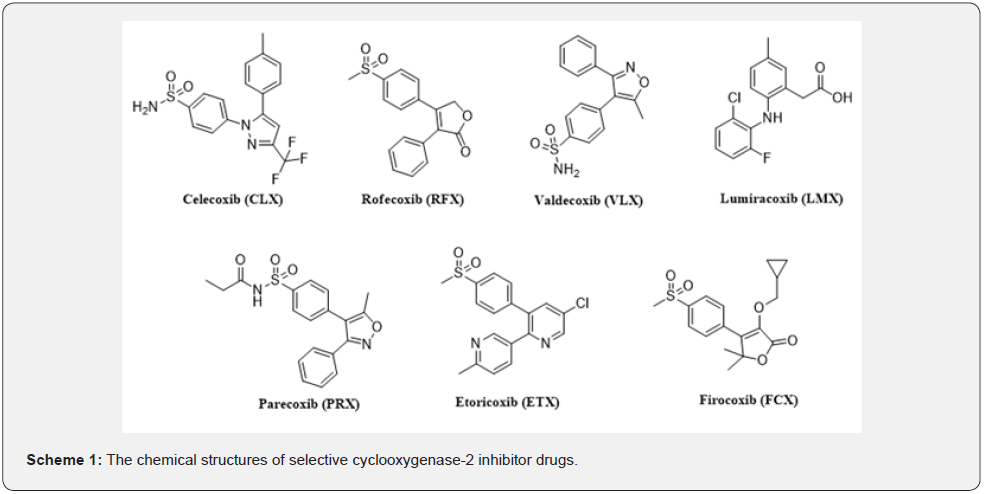
Material and Methods
The AutoDock 4.2 molecular docking program was used to find best binding interactions of selected selective cyclooxygenase-2 inhibitor drugs against SARS-CoV-2. The three-dimensional (3D) crystal structure of the protein Mpro was retrieved from Protein Data Bank (PDB) (PDB ID: 6LU7) [18]. The 3D structure of the drugs was downloaded from the PubChem (https://pubchem. ncbi.nlm.nih.gov/) in structure-data file format. The most suitable of the possible binding modes obtained as a result of the Molecular Docking processes were determined with Autodock 4.2, and their analyzes and visuals were obtained with the Biovia Discovery Studio Visualizer 2020 program [19-21]. In the present study, a selective cyclooxygenase-2 (COX-2) inhibitor drugs Celecoxib, Rofecoxib, Valdecoxib, Lumiracoxib, Parecoxib, Etoricoxib, and Firocoxib molecules were used for docking procedures. Also, Favipiravir, Chloroquine and Hydroxychloroquine were used as standard drugs for comparison.
Results and Discussion
The docking analysis result of the molecules and standards Celecoxib, Rofecoxib, Valdecoxib, Lumiracoxib, Parecoxib, Etoricoxib, Firocoxib, Favipiravir, Chloroquine and Hydroxychloroquine as inhibitors of SARS-CoV-2 (PDB: 6LU7) including binding energy, inhibition constant and important interactions at the active site are demonstrated in Table 1.
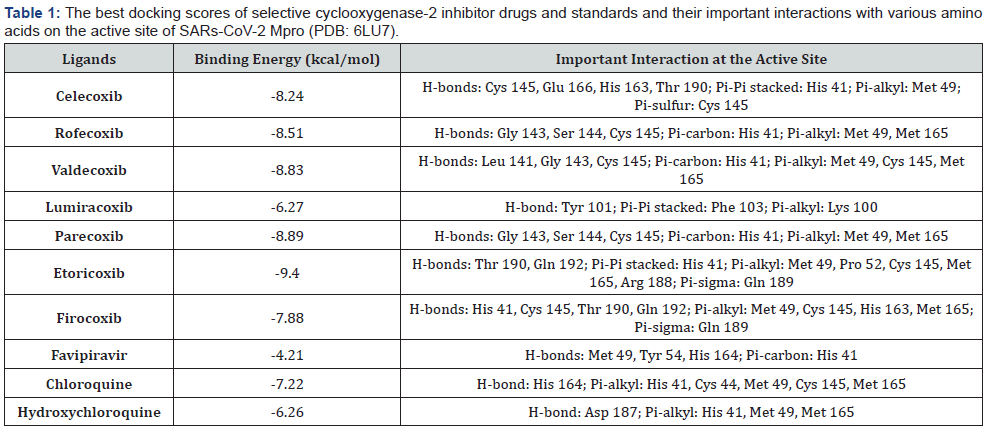

The protein-ligand interaction study revealed that the selective cyclooxygenase-2 inhibitor drugs are binding at the active site of SARS-CoV-2 Mpro protein with the best poses ranging from -6.27 to -9.40 kcal/mol (Table 1). In the current work, all the selective cyclooxygenase-2 inhibitor drugs showed better binding affinity then the standard drugs Favipiravir, Chloroquine, and Hydroxychloroquine (binding affinities of -4.21, -7.22, and -6.26 kcal/mol, respectively), except the Lumiracoxib. Among the best docking scores, only one drug (Lumiracoxib) has been shown the bind in a different region, then the rest of drugs (including standard drugs) and the binding energy of this drug was lowest (-6.27 kcal/ mol) among other drugs that were docked in this study (Figure 1). The best binding affinity (-9.40 kcal/mol) was observed with the drug of Etoricoxib, which have several important amino acid interactions, including hydrogen bonds with Thr 190 and Gln 192, and pi-alkyl interaction with Met 49, Pro 52, Cys, 145, Met 165, and Arg 188. The great binding affinities were also obtained with the drugs of Celecoxib, Rofecoxib, Valdecoxib, and Parecoxib, which had close binding energies to each other’s (-8.24, -8.51, -8.83, and -8.89 kcal/mol, respectively). The most important interactions with these compounds were with Cys 145 (hydrogen bonding), His 41 (pi-carbon bonding), Met 49 and Met 165 (Pialkyl interactions) (Figure 2). The moderate binding affinity was observed with the drug of Firocoxib (-7.88 kcal/mol). This drug also has some hydrogen bounds (His 41, Cys 145, Thr 190 and Gln 192), Pi-alkyl interactions (Met 49, Cys 145, His 163, and Met 165) and Pi-sigma interaction with Gln 189 (Figure 2).
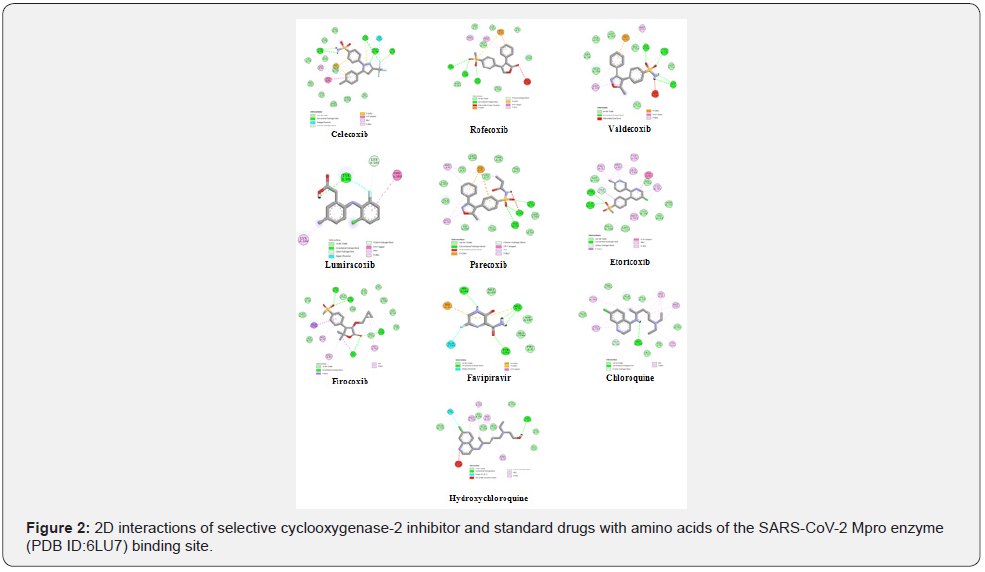
In general, all the docked selective cyclooxygenase-2 inhibitor drugs showed great binding affinities against SARS-CoV-2 Mpro enzyme by having important interactions on the active site. As shown in Figure 1, only one drug (Lumiracoxib) found to bind different binding site which is not favorable for high binding affinities. Some key amino acids are important on the active site for great binding affinities such as Gly 143, Cys 145, Thr 190, and Gln 192 for hydrogen bonding, His 41 (for Pi-carbon interactions), and Met 49 and Met 165 (for Pi-alkyl interactions).
Conclusion
In summary, we have performed molecular docking of the selective cyclooxygenase-2 inhibitor drugs (Celecoxib, Rofecoxib, Valdecoxib, Lumiracoxib, Parecoxib, Etoricoxib, and Firocoxib) with the important therapeutic target protein of SARS-CoV-2 and compared them with the standard drugs Favipiravir, Chloroquine and Hydroxychloroquine. The obtained dock scores demonstrated that all the selective cyclooxygenase-2 inhibitor drugs (except Lumiracoxib) showed a better binding affinity against SARSCoV- 2 Mpro enzyme than the standard drugs. More specifically, Etoricoxib (-9.40 kcal/mol) have shown the best binding affinity. This docking study indicates that these selective cyclooxygenase-2 inhibitor drugs might be useful lead molecules to discover potent and less toxic SARS-CoV-2 drugs in the near future.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgment
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
References
- Wang MY, Zhao R, Gao LJ, Gao XF, Wang DP, et al. (2020) SARS-CoV-2: Structure, Biology, and Structure-Based Therapeutics Development. Front Cell Infect Microbiol 10: 587269.
- Rothan H, Byrareddy SN (2020) The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun 109: 102433.
- Wanga F, Zheng S, Zheng C, Sun X (2020) Attaching clinical significance to COVID-19-associated diarrhea. Life Sci 260: 118312.
- Hoxha M (2020) What about COVID-19 and arachidonic acid pathway?. Eur J Clin Pharmacol 76(11): 1501-1504.
- Hu B, Guo H, Zhou P, Shi Z-L (2021) Characteristics of SARS- CoV-2 and COVID-19. Nat Rev Microbiol 19(3): 141-154.
- Chen N, Zhou M, Dong X, Qu J, Gong F, et al. (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395(10223): 507-513.
- Hopkins C, Surda P, Kumar N (2020) Presentation of new onset anosmia during the COVID-19 pandemic. Rhinology 58(3): 295-298.
- Kayaalp O (2018) Akılcıl Tedavi Yönünden Tıbbi Farmakoloji. Ankara: Pelikan Yayınevi.
- Korbecki J, Baranowska-Bosiacka I, Izabela Gutowska I, Chlubek D (2014) Cyclooxygenase pathways. Acta Biochim Pol 61(4): 639-649.
- Lasa M, Mahtani KR, Finch A, Brewer G, Saklatvala J, et al. (2000) Regulation of cyclooxygenase 2 mRNA stability by the mitogen-activated protein kinase p38 signaling cascade. Mol Cell Biol 20(12): 4265-4274.
- Kuwano T, Nakao S, Yamamoto H, Tsuneyoshi M, Yamamoto T, et al. (2004) Cyclooxygenase 2 is a key enzyme for inflammatory cytokine-induced angiogenesis. FASEB J 18(2): 300-310.
- Das UN (2020) Can Bioactive Lipids Inactivate Coronavirus (COVID-19)? Arch Med Res 51(3): 282-286.
- Das UN (2018) Arachidonic acid and other unsaturated fatty acids and some of their metabolites function as endogenous antimicrobial molecules: A review. J Adv Res 3(11): 57-66.
- Verrall GM (2020) Approach to Target the Host Response in Order to Try and Reduce the Numbers Presenting With Adult Respiratory Distress Syndrome Associated With COVID-19. Is There a Role for Statins and COX-2 Inhibitors in the Prevention and Early Treatment of the Disease? Front Immunol 2(11): 2167.
- Ong SWX, Tan WYT, Chan YH, Fong SW, Renia L, et al. (2020) Safety and potential efficacy of cyclooxygenase-2 inhibitors in coronavirus disease 2019. Clin Transl Immunology 9(7): e1159.
- Smeitink J, Jiang X, Pecheritsyna S, Renkema H, Maanen RV, et al. (2020) Hypothesis: mPGES-1-derived Prostaglandin E2, a so far missing link in COVID-19 pathophysiology?. Preprints. 2020040180.
- Pace S, Rossi A, Krauth V, Dehm F, Troisi F, et al. (2017) Sex differences in prostaglandin biosynthesis in neutrophils during acute inflammation. Sci. Rep 7(1):3759.
- Jin Z, Du X, Xu Y, Deng Y, Liu M, et al. (2020) Structure of M pro from SARS-CoV-2 and discovery of its inhibitors. Nature 582(7811): 289-293.
- Lolak N, Akocak S, Turkes C, Taslimi P, Isik M, et al. (2020) Synthesis, characterization, inhibition effects, and molecular docking studies as acetylcholinesterase, α-glycosidase, and carbonic anhydrase inhibitors of novel benzenesulfonamides incorporating 1,3,5-triazine structural motifs. Bioorg Chem 100: 103897.
- Isik M, Akocak S, Lolak N, Taslimi P, Turkes C, et al. (2020) Synthesis, characterization, biological evaluation, and in silico studies of novel 1,3-diaryltriazene-substituted sulfathiazole derivatives. Arch Pharm 353(9): 2000102.
- Koyuncu I, Durgun M, Yorulmaz N, Toprak S, Gonel A, et al. (2021) Molecular docking demonstration of the liquorice chemical molecules on the protease and ACE2 of COVID-19 virus. Current Enzyme Inhibition 17(2): 98-110.






























