Bilayer Floating Tablet: A Novel Pharmaceutical Approaches for Gastroretention
Deepika Joshi1*, Kamal Kaushik1,Bhavana Singh1, Nidhi Semwal1
1School of Pharmaceutical Sciences, SGRR University, Dehradun, 248001, India
Submission: September 1, 2021; Published: September 27, 2021
*Corresponding author: Deepika Joshi, Assistant Professor, School of Pharmaceutical Sciences, SGRRU, Dehradun, 248009, India
How to cite this article: Deepika J, Kamal K, Bhavana S, Nidhi S. Bilayer Floating Tablet: A Novel Pharmaceutical Approaches for Gastroretention. Nov Appro Drug Des Dev. 2021; 6(2): 555683. DOI: 10.19080/NAPDD.2021.06.555683
Abstract
Bilayer floating tablet is a gastro retentive novel delivery system developed for controlled release formulation. This review deals with study upon updated literatures on bilayer floating drug delivery system, its advantages and disadvantages, concept of gastroretention, rationale for drug selection, pharmacokinetic aspect, mechanism, approaches, list of polymers and other ingredients used, in-vitro and in-vivo evaluation, literature survey, marketed products. Incorporation of drugs in bilayer floating tablet remain in gastric region for several hours would significantly prolongs the gastric residence time of drug thereby improving bioavailability/solubility of drugs. This approach may be extensively used to improve therapy with several narrow absorption windows drugs.
Keywords: Gastric retention; Bilayer floating tablet; Stomach; Residence time; Gastric; Controlled drug delivery system
Introduction
Bilayer floating tablets
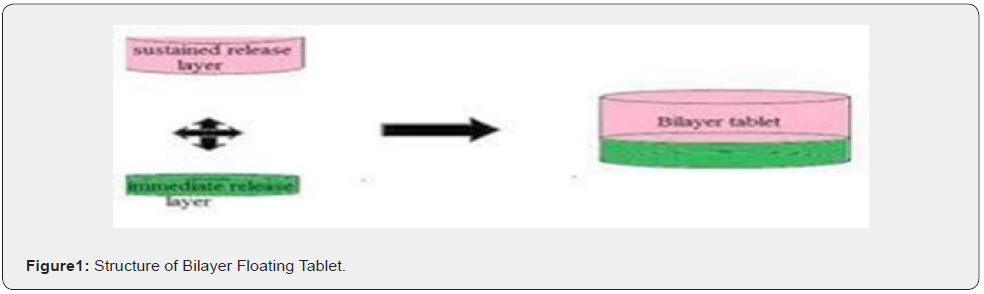
Bilayer tablet are multilayer tablets used in controlled drug delivery system. Bilayer floating tablets consisting of two layer i.e., immediate release layer which releases initial dose from system while the another sustained release layer absorbs gastric fluid (Figure 1), forming an impermeable colloidal gel barrier on its surface and maintain a bulk density of less than unity and thereby it remain buoyant in the stomach [1]. The immediate release layer is comprised of gas generating system i.e., sodium bicarbonate and citric acid control release layer comprised of low density release retardant polymers like HPMC K4M, K15M, E50LV. Bilayer floating tablets can be primary option to avoid chemical in compatibility between active pharmaceutical ingredients by physical separation and to unable the development of different drug relief profile [2,3].
Floating drug delivery system
Floating drug delivery system are low density system and having bulk density less than gastric fluid and remain they have sufficiently buoyancy to float over the gastric contents and remain buoyants in the stomach without affecting the gastric emptying rate for a longer period of time [4]. The increase in gastric retention time and a better control of the fluctuations in plasma drug concentrations can be seen. While the system is floating on the gastric content the drug is released slowly at the desire rate reliably buoyant on the surface of the meal. Many buoyant systems developed based on the granules, powder, capsules, tablets, laminated films and hallow microsphere. [5] Flotation of drug is achieve by in cooperating floating chamber filled with vaccum, air or inert –gas from the system. After release of drug, residual is emptying from the stomach.
GIT physiology
The main function of the stomach is to process and transport food.it serve as a short-term storage reservoir, allowing a rather large meal to be consumed quickly. The metabolism of enzymes is promoted in stomach of proteins. The peristalsis of stomach mixes up and grind consumed food with secretions of the stomach, turning food in liquid form. The liquefied bulk is transported to the small intestine for further digestion. [6] The human anatomy categorizes stomach (Figure 2) in three main parts: fundus, body and antrum (pylorus) [7].
Fundus: the upper part of the stomach, which forms a bulge above the level of the opening of the oesophagus (furthest from the pylorus).
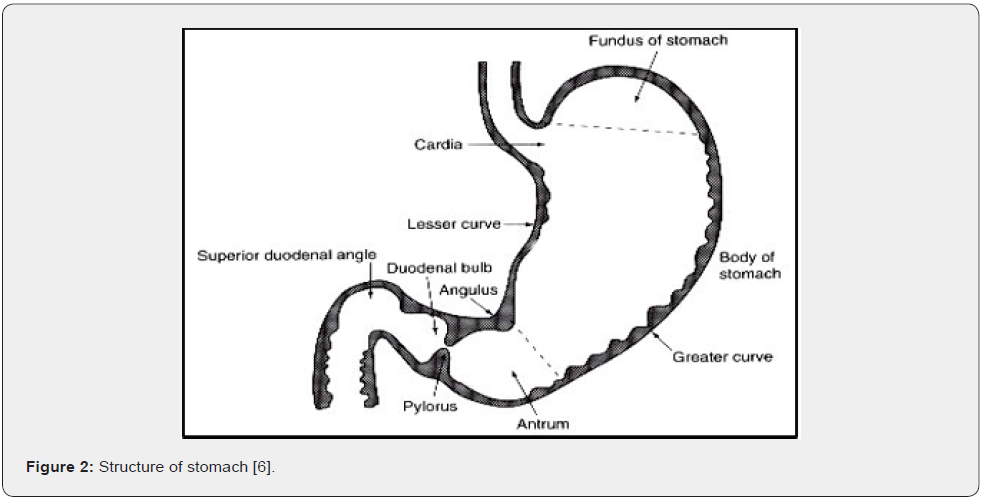
Body: below the fundus is the body, the main part of the stomach. The wider end of the funnel, the pyrolicantrum, connects to the body of the stomach.
Antrum: Provides for the main site for mixing motions and acts as gastric emptying pump by propeller actions. Both the fasting and fed states cause gastric emptying [8].
The proximal portion referred to as fundus and the body functions as storage for undigested food.
In this phenomenon, series of electric events takes pace in cycle via stomach and intestine every 2 to 3 hours. There occurs a phenomenon of inter digestive myloelectric cycle or migrating myloelectric cycle (MMC) and is divided into 4 phases:
Phase 1- Basal phase, last from 30 to 60 minutes with rate contractions and is characterized by a lack of secretory, electrical, and contractile activity.
Phase2- Preburst phase, lasts for 20 to 40min with intermittent contractions, during which contractile motions increase in frequency and size.
Phase 3- Burst phase, last for 10 to 20mins with intense and regular concentrations for shorts periods, termed housekeeper waves that sweep off undigested food.
Phase 4- Lasts from 0 to 5 minutes and is the transition periods between phases 3 and 1.
Upon food being ingested, the stomach motions vary fasted to fed state. It termed as digestive motility pattern and constituted regular peristalsis as in phases 2 of the states of fast. It reduces food size (<1mm) propelling food towards pylorus [9].
Factors affecting gastric retention
a) Density: Dosage form with lower density in the gastric content can float to the surface while high-density sink to the bottom of the stomach. Suitable density required for floating property is less than 1.0gm/cm3.
b) Nature of meal: High amount of fatty acid and other indigestible polymer slow down the gastric retention time (GRT ) due to variation in gastric motility.
c) Frequency of feed: Gastric retention time can rise by over 400minutes, when consecutive meals are given related with a single meal because of the low frequency of MMC.
d) Disease state: Gastric disease such as diabetes, chron’s disease, hypothyroidism, hyperthyroidism, duodenal ulcer etc fluctuates the GRT.
e) Size: Size should be more than 7.5mm in diameter to have improved GRT.
f) Shape: Either round or spherical shaped dosage from exhibit better property related to other shapes.
g) Single or multiple unit formulation: Multiple unit formulations show a more expectable release profile and insignificant damaging of performance because of failure of units, allow co-administrations of units that have dissimilar release profiles related with single unit dosage forms.
Advantage of bilayer floating tablets
a) Lighter and compact
b) Easiest and cheapest to package and strips
c) Potentials use of single entity feed granules.
d) Traditional delivery system.
e) Maintain physical and chemical stability [10].
f) They offer the most flexible dosage form.
g) Masking of bitter taste and bad odour by coating.
h) Swallowing of tablets is easy.
i) Maintains constant blood level.
j) Better suited for large-scale production.
k) Less cost other than other oral dosage form
l) Chemically, mechanically and microbiologically tablets are very stable.
Disadvantage of bilayer floating tablets
a) Difficult to swallow in case of children and unconscious patients.
b) Insufficient hardness, layer separation, reduced yield.
c) Individual layer weight control.
d) Cross contamination between the layers [11].
e) Capping is the major problem in bilayer tablets.
f) Increased fluid levels are required in the stomach so that the system floats properly.
g) Complex and bilayer rotary press are expensive.
h) Due to low density and amorphous nature of some drugs do not form because they resist compression.
i) Sometimes encapsulation or coating is required for the drugs that are oxygen sensitive, bitter tasting and bad odour.
Rationale for selecting drug candidates for bilayer floating tablets
a) Unstable at intestinal pH.
b) Drug has less bioavailability in gastric region.
c) Less dose of drug.
d) Less gastric retention time [14].
e) Narrow absorption window in GI tract ex-ribolavin and levodopa.
f) Basic absorbed from stomach and upper part of GIT ex: chlordiazoperoxide, cinnarazine.
g) Drug that disturbs normal colonic bacteria ex: amoxicillin Trihydrate.
Limitations of bilayer floating tablets
a) Lack of sufficient bonding and adhesion at the interface between the layer result in interfacial crack and layer separation.
b) Drug, which are irritants to gastric mucosa, are not desirable.
c) The drug, which undergoes first fast metabolism, is not desirable for the preparation of these system.
d) The drugs that are unstable in the acidic environment of the stomach is not suitable.
e) Drug which has stability and solubility problem in GIT is not suitable for this system [13].
f) If the layer is too soft or too hard, they will not bind properly with each other which can lead to separation of the layer.
g) Bilayer tablets don’t permit the termination of therapy.
h) It has less flexibility on adjusting the dose regiments [12].
Need of bilayer floating tablets
a) To control the delivery rate of either single or two different active pharmaceutical ingredients.
b) To modify the total surface area available API layer either by imposing with one or two inactive layers in order to achieve swellable /erodible for modified release.
c) T separate incompatible active pharmaceutical indigents from each other, to control the release of API from one layer by utilizing the functional property of other layer (such as osmotic property) [15].
d) For the administration of fixed dose combination of drug, prolong the product life cycle, buccal/mucoadhesive delivery systems, fabricate drug delivery system such a chewing device and floating tablets for gastroretentive drug delivery systems [16].
Pharmaceutical approaches for bilayer floating
Various technique for bilayer floating tablet
A. Osmotic-controlled release oral delivery system
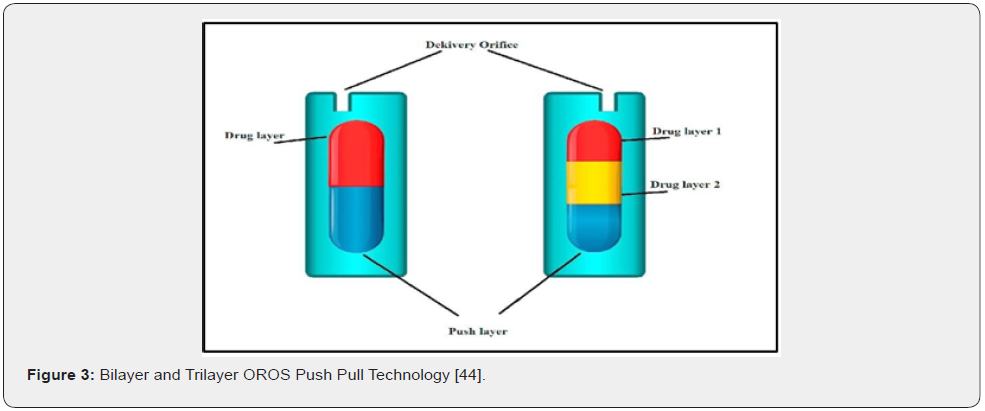
In this technology the system is comprised of two or three layers among which one or more layer is of drug and other layer consist of push layer. The drug layer consists of poorly soluble drug along with diluents having low molecular weight polymer, suspending agent and osmotic agents. The push layer is of higher molecular weight osmopolymer and an osmogents. A semipermeable membrane surrounds the tablet core and is interposed with an osmotic agent that swells in presence of water. Laser is used to drill a tiny hole through the membrane. In the stomach, water passes through the membrane into the pill, causing the osmotic material to swell, that pushes the drug out of the whole. The drug is delivered to the body at a constant rate. The product manufactured using this technology is Glucotrol XI and Procardia XL.
B. Elan drug technology
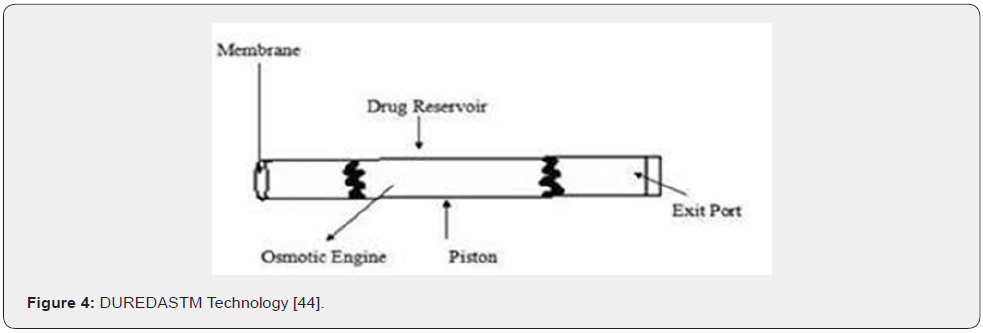
DUREDASTM or Dual Release Drug Absorption System using by layer tableting technology, which has been developed to provide two different release rates from a single dosage form. In this the tablet are prepared by two separate direct compression steps that combined and immediate release granulate and a controlled-release hydrophilic matrix complex with in one tablet. The immediate release layer, release the drug immediately after going into the GIT and the controlled release matrix remain intact and slowly absorbs fluid from the GI tract, which is return causes the matrix to expand and transform the hydrophilic polymer into a porous, discuss gel that’s a serves as a barriers between the drug and fluid and the gel continue to expand, the fluid penetrates into the dosage form, dissolving the drug and allowing the solution to diffuse out [17]. The immediate release and controlled release combination of two drugs are feasible.
C. PRODAS or programmable oral drug absorption system
It is a multi-particulate drug delivery technology based on the encapsulation of controlled release mini tablets in the size of 1.5 to 4mm in diameter this technology can be used to pre programme the release rate of a drug.it is possible to incorporate different mini tablet at difference sites within the GIT. This represents a combination of multi-particulate and hydrophilic matrix tablet technology and provide the benefit of both system in one dosage form. Such combination includes immediate release, delayed release, and controlled release minitablet.
D. Rotab bilayer
RoTab bilayer when using is switched to production mode. Dose and compression force is automatically regulated by adjusting filling speed and die table. Hardness is also regulated when its required [18].

Polymer and other excipients used for formulation of bilayer floating tablets
The polymer used for coating or fabrication of bilayer tablets are hydroxypropyl methyl cellulose (HPMC 1000, HPMC 4000, HPMC K15, HPMC K4,); beta cyclodextrin, sodium alginate, hydroxypropyl cellulose (HPC-H, HPC-M), Eudagrit S, Metolose S.M.100, polyvinylpyrrolidone (PVP), acrylic polymer and Carbopol.
Inert fatty materials: Beewax, Fatty acids, long chain fatty alcohols, gelucires 39/01 and 43/01.
Effervescent agents: Sodium bicarbonate, Citric acid, Tartaric acid, Di-SGC (Di-sodium Glycine Carbonate, CG).
a) Release rate accelerants (5%-60%) Eg: lactose, mannitol.
b) Release rate retardants (5%-60%) Eg: Dicalcium Phosphate, Talc, magnesium stearate.
c) Buoyancy increasing agents (upto 80%) Eg: Ethyl cellulose.
d) Low density material: Polypripylene foam powder (Accurel) MP1000.
Pharmacokinetic aspect of bilayer floating tablets
a) Absorption window
He candidates for GRDDS are molecules that have poor colonic absorption but are characterized by better absorption properties at upper part of GIT.
b) Enhance bioavailability
The compound having narrow absorption window having the possibility of continuous administration of the compound at specific site.
c) Enhance first pass biotransformation
The pre-systemic metabolism of the tested compound is increased. When the drug is presented to metabolic enzyme (cytochrome p-450) in a sustained manner.
d) Improve bioavailability due to reduced p-glycoprotein activity in 16he duodenum
The drug that P-gp substrate do not undergoes oxidative metabolism GRDDS may elevate absorption compaired to immediate and CR dosage form.
e) Reduce frequency of dosing
For drugs with relatively short biological half-life. Sustained and slow input from GRDDS results flip-flop pharmacokinetic and enable reduced dosing frequency.
f) Targeted therapy for local elements in upper GIT tract
The prolonged and sustained administration of the drug from GRDDS to the stomach may produce local therapy in the stomach and small intestine.
Pharmacodynamic aspect of bilayer floating tablet
a) Reduce fluctuation of drug concentration.
b) Are associated with peak concentration can be prevented. Improved selectively in receptor activation.
c) Reduce counter activity of the body.
d) Slow input of drug into the body was shown to minimize the counter activity leading to higher drug efficiency.
e) Minimize adverse activity of colon- The pharmacodynamics aspect provides the rationale for GRDDS formulation for beta-lactumantibiotics that are only absorbed from the small intestine and due to presence at colon it develop of microorganism is resistance.
Evaluation techniques of bilayer floating tablets
Vitro evaluation: In this technique the evaluation was done to access the physiochemical properties and release characteristic of the formulation.
A. Pre-compression parameter
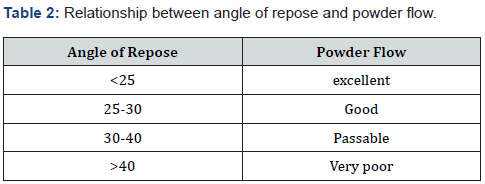
i. Angle of repose: With the help of angle of repose, we can estimate the frictional force in powder. It can be defined as the maximum angle formed between surface pile of powder and horizontal plane i.e., height. We can find out the angle of repose by using the formula.
tanθ= h/r
θ= tan-1h/r
Where, θ=angle of repose h=height of pile
r= radius of pile29.
The relationship between angle of repose and powder flow is follows in table:
ii. Compressibility index: The propensity of the powder to be compressed is estimated by compressibility index and it also help in measurement of settling and interparticulate interaction
Compressibility index (%) = pt-p0*100/pt
Where pt = trapped density gram/ml
Po= Bulk density gram/ml
iii. Bulk density
It is defined as the ratio of mass of powder to bulk volume. in this 50cm3 of powder is passed through sieve no.20 than this introduces in 100ml graduated cylinder [19].
iv. Tapped density
The increase in bulk density attained after mechanical tapping in measuring cylinder is called tapped density [21].
Tapped density= weight of powder taken/tapped volume
v. Hausner ratio
The hausner ratio is the no. that is related to flowability of powder material. Hausner ratio = tapped density/bulk density
Hausner ratio = Vo/Vf
Where, V0= unsettled apparent volume Vf = Final tapped volume.
vi. Particle size distribution
Sieving method is used to determine particle size distribution.
B. Post-compression parameters
i. Tablets thickness: Three tablets are randomly taken and then their thickness and diameter are measured by vernier caliper or by using calibrated screw gauze.
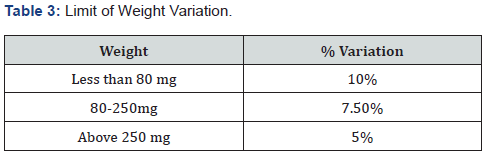
ii. Weight variation test: Twenty tablets are selected and weight individually. Then the average weight and standard deviation is calculated. Test passes when not more than two tablets deviate from average weight. In all formulation, the tablet weight was more than 324mg, hence 5% maximum difference allowed [20].
iii. Hardness: Hardness indicates the ability of a tablet to withstand mechanical shock while handling [22]. It is expressed in kg/cm2 and checked using Monsanto hardness tester by randomly picking three tablets [22]. In all the cases, mean of five replicate determinations were taken.
iv. Friability test: This was determined by weighing 26 tablets after dusting, placing them in the roche friabilator and rotating the plastic cylinder vertically at 25 rpm for 4 min [23]. After dusting, the total left weight of the tablet was recorded, and percent friability was calculated by formula:
% Friability= intial wt of tablets- final wt of tablets/initial weight of tablet × 100
Acceptance limit: % Friability of tablets less than 1 % are considered acceptable [24].
v. Disintegration time: One tablet is placed in disintegration apparatus containing buffer 0.1N HCl or PBS pH 6.8 and test is carried out at 37 °C [25].
vi. Dissolution studies: It is performed using USP paddle apparatus at 50 rpm rotational speed maintained at 37°C. At different time interval 5ml samples is withdrawn and is replaced with same amount of buffer [26].
vii. Floating lag time: It is basically the time interval of tablet to starting floating. It should be less than 1min. It is measured by the dissolution test apparatus containing 0.1N HCl (900ml).
viii. Floating time: It is the total time taken by tablets that remains floating in the media [27].
ix. Drug content uniformity: 10 tablets are taken and powdered equivalent weight of drug dose is taken and is transferred to volumetric flask then buffer is added, and absorbance is determined using UV spectrophotometer [28].
C. In vivo evaluation of bilayer floating tablets:
i. Radiology: X-ray is widely used for examination of internal body systems. Barium sulphate is widely used Radio Opaque Marker. So, BaSO4 is incorporated inside dosage form and x-ray images are taken at various intervals to view gastric retention.
ii. Scintigraphy: It is similar to x-ray, emitting materials are incorporated inter dosage form and then images are taken by scintigraphy. A widely used emitting material is 99Tc.
iii. Gastroscopy: Gastroscopy is peroral endoscopy used with fiber optic or video systems. Majorly gastroscopy is used to inspect visually the effect of prolongation in stomach. It can also provide the detail information of evaluation of GRDDS.
iv. Magnetic marker monitoring: In this technique, dosage form is magnetically marked with incorporating iron powder inside, and image is taken by very sensitive bio-magnetic measurement equipment.
v. Ultrasonography: It is not used generally because it is not traceable at intestine.
vi. C13 octanoic acid breath test: C13 Octanoic acid is absorbed into GRDDS where it undergoes chemical reaction and liberates CO2 gas. The important carbon atom which will come in CO2 is replaced with C13 isotope. So, time up to which C1302 gas is observed in breath can be considered as gastric retention time of dosage form. As the dosage form moves to intestine, there is no reaction and no CO2 release. So, this method is cheaper than other [29].
Application of bilayer floating tablet
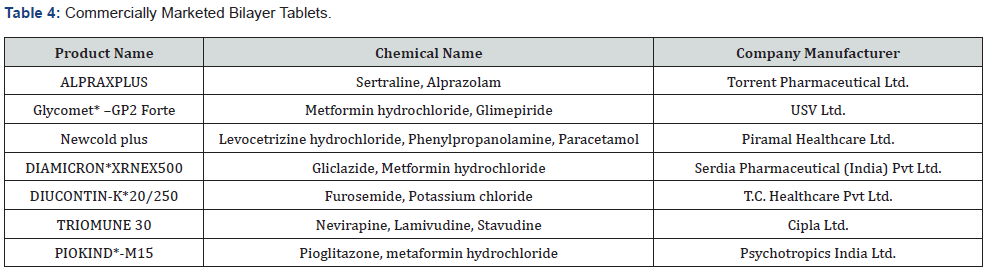
Bilayer tablets are suitable for the sequential release of two drugs to be given combined. It separates the two mismatching drugs. The sustained-released tablets whose one layer provides instant drug release as the initial loading dose while the second layer is containing the sustained dose. Bilayer tablets are latest technology that helps in overcoming the limitations of a singlelayered tablet. Bilayer tablets help in the combined delivery of two different drugs that have different release profiles. Bilayer tablets are utilized to administer fix dosage containing different APIs. They are employed to increase and modify the surface area for active pharmaceutical ingredients by erodible barriers for custom release [30,31].
A). Herbal bilayer floating tablets
Bilayer floating tablet is the best option for herbal drug delivery. It could release drug upto 12-24 hours. It improves the therapeutic effect of drug.
Some of the herbal drugs that can be delivered as bilayer floating tablets are:
i. Forskolin: It is used as anti-obesity agent reducing fat in body muscles. It may enhance fat loss without loss of muscle mass.
ii. Black myrobalan: It shows uniform anti-bacterial activity against ten clinical strains of H.pylori.
iii. Ginger root: It is used for the treatment of gastrointestinal ailments such as motion sickness, dyspepsia and hyperemesis gravidarum and it also have chemopreventative activity in animal models.
iv. Turmeric: It prevents gastric and colon cancers in rodents.
v. Berberine: It shows variety of activity against bacteria, viruses, fungi, protozoans, and helminthes [32].
B). Treatment of diseases
i. Hypertension and angina pectoris
Nifedipine is a calcium channel blocker of the di-hydropyridine type which is mainly used for the treatment of hypertension and angina pectoris. Nifedipine is a suitable candidate for CR administration due to its short elimination half-life of 2-hrs, its rapid and complete drug absorption over the entire gastrointestinal tract, despite its low water solubility and the relationship between drug plasma concentration and blood pressure reduction [32,33]. Controlled release formulation of nifedipine would be effective in overcoming the dissolution limitation by slowly supplying the drug from the intact matrix base during its sojoum in the gastrointestinal tract and its thus expected to decrease side effect and improve patient compliance. Control release tablet for oral administration designed to deliver the drug at gastric region for treatment of hypertension [34].
ii. Cardiovascular disease: Atorvastatin immediate release and aspirin pulsatile release for the treatment of cardiovascular diseases. Four formulations were prepared for immediate release layer of atorvastatin using different concentrations of microcrystalline cellulose and talc by different compression method [35-37].
iii. Peptic ulcer: The major target of bilayer tablet is to decrease the pain and promotes ulcer healing, prevention of complications/relapse.
C). Targeted drug delivery system
It is the method of delivering medications to the patients in the manner that increases the concentration of medication in some part of body related to other. The aim of targeted drug delivery system is to prolong, localize, target and have protected drug interaction with the diseased tissue. It reduces the frequency of dosage taken by patient. There are different types of drug delivery route such as polymeric micelles, liposomes, lipoproteinbased drug carrier, nano-particle drug carrier, dendrimers, etc. It is also used to treat cardiovascular disease and diabetes. The most important application is to treat cancerous tumours [38-40].
D). Controlled drug delivery system
It aims at releasing the dose of therapeutic directly in the desired zone during the required period of time. It allows the maximizing the efficacy of the therapeutic and minimizing e side effects. For floating drug delivery system, the polymers used must be highly swellable in shortest time [41]. The control release matrix tablet containing uniform mixture of drug, polymer and excipients including gas–generating agents. Nifedipine was mixed using variable amount of Carbopol p 934 and HPMC (K4M, K15M) properly in a mortar with weighed number of excipients. The well will mix powder was compressed by direct compression technique and used as controlled release layer [42,43].
Conclusion
Drug release is the major area in the pharmaceutical research work. Through floating bilayer tablets both type of release i.e., sustained as well as immediate release can be obtained, and sustained release can be increased up to 24 hours. It is also beneficial in providing gastric retention thereby increasing gastric emptying time as well as increasing bioavailability. Another advantage is that two drugs can be administrated concurrently at the same time, which provides better patient compliance. Drugs with narrow absorption window such as antiviral, antibiotic and antifungal stand out as better candidate for floating bilayer dosage form. In the last decade, interest in developing a combination of two or more active pharmaceutical ingredients (API) in a single dosage form (bilayer tablet) have increased in the pharmaceutical industry, promoting patient convenience and compliance. Due to the use of different materials and complex geometric boundaries between adjacent layers, the mechanical structures of this drug delivery system have become quite intricate, requiring complicated tablet architectures as well as patient friendly. Gastro retentive drug delivery system prolongs the retention time of dosage forms in the stomach or upper gastro-intestinal tract, as to improve solubility, bioavailability and the therapeutic efficacy of the drugs. Several pharmaceutical companies are currently developing bilayer tablets. For a variety of reasons: patients’ extension, therapeutic marketing to name a few.
Bilayer tablet is improved beneficial technology to overcome the shortcoming of single layered tablet. Bilayer tablets provide one of the important design approaches where incompatible drugs, with different indication, and same drug with different release rate can be incorporated in a single unit. Bilayer tablet is suitable for sequential release of one or two drugs in combination, separate two incompatible substances and also for sustained release tablet in which one layer is immediate release as initial dose and second layer is maintenance dose. The preparation of tablets in the form of bilayer is used to provide systems for the administration of drugs, which are incompatible and provide controlled release tablet preparations by providing surroundings or multiple swelling layers.
Acknowledgment
All authors have contributed significantly in the preparation of the manuscript and are in agreement with the content of the manuscript and agree to submission to Novel Approaches in Drug Designing & Development.The authors express gratitude to School of Pharmaceutical Sciences, SGRR University, Dehradun.
References
- Biswal B (2011) Design development and evaluation of trimetazidinedihydrochloride floating bilayer MR tablets. IRJP 2(7): 92-97.
- Lende LK, Banarjee SK, Gandhave MV, Gaikwad DD, AJ Gaykar (2013) Review on: Bilayer floating tablet. AJPRD 1(1): 31-39.
- Pethkar SV, Suggala V, Gholap S, Salunke R, et al. (2016) Review on Bilayer floating tablet. WJPPR 5(2): 1411-1423.
- Vyas SP, Khar RK (2006) Essentials of Controlled Drug Delivery in: S. P. Vyas. Editors. Controlled Drug Delivery – Concepts and Advances. Vallabh Prakashan, Delhi, pp. 1- 53.
- Sharma AR, Khan A (2014) Gastroretaintive drug delivery system: An approach to enhance gastric retenti on for prolonged drug release. IJPSR 5(4): 1095-1106.
- Bhowmik D, Gautam D (2018) Gastro Retentive Drug Delivery Systems-a Novel Approaches of Control Drug Delivery Systems. Research Journal of Science and Technology 10(2): 1-13.
- Pandey A, Kumar G, Kothiyal P, et al. (2012) A Review on current approaches in gastro retentive drug delivery system. Asian Journal of Pharmacy and Medical Science 2(7): 60-77.
- Makwana A, Sameja K, Parekh H, Pandya Y (2012) Advancements in controlled release gastroretentive drug delivery system. A review. Journal of Drug Delivery and Therapeutics 2: 12-21.
- Nayak KP, Upadhyay P (2012) Gastroretentive drug delivery systems and recent approaches. A review. Journal of Pharmaceutical Research and Opinion 2: 1-8.
- Deshpande AA, Shah NH, Rhodes CT, Malick W (1997) Development of A Novel Controlled Release System for Gastric Retention. Pharm Res 14(6): 815-819.
- Sharma SK, Mohan S, Jaimin Mi, Chauhan BS, Chatterjee A (2014) Formulation and In-Vitro Evaluation of Bilayer Tablets containing Pioglitazone HCl and Gliclazide for Type II Diabetes. Int J Pharm Tech Res 6(2): 607-622.
- Panchal AH (2013) A Novel Approache of Bilayer Tablet: A Review. International Research Journal of Pharmacy 3(5): 44-59.
- Gosh R, Dollery (1999) Therapeutics Drugs, Churchill Livingstone, New York 6: 38-43.
- Aulton ME (2002) Bilayer Tablet in pharmaceutics, the sciences of dosage from design. (2nd edn), Churchill Livingstone 2(6): 414-418.
- Garg RK (2015) Bilayer floating tablet Technology: An overview. IJPPR 3(3): 302-322.
- Kulkarni A, Bhatia M (2009) Development and evaluation of bilayer floating tablets of atenolol and lovastatin for biphasic release profile. Iranian Journal of Pharmaceutical Research 8(1): 15-25.
- Panchal AH (2013) A Novel Approach of Bilayer Tablet: A Review, International Research Journal of Pharmacy 3(5): 44-59.
- Parnapalli Mand, Arshad BK (2012) Recent Apporaches in Bilayered Technology: Review. International Journal of Pharmaceutical Science and Research 3: 4681-4688.
- Desai S, Bolton S (1993) A Floating Controlled Release System: In-Vitro – In-Vivo Pharm Res 10: 1321-1325.
- Indian Pharmacopoeia (1996) (2nd edn), Published by the Controller Publication, Delhi 1996: 736.
- The United States Pharmacopeial Convention (2012) Stage 6 Harmonization Official December 1: 616.
- Gahiwade HP, Patil MV, Thakare BW, Vinod MT, et al. (2012) Formulation and In-vitro evaluation of Trifluperazine HCl bilayer floating tablets. International Journal of Pharma and Bio Science 2: 166-172.
- The United States Pharmacopoeia (2000) United states Pharmacopoeial convention, Inc., Rockville, MD, pp. 19-44.
- Chaudhari S, Bawaskar M, Shirsat A (2012) Formulation and Evaluation of bilayer Floating tablets of Carvedilol Phosphate. Journal of Drug Delivery and Therapeutics 2: 9-19.
- Chandira RM, Palanisamy P, Jayakar B (2012) Formulation and Evaluation of Bilayered Floating tablets of Metformin Hydrochloride. International Research Journal of Pharmacy 3: 257-266.
- Narendra C, Srinath MS, Ganesh B (2006) Optimization of Bilayer Floating Tablet containing Metoprolol Tartrate as a Model Drug for Gastric retention. AAPS Pharma Sci Tech 7: 1-17.
- Lokendra PS, Rakesh KS, Deepak GU, Vijay KC, et al. (2011) Floating effervescent tablet: A Review”. Journal of Pharmaceutical and Biomedical Sciences 5: 345-357.
- Dinakaran SK (2011) Formulation and evaluation of bi-layer floating tablets of ziprasidone HCl and trihexyphenidyl HCl. Brazilian Journal of Pharmaceutical Sciences 4: 545-555.
- Patel N, Nagesh C, Chandrashekhar S, Patel J, Jani D (2012) Floating drug delivery systems: An innovative acceptable approach in Gastro retentive drug delivery. Asian J Pharm Res 2: 07-18.
- Chawla G, Gupta AP, Bansal K (2004) Gastroretentive Drug Delivery System. In: Progress in Controlled and Novel Drug Delivery System, NK Jain First Edition, pp. 76-97.
- Rao SV, B Priyanka, K Padmalatha (2019) Bilayer tablet technology: a novel approach, GSC Bio Pharm Sciences 7(2): 022-028.
- Desai S, Bolton S (1993) A Floating Controlled Release System: In-Vitro – In-Vivo Pharm Res 10: 1321-1325.
- Parnapalli M, Arshad BK (2012) Recent Apporaches in Bilayered Technology: Review. International Journal of Pharmaceutical Science and Research 3: 4681-4688.
- Patwekar SL, Baramade MK (2012) Controlled Release Approach to Novel Multiparticulate. Drug Delivery System International Journal of Pharmacy and Pharmaceutical Sciences 4: 757-763.
- Ahmed SI, Mangamoori LN, Rao YM (2010) Formulation and characterization of matrix and triple-layer matrix tablets for oral controlled drug delivery. International Journal of Pharmaceutical Sciences 2(7): 137-143.
- Panchal AH (2013) A Novel Approach of Bilayer Tablet: A Review. International Research Journal of Pharmacy 3(5): 44-59.
- Agyilirah GA, Green M, duCret R, Banker GS (1991) Evaluation of the gastric retention properties of a cross linked polymer coated tablet versus those of a non‐disintegrating tablets. Int J Pharm 75: 241‐247.
- Shrikant M, Shah S, Upadhay P (2012) Floating bilayer drug delivery systems- An unconventional approach in conventional form. Am J Pharm Tech Res 2(2): 609-628.
- Pattanayak DP, Dinda SC (2011) Bilayer tablet formulation of Metformin HCl and Glimepiride: A novel approach to improve therapeutic efficacy. Int J Drug Discovery Herb 1(1): 1-4.
- Gahiwade HP, Patil MV, Thakare BW, Vinod MT, et al. (2012) Formulation and In-vitro evaluation of Trifluperazine HCl bilayer floating tablets. International Journal of Pharma and BioScience 2: 166-172.
- Shukla S, PandyaV, Bhardia P, Jonwal N, Bhatt D (2013) Bi-layer Tablet system – An Innovative trend. Asian J Pharm Res 3(2): 49-56.
- Parnapalli M, Arshad BK (2012) Recent Apporaches in Bilayered Technology: Review. International Journal of Pharmaceutical Science and Research 3: 4681-4688.
- Karudumpala S, Chetty M, Gnanaprakash K, Venkatesh B, Sankar P (2013) A review on bilayer floating tablets. Int J Res Pharm Sci 4(2): 354-360.






























