Perspectives of β-glucans for Health Promotion
Yitong Li, Tong Li and Rui Hai Liu*
Department of Food Science, Cornell University, Ithaca, New York, USA
Submission:October 01, 2020;Published: October 21, 2020
*Corresponding author: Rui Hai Liu, Department of Food Science, 245 Stocking Hall, Cornell University, Ithaca, NY, USA
How to cite this article: Yitong L, Tong L, Rui Hai L. Perspectives of β-glucans for Health Promotion. Nov Appro Drug Des Dev. 2020; 5(3): 555665. DOI: 10.19080/NAPDD.2020.05.555665
Abstract
Dietary consumption of whole grains has been linked to reducing the risks of developing chronic diseases including diabetes, obesity, cardiovascular disease, and some cancers. One of the bioactive compounds of whole grains, β-glucan, has been proposed to be responsible for their health benefits. β-glucan is a non-digestible and non-starch polysaccharide, widely distributed in grains, bacteria, yeast, and fungi. This article will review current research about β-glucans originated from different sources and their health benefits, especially on innate immune modulation, metabolic syndrome, and gut health, and focus on mechanisms of action of soluble and insoluble β-glucans on their biological functions.
Keywords: β-glucan; Whole grain; Health benefits; Immune system; Metabolic syndrome; Gut health
Abbreviations: MS: Metabolic syndrome; HFD: High-fat Diet; LDL: Low-density lipoprotein; HDL: High-density lipoprotein; SCFAs: Short-chain fatty acids
Introduction
Dietary consumption of whole grains has been linked to reducing the risks of developing chronic diseases including diabetes [1], obesity [2], cardiovascular disease [3], and some cancers [4]. Whole grains include three major parts: the bran as the outer layer rich in fiber and phytochemicals; the germ which contains lipid soluble vitamins and minerals; and the endosperm rich in starch [5]. However, Bran and germ are often removed by the grain-refining process that significantly reduces the health advantages of whole grains. One of the most valuable fiber components naturally occurring in the outer bran of grains is β-glucan [6], which has been removed as a byproduct during the milling process of grains that has been underestimated for a long time [7]. In recent decades, β-glucan has risen much attention for its unique properties of being both a dietary fiber and bioactive compound of whole grains.
As a non-starch polysaccharide, β-glucan is widely distributed in brans of grains, but also produced by many bacteria, yeast, and fungi [6]. It has been reported by numerous studies that β-glucan promotes health in different ways depending on its source, structure, solubility, and molecular weight [8]. β-glucan from mushrooms was found to possess immune-modulation effects in both animal studies and human trials [9,10]. More research reported the cholesterol-lowering ability of cereal β-glucan with different structure and solubility compared to the microorganism sources [11,12]. Nowadays, β-glucan has been widely studied for its abilities to modulate the immune system, gut health, and to prevent metabolic syndrome [13].
This review summarized the latest knowledge about β-glucan originated from different sources and their health implications, giving some insights to the future research directions and applications of β-glucan. The soluble and insoluble forms of β-glucans were also compared to understand the correlations between the physiochemical properties of β-glucans and its health-promoting effects.
Chemical Properties and Food Sources of β-glucan
β-glucan is a group of polysaccharides composed of glucose molecules connected by β-(1-3) and β-(1-4) or β-(1-6) linkages. The structure and solubility of β-glucan highly differed among grains and microorganisms [14]. Cereal β-glucans mainly consist of linear polymers of glucose connected by 70% of β-(1-4) and 30% β-(1-3) linkages [5]. In contrast, yeast or fungal β-glucans are connected by β-(1-3) and β-(1-6) linkages. Because of the structural variance, grain β-glucans exhibited much higher solubility than the yeast and fungal types, thus being considered as water-soluble β-glucans [14]. Water soluble and insoluble β-glucans demonstrate different biological functions and physiological properties [8]. Soluble β-glucans are known to increase the viscosity in the gut and contribute largely to reduced blood glucose and cholesterol levels [7,15]. Insoluble β-glucans with β-(1-6) branching has been predominantly proved to enhance the immune responses via elevated connections to specific receptors [16].
In addition to solubility, molecular weight (MW) of β-glucans is another determinant of its physiological functions [17]. The MWs of β-glucans vary within and among species. It was reported that the cholesterol-lowering effects of β-glucans were reduced by half in the low MW (210kDa) relative to the high MW polymers (530 kDa) [12]. This fact can be partly attributed to the positive association between MW and viscosity of β-glucans in aqueous solution [14,18].
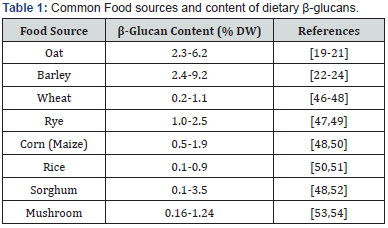
Among all the grains types, oat has been studied the most for its considerable soluble β-glucan levels (4.4-5.8%, DW) [19- 21]. Compared to oat, barley contains relatively higher amounts of β-glucans (2.4-9.2% DW). However, it has been less utilized mainly due to its sensory acceptability and difficulty in processing [22-24]. Noticeably, highland barley (also known as Tibet hull-less barley) has been reported to have higher β-glucan levels, as high as 9.2% [24]. Other grains and edible mushrooms also contain certain amounts of β-glucans but much lower than those of oat and barley. The β-glucan contents of common food sources are summarized in Table 1.
Health benefits of β-Glucans
Immune Modulation
Innate immune modulation is important for the defense system against the invasion of pathogens to the host [25]. β-Glucan has been widely investigated as an immune-modulator which participates in the upregulation or downregulation of innate immune responses [26]. Macrophage, an essential member of the innate immune defense system, was found to be the most important biological target in the immune-regulatory effects of β-Glucans [25].
β-Glucan activates macrophages in multiple ways. Dectin-1, as a C-type surface receptor of macrophages, can be specifically targeted by the β-(1-3) linkages [16]. Compared to soluble β-glucans, β-(1-3) and β-(1-6) linkages in insoluble yeast and fungi β-glucan exhibited higher affinity to Dectin-1 because the branching structures facilitated the recognition [27,28]. Upon the binding of β-glucans and Dectin-1, immune responses can be induced, subsequently leading to the secretion of proinflammatory cytokines such as TNF-α [29]. Recent studies also found that synergic effects existed between Dectin-1 and Tolllike receptors (TLRs)-a type of extracellular pattern recognition receptors [28]. Insoluble β-glucans stimulated a stronger immune response in receptor cell lines than soluble β-glucans. Insoluble β-glucans were capable of activating the co-expressions of TLR4 and both Dectin-1 variants: Dectin-1A and Dectin-1B. Contrastingly, soluble β-glucans were only active when Dectin-1A and TLR4 were co-expressed. Likewise, the study by Patidar et al. indicated that, compared to soluble barley β-glucan, insoluble yeast β-Glucan exerted stronger stimulation for the co-localization of Dectin-1 and TLR2 in macrophages infected by L. donovani with increased production of nitric oxide, an inflammatory mediator [27]. In their in vivo study using L. donovani infected BALB/c mice, soluble β-glucans showed significantly higher parasite-killing ability and stronger anti-leishmanial functions.
Monocyte is another member of innate immunity system and can differentiate into macrophages in certain conditions. Based on an in vivo human endotoxemia model, Novakovic et al. reported that the administration of fungal β-glucan successfully reversed the monocytes’ immunological tolerance induced by lipopolysaccharides (LPS), and restored the release of cytokines like IL-6 and TNF-α [30]. In a clinical trial, yeast β-glucan showed a good performance on improving LPS induced responses in monocytes from human subjects [31]. Taking 250 mg/day β-glucan for 10 days effectively increased the production of cytokines such as IL-2, IL-4, IL-5 and IFN-γ upon the upregulation of blood monocytes.
Collectively, it was suggested that β-glucans exerted immune protective functions mostly via the regulation of macrophages and monocytes and recovery of the secretion of cytokines. The structural conformation strongly influences its mechanisms towards immune regulation, thus requires a better understanding to make it a persuasive therapeutic strategy for humans.
Metabolic syndrome Prevention
Metabolic syndrome (MS), as one of the leading public health concerns around the world, is identified in obese patients with metabolic disorders including insulin resistance, hyperglycemia, dyslipidemia, and hypertension [32]. As early as 1997, β-glucan from grains source was authorized by FDA to lower blood cholesterol levels when consumed 3 g daily [33]. The protective effects against MS of β-glucan are closely connected to its ability to lower cholesterol and regulate blood glucose.
A meta-analysis of 28 randomized controlled trials assessed the cholesterol-lowering effect of oat β-glucan [17]. They confirmed that an intake of ≥3 g oat β-glucan significantly decreased total cholesterol and LDL levels by 0.3 and 0.25mmol/L, respectively, without effects on the HDL levels. Besides, the protective effects of β-glucan were elevated in diabetic individuals. Consistently, a meta-analysis of barley β-glucan including 615 subjects revealed that, to achieve a 0.25 mmol/L reduction in LDL level, a median dose of 6.5 g/day barley β-glucan should be taken for 4 weeks, which was a larger effective dose and longer treatment time compared to oat β-glucan [34].
So far, most MS research about β-glucan used the grain sources, only a few focused on the insoluble sources. Cao et al investigated the effects of yeast β-glucan on mice with MS induced by a high-fat diet (HFD) [35]. It was observed that 50 mg/kg/ day yeast β-glucan decreased the blood glucose level of the MS mice through the reduction of a major glucose transporter-SGLT-1 in the small intestine. In their follow-up experiments using ob/ ob mice model, yeast β-glucan was found to downregulate key regulatory proteins involved in glucose or lipid metabolisms such as p-AMPK, Fas and p-ACC [36].
The key mechanism involved in the effects of soluble β-glucans to prevent MS is associated with the formation of high viscosity solution in the gut, which on the one hand, inhibited the uptake of glucose and cholesterol to the blood circulation; on the other hand, made the subjects fell satiety easier thus decreasing food intake and total energy [17,37].
Gut microbiota has been a popular topic in recent years, which was speculated to be one of the mechanisms responsible for the protective effects against MS of both soluble and insoluble β-glucans [35,36,38]. It was found that the genus positively related to diabetes- Bacteroides, was decreased by soluble β-glucans. Consistently, the relative abundance of Akkermansia, which is beneficial to glucose and insulin homeostasis, was increased by insoluble β-glucans in both HFD induced mice and ob/ob mice models [35,36].
β-glucan serves as an important food component in the prevention and dietary treatment of MS. However, its effects are strongly affected by MW, solubility, processing methods, and food matrices. Further research about the interactions of these aspects still needs to be developed.
Gut Health Regulation
Emerging evidence suggested that β-glucans can serve as a prebiotic to assist gut health preservation. Gut microbiota regulation is a predominant factor contributing to not only gut health preservation, but also other health benefits [39]. Shortchain fatty acids (SCFAs), as the key metabolites of gut microbiota fermentation, play an important role in the process [40]. Chen et al. reported the gut protective effects of β-glucans from highland barley on mice with ethanol-induced gastric lesions [41]. Their results indicated that a pre-treatment of 250 mg/kg β-glucans alleviated the gastric injuries via several mechanisms: to enhance the antioxidant ability of the system by upregulating the levels of SOD and CAT; to stimulate immune responses by increase the release of nitric oxide; and to enrich the SCFAs in the gut. SCFAs enrichment was also observed in many studies using β-glucans supplementation [42,43]. In a human study comparing β-glucans with other dietary fibers, β-glucans showed a superior impact on the production of propionate in the gut, supporting the robust role of β-glucans as a prebiotic [43]. However, contradictory results were observed in terms of the gut bacteria strains boosted by β-glucans. In a study where in vitro human feces were used to study the fermentation of insoluble fungal β-glucans, both Bacteroides and Clostridium were found to be elevated by β-glucans treatment [44]. Inversely, genus Bacteroides was decreased in the gut of HFD induced diabetic mice model fed with oat β-glucans [38]. The discrepancy is probably attributed to the differences in the physiochemical properties between fungal and grain β-glucans.
Generally, β-glucans, especially those soluble ones from grains help maintain gut health mainly through the production of SCFAs such as propionic acids. The production of SCFAs can, in turn, enrich the beneficial species of gut microbiota and form a positive feedback loop [45].
Summary and Future Direction
In summary, the current review provided direct comparisons among β-glucans from different sources regarding their health benefits. It has been well-documented that β-glucans have protective effects on the modulation of the immune system, especially the innate immune system, the prevention of metabolic syndrome, and the regulation of gut health. The MW, solubility, viscosity, and sources of β-glucans all strongly influence the biological functions and physiological properties of β-glucans. However, the elucidation of their associations remains unclear. Besides, the impact of colon digestion on the bioactivity and functions of β-glucans within the food matrices are still poorly understood and requires better illustrations in the future.
References
- De Munter JSL, Hu FB, Spiegelman D, Franz M, Van Dam RM (2007) Whole grain, bran, and germ intake and risk of type 2 diabetes: A prospective cohort study and systematic review. PLoS Med 4(8): e261.
- Philip Karl J, McKeown NM (2014) Whole Grains in the Prevention and Treatment of Abdominal Obesity. In: Nutrition in the Prevention and Treatment of Abdominal Obesity.; Elsevier Inc.: Amsterdam, The Netherlands: 515-528.
- Mellen PB, Walsh TF, Herrington DM (2008) Whole grain intake and cardiovascular disease: A meta-analysis. Nutr Metab Cardiovasc Dis 18(4): 283-290.
- Jonnalagadda SS, Harnack L, Hai Liu R, McKeown N, Seal C, et al. (2011) Putting the Whole Grain Puzzle Together: Health Benefits Associated with Whole Grains-Summary of American Society for Nutrition 2010 Satellite Symposium. J Nutr 141(5): 1011S-1022S.
- Liu RH (2007) Whole grain phytochemicals and health. J Cereal Sci 46(3): 207-219.
- Rondanelli M, Opizzi A, Monteferrario F (2009) The biological activity of beta-glucans. Minerva medica 100(3): 237.
- Lee YT, Puligundla P, Schwarz PB (2017) Molecular weight, solubility and viscosity of β-Glucan preparations from barley pearling byproducts. SainsMalaysiana 46(5): 713-718.
- Maheshwari G, Sowrirajan S, Joseph B (2019) β-Glucan, a dietary fiber in effective prevention of lifestyle diseases -An insight. Bioact Carbohydrates Diet Fibre (19): 100187.
- Akramiene D, Kondrotas A, Didziapetriene J, Kevelaitis E (2007) Effects of beta-glucans on the immune system. Medicina (Kaunas) 43(8): 597.
- Volman JJ, Ramakers JD, Plat J (2008) Dietary modulation of immune function by β-glucans. PhysiolBehav 94(2): 276-284.
- Sima P, Vannucci L, Vetvicka V (2018) β-glucans and cholesterol (Review). Int J Mol Med 41(4): 1799-1808.
- Wolever TMS, Tosh SM, Gibbs AL, Brand-miller J, Duncan AM, et al. (2010) Physicochemical properties of oat beta-glucan influence its ability to reduce serum LDL cholesterol in humans : a randomized clinical trial. Am J Clin Nutr 92(4):723-732.
- El Khoury D, Cuda C, Luhovyy BL, Anderson GH (2012) Beta glucan: Health benefits in obesity and metabolic syndrome. J Nutr Metab (2012).
- Suchecka D, Gromadzka-Ostrowska J, Zyła E, Harasym JP, Oczkowski M (2017) Selected physiological activities and health promoting properties of cereal beta-glucans. A review. J Anim Feed Sci 26(3):183-191.
- Wang Q, Ellis PR (2014) Oat β-glucan: Physico-chemical characteristics in relation to its blood-glucose and cholesterol-lowering properties. Br J Nutr 112(S2): S4-S13.
- Nakashima A, Yamada K, Iwata O, Sugimoto R, Atsuji K, et al. (2018) β-Glucan in Foods and Its Physiological Functions. J Nutr Sci Vitaminol (Tokyo) 64(1): 8-17.
- Whitehead A, Beck EJ, Tosh S, Wolever TMS (2014) Cholesterol-lowering effects of oat β-glucan: A meta-analysis of randomized controlled trials1. Am J Clin Nutr 100(6):1413-1421.
- Mäkelä N, Brinck O, Sontag-Strohm T (2020) Viscosity of β-glucan from oat products at the intestinal phase of the gastrointestinal model. Food Hydrocoll (100): 105422.
- Herrera MP, Gao J, Vasanthan T, Temelli F, Henderson K (2016) β-Glucan content, viscosity, and solubility of Canadian grown oat as influenced by cultivar and growing location. Can J Plant Sci 96(2):183-96.
- Beer MU, Wood PJ, Weisz J (1997) Molecular weight distribution and (1→3)(1→4)-β-D-glucan content of consecutive extracts of various oat and barley cultivars. Cereal Chem 74(4):476-80.
- Andersson AAM, Börjesdotter D (2011) Effects of environment and variety on content and molecular weight of β-glucan in oats. J Cereal Sci 54(1):122-8.
- Nishantha MDLC, Zhao X, Jeewani DC, Bian J, Nie X, et al. (2018) Direct comparison of β-glucan content in wild and cultivated barley. Int J Food Prop 21(1):2218-28.
- Martínez M, Motilva MJ, López de las Hazas MC, Romero MP, Vaculova K, et al. (2018) Phytochemical composition and β-glucan content of barley genotypes from two different geographic origins for human health food production. Food Chem 245: 61-70.
- Zhang G, Yang K, Xue W, Zeng X, Xu Q, et al. (2020) Comparative proteomic analysis of hulless barley cultivars (Hordeum vulgare L.) differing distinctly in β-glucan content. LWT: 110085.
- Yin M, Zhang Y, Li H (2019) Advances in research on immunoregulation of macrophages by plant polysaccharides. Front Immunol (10):145.
- Wismar R, Brix S, Frøkiær H, Lærke HN (2010) Dietary fibers as immunoregulatory compounds in health and disease.Annals of the New York Academy of Sciences 1190(1):70-85.
- Patidar A, Mahanty T, Raybarman C, Sarode AY, Basak S, et al. (2020) Barley beta-Glucan and Zymosan induce Dectin-1 and Toll-like receptor 2 co-localization and anti-leishmanial immune response in Leishmania donovani-infected BALB/c mice. Scand J Immunol: e12952.
- Kanjan P, Sahasrabudhe NM, de Haan BJ, de Vos P (2017) Immune effects of β-glucan are determined by combined effects on Dectin-1, TLR2, 4 and 5. J Funct Foods (37): 433-440.
- Yadav M, Schorey JS (2006) The β-glucan receptor dectin-1 functions together with TLR2 to mediate macrophage activation by mycobacteria. Blood 108(9):3168-3175.
- Novakovic B, Habibi E, Wang SY, Arts RJW, Davar R, et al. (2016) β-Glucan Reverses the Epigenetic State of LPS-Induced Immunological Tolerance. Cell 167(5): 1354-1368.
- McFarlin BK, Carpenter KC, Davidson T, McFarlin MA (2013) Baker’s yeast beta glucan supplementation increases salivary IgA and decreases cold/flu symptomatic days after intense exercise. J Diet Suppl 10(3):171-83.
- Ibitoye OB, Ajiboye TO (2018) Dietary phenolic acids reverse insulin resistance, hyperglycaemia, dyslipidaemia, inflammation and oxidative stress in high-fructose diet-induced metabolic syndrome rats. Arch Physiol Biochem 124(5):410-417.
- Food and Drug Administration (1997) Department of Health and Human Services (Food and Drug Administration) Food Labeling: Health claims; Oats and Coronary Heart disease; Final Rule. Fed Regist.
- Ho HVT, Sievenpiper JL, Zurbau A, Mejia SB, Jovanovski E, et al. (2016) A systematic review and meta-analysis of randomized controlled trials of the effect of barley β-glucan on LDL-C, non-HDL-C and apoB for cardiovascular disease risk reductioni-IV. Eur J Clin Nutr 70(11): 1239-1245.
- Cao Y, Zou S, Xu H, Li M, Tong Z, et al. (2016) Hypoglycemic activity of the Baker’s yeast β-glucan in obese/type 2 diabetic mice and the underlying mechanism. Mol Nutr Food Res 60(12):2678-2690.
- Cao Y, Sun Y, Zou S, Li M, Xu X (2017) Orally Administered Baker’s Yeast β-Glucan Promotes Glucose and Lipid Homeostasis in the Livers of Obesity and Diabetes Model Mice. J Agric Food Chem 65(44):9665-9674.
- Marasca E, Boulos S, Nyström L (2020) Bile acid-retention by native and modified oat and barley β-glucan. CarbohydrPolym 236:116034.
- Zhu Y, Dong L, Huang L, Shi Z, Dong J, et al. (2020) Effects of oat β-glucan, oat resistant starch, and the whole oat flour on insulin resistance, inflammation, and gut microbiota in high-fat-diet-induced type 2 diabetic rats. J Funct Foods 69:103939.
- Jayachandran M, Chen J, Chung SSM, Xu B (2018) A critical review on the impacts of β-glucans on gut microbiota and human health. J Nutr Biochem61:101-110.
- Makki K, Deehan EC, Walter J, Bäckhed F (2018) The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe 23(6):705-715.
- Chen H, Nie Q, Xie M, Yao H, Zhang K, et al. (2019) Protective effects of β-glucan isolated from highland barley on ethanol-induced gastric damage in rats and its benefits to mice gut conditions. Food Res Int 122:157-166.
- Aoe S, Yamanaka C, Fuwa M, Tamiya T, Nakayama Y, et al. (2019) Effects of BARLEYmax and high-β-glucan barley line on short-chain fatty acids production and microbiota from the cecum to the distal colon in rats. PLoS One 14(6): e0218118.
- Carlson JL, Erickson JM, Hess JM, Gould TJ, Slavin JL (2017) Prebiotic dietary fiber and gut health: Comparing the in vitro fermentations of beta-glucan, inulin and xylooligosaccharide. Nutrients 9(12):1361.
- Cantu-Jungles TM, Ruthes AC, El-Hindawy M, Moreno RB, Zhang X, et al. (2018) In vitro fermentation of Cookeina speciosa glucans stimulates the growth of the butyrogenic Clostridium cluster XIVa in a targeted way. CarbohydrPolym (183):219-229.
- Bishehsari F, Engen PA, Preite NZ, Tuncil YE, Naqib A, et al. (2018) Dietary fiber treatment corrects the composition of gut microbiota, promotes SCFA production, and suppresses colon carcinogenesis. Genes (Basel) 9(2):102.
- Marcotuli I, Houston K, Schwerdt JG, Waugh R, Fincher GB, et al. (2016) Genetic diversity and genome wide association study of β-glucan content in tetraploid wheat grains. PLoS One 11(4): e0152590.
- Henry RJ (1987) Pentosan and (1 → 3),(1 → 4)-β-Glucan concentrations in endosperm and wholegrain of wheat, barley, oats and rye. J Cereal Sci 6(3):253-258.
- Hamid A, Ilyas M, Kalsoom S (2017) Effect of wheat and corn bran and barley and sorghum ß-glucan extracts on the plasma cholesterol level of dietary-induced hypercholesterolemic rats. Pak J Zool 49(5).
- Saastamoinen M, Plaami S, Kumpulainen J (1989) Pentosan and β-glucan content of finnish winter rye varieties as compared with rye of six other countries. J Cereal Sci 10(3):199-207.
- Demirbas A (2005) β-Glucan and mineral nutrient contents of cereals grown in Turkey. Food Chem 90(4):773-777.
- Jung TD, Shin GH, Kim JM, Choi S Il, Lee JH, et al. (2017) Comparative analysis of γ-oryzanol, β-glucan, total phenolic content and antioxidant activity in fermented rice bran of different varieties. Nutrients 9(6):571.
- Niba LL, Hoffman J (2003) Resistant starch and β-glucan levels in grain sorghum (Sorghum bicolor M.) are influenced by soaking and autoclaving. Food Chem 81(1):113-118.
- Bach F, Helm CV, Bellettini MB, Maciel GM, Haminiuk CWI (2017) Edible mushrooms: a potential source of essential amino acids, glucans and minerals. Int J Food Sci Technol 52(11):2382-2392.
- Khan AA, Gani A, Khanday FA, Masoodi FA (2018) Biological and pharmaceutical activities of mushroom β-glucan discussed as a potential functional food ingredient. Bioact Carbohydrates Diet Fibre (16):1-13.






























