Pandemics and Peptide Vaccines
Ashesh Nandy*
Centre for Interdisciplinary Research and Education, Kolkata, India
Submission:May 18, 2021; Published: June 01, 2021
*Corresponding author: Ashesh Nandy, Centre for Interdisciplinary Research and Education, Kolkata, India. Email: anandy43@yahoo.com
How to cite this article: Nandy A. Pandemics and Peptide Vaccines. Nov Appro Drug Des Dev. 2021; 6(1): 555676. DOI: 10.19080/NAPDD.2021.06.555676
Keywords: Covid-19; Vaccines; Drugs; mRNA; Peptide; Immune system
Opinion
The covid-19 pandemic has galvanized the world research community into a single-minded motivation to design and develop a vaccine against it. This has led to the unprecedented result in myriad vaccines being offered to the world within a year’s time, where normally it takes several years and billions of dollars to market a viable vaccine. The alacrity with which nations have embraced these vaccines generally after Phase III trials from the emergency use clause in drug regulation protocols expedited the process.
Traditionally vaccines have been developed since the beginning by using inactivated, or attenuated viruses and assumed that if the vaccine succeeds against the target group it will work for all communities – a “one size fits all” concept. The results, to no surprise, differ from communities to communities, but generally have had spectacular successes like smallpox eradication and near eradication of poliomyelitis. Another problem is that viruses, especially RNA viruses mutate very rapidly rendering drugs and vaccines obsolete in short time; in the case of coronavirus SARS-CoVID-2, which is three times the average RNA virus length, the mutations per generation are even greater in number. Hence, we have the USA variant (D614G) of the covid-19, the UK variant (B.1.1.7), the South African variant (B.1.351), the Indian variant (B.1.1.617) among others originating within the past year [1]; fortunately, the main vaccines still seem to offer good protection against these variants so far.
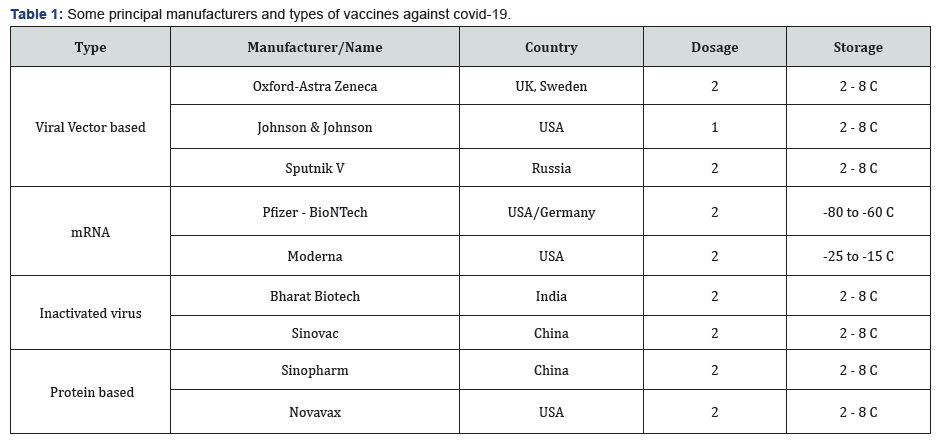
However, recent advances in genetics, immune informatics, bioinformatics, information technology and allied sciences have given us insights into how vaccines work, ushering in a new paradigm in vaccinology where the focus is on design and development of the silver bullet of vaccines that target precisely where antibodies, the body’s immune system, are generated. The new vaccines against covid-19 reflect this view (Table 1). All the new vaccines target the spike glycoprotein of the virus as the principal agent to enable the whole virus to enter the host cell. The principle of the vector virus vaccines is using adenovirus engineered to carry the genetic information like a Trojan horse to get the host cell to make the spike protein and thereby generate antibodies against it; the Oxford-Astra Zeneca vaccine, Sputnik V and Janssen (Johnson & Johnson) vaccines are of this type. On the other hand, Pfizer and Moderna use, for the first time, a new mRNA technology to create the spike glycoprotein in the host cell, only that it doesn’t carry any live virus. The two Chinese vaccines, Sinovac and Sinopharm, and the Indian COVAXIN vaccine are throwbacks to the more traditional inactivated viruses to boost the body’s immune system.
Vaccination has yielded good results for the developed nations, but the economic demands militate against the less developed and underdeveloped nations. Given the rate of production and the supply-demand equation for as infectious and virulent disease as covid-19, it may take several years to cover them all. In the meantime, more variants will keep cropping up and it is likely that some variants will be resistant to the structure of the spike protein on which the current crop of vaccines are based. Which means back to the drawing board again, this time with commercially less viable terms and all that implies.
Clearly a new more manageable scenario is called for. One of the approaches that has not been addressed adequately so far is peptide vaccines. One of the earliest successes of peptide vaccines was that against canine parvovirus and subsequently other veterinary applications, but no license has been given for use against human viruses, although the NIH ClinicalTrials.gov regularly list 500+ trials of various vaccines, mostly against cancers [2]. Significant success has been seen in addressing several types of human cancer tumours [3] and it is believed that that is where the first licenses will be received but as of now that is a distant dream in spite of many benefits of peptide vaccines such as (a) being a chemical can be rather easily and rapidly manufactured; (b) can be scaled up as demand grows more readily than biological vaccines; (c) being factory assembled, can be modified easily as required; (d) being a chemical, purity can be assured and there is no question of biological contamination; and (e) storage is not an issue [4]. Overall, the costs for peptide vaccines are significantly less compared to conventional vaccine development and deployment strategies.
These advantages make peptide vaccines a ready alternative for epidemic and pandemic situations. Rational design of peptide vaccines calls for peptides that can target segments of the spike protein that are sufficiently surface exposed and have good epitope potential while being as conserved as possible against mutational changes [5]. This calls for good computational approach like graphical representations and numerical characterization using which one can rapidly scan hundreds and thousands of protein sequences and determine segments that remain most conserved [2], then check them for surface exposure and epitope potential and subject the selected regions to check against autoimmune threats [6]. Only those that pass these tests can be considered for vaccine development. In a beginning pandemic situation even a few reasonably accurate sequences can be a starting point with more sequences and variants added as they become accessible.
The search for peptide vaccine targets needs to be improved in several ways [2]: Depending on the model used the demands on computing resources can be quite high, the quantity and quality of sequence data need to be of high standard, peptide folding has to be guarded against and cautions in storage need to be ascertained. These and other issues need to be resolved before the call can be given for peptide vaccine licensure.
But given an eventual go-ahead for peptide vaccines, there are several advantages that one can plug into. A peptide vaccine can be clubbed with other peptide vaccines into a multivalent dose. The vaccine being a synthetic peptide, one can make small changes as required to best fit the HLA profile of a community and tailor the vaccine to suit the circumstances. This has the advantage of maximizing the effect of a vaccine designed and developed for a particular area to wider reach. If this can be institutionalized one can envisage a possible scenario where a series of vaccine manufacturing facilities are set up at vantage points across the globe and a prophylactic for a particular pandemic is designed for a particular region at some primary labs, then distributed amongst these facilities to alter where possible the peptides to maximize the effect on the local population. To carry the implications a little further, such post-development adjustment can also be done for individuals according to their individual HLA profile and thus create personalized medicines [7].
Thus, rational design of peptide vaccines carries the seeds for future growth and campaign against epidemic and pandemic viruses. These are surely to come, perhaps with increasing frequency, and the sooner we overcome the initial hurdles, the more secure it will be for mankind.
References
- https://www.gavi.org/vaccineswork/results-novavax-vaccine-trials-uk-and-south-africa-differ-why-and-does-it-matter (Accessed May 16, 2021)
- Nandy A, Basak SC (2016) A Brief Review of Computer-Assisted Approaches to Rational Design of Peptide Vaccines. Int J Mol Sci 17(5): 666.
- Maeng HM, Berzofsky JA (2019) Strategies for developing and optimizing cancer vaccines. F1000Research 8: 654.
- Moisa AA, Kolesanova EF (2012) Synthetic Peptide Vaccines. In: Priti R, editor. Insight and Control of Infectious Disease in Global Scenario. London (UK): In Tech. pp. 201-28. ISBN: 978-953-51-0319.
- Skwarczynski M, Toth I (2016) Peptide-based synthetic vaccines. Chem Sci 7(2): 842-54.
- Oany AR, Emran AA, Jyoti TP (2014) Design of an epitope-based peptide vaccine against spike protein of human coronavirus: An in-silico approach. Drug Des Dev Ther 8: 1139-1149.
- Nandy A, Basak SC (2019) Towards Personalized Vaccines-Tailoring Peptide Vaccines to Demographic Groups and Individuals. Med One 4: e190026.






























