Literature Review of Analytical Methods for Determination of Triamcinolone Acetonide and Benzyl Alcohol
Yara Elkady, Sobhy M El-Adl, Mohamed Baraka and Mahmoud M Sebaiy*
Department of Medicinal Chemistry, Zagazig University, Egypt
Submission:August 31, 2020; Published: September 08, 2020
*Corresponding author: Mahmoud M Sebaiy, Department of Medicinal Chemistry, Faculty of Pharmacy, Zagazig University, Zagazig, Egypt
How to cite this article: Yara E, Sobhy M E, Mohamed B, Mahmoud M S. Literature Review of Analytical Methods for Determination of Triamcinolone Acetonide and Benzyl Alcohol. Nov Appro Drug Des Dev. 2020; 5(3): 555663. DOI: 10.19080/NAPDD.2020.05.555663
Abstract
In this review article, we will introduce all reported methods that have been developed for determination of triamcinolone acetonide and benzyl alcohol in their pure form, combined form with other drugs, combined form with degradation products, and in biological samples. We also will shed the light on the most important combination of our cited drugs.
Keywords: Drugs; Benzyl alcohol; Acetonitrile; phosphoric acid; Benzoic acid
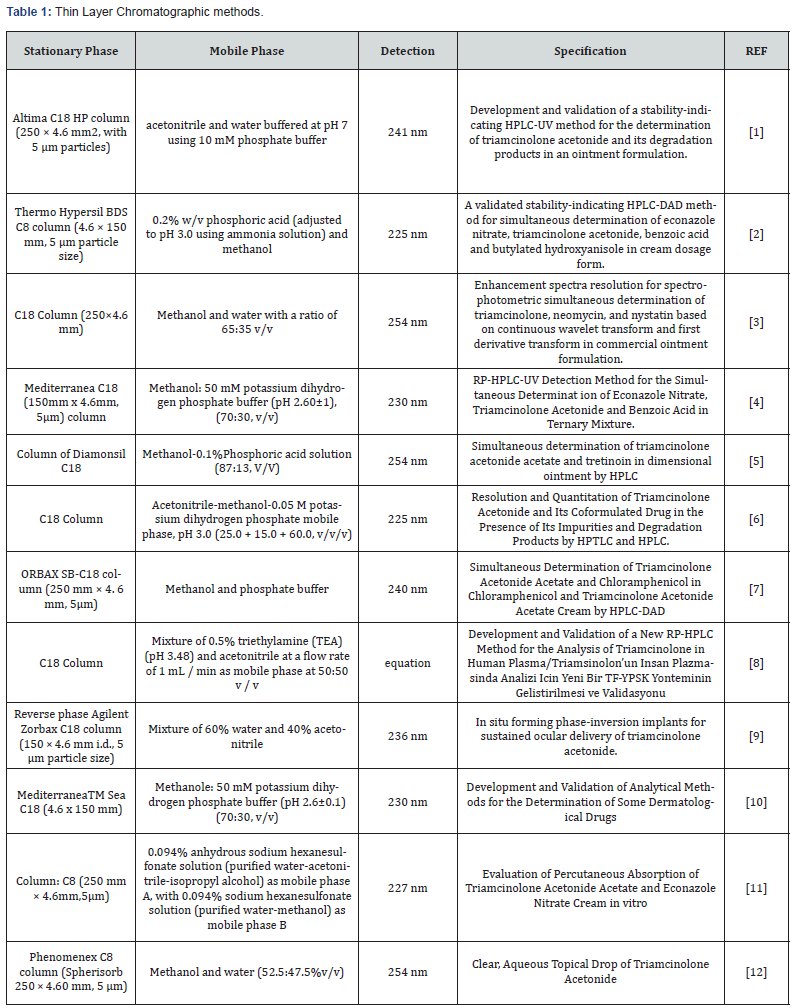
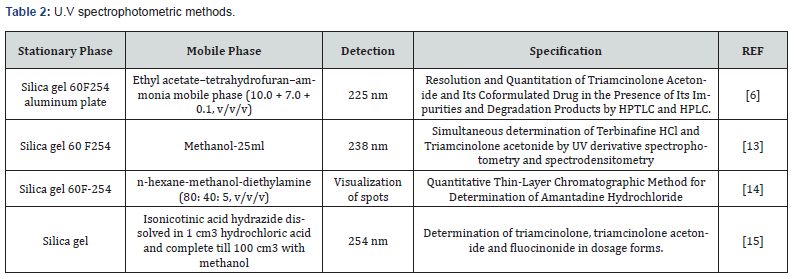
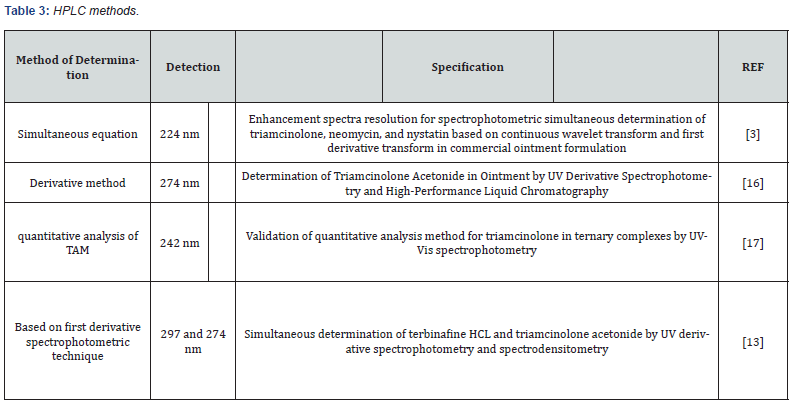
Metods for Analysis of Triamcinolone
Chromatographic methods:
HPLC methods:
a)Spectrofluorimetric methods: The methodology depends on the fluorescence of creation of triamcinolone acetonide oxidized by concentrated sulfuric corrosive. The impact of H2SO4, β-cyclodextrin (β-CD), dissolvable and cetyltrimethylammonium bromide (CTMAB) were talked about. Furthermore, two diagnostic frameworks were set up. One is CTMAB framework, the straight range is 0-4.6×10-6 mol/L, and as far as possible is 3.59×10−8 mol/L (Table 1-3). The other is β-CD and ethanol framework, the straight range is 0–2.3×10−6 mol/L and as far as possible is 1.91×10−8 mol/L. The selectivity of examination, the investigative figures of improvement, and the exactness of the technique are exhibited with the assurance of triamcinolone acetonide in pharmaceutical arrangements [18].
b)Luminescence methods: A rapid lateral flow immunochromatographic measure (ICA) was created for the quantitative identification of TAM utilizing a test dependent on up conversion radiance nanoparticles. Lanthanide-doped up conversion nanoparticles (UCNPs) were incorporated in a framework involving water and ethylene glycol, and a silicon dioxide layer was secured at the carboxyl site. A coupling site assurance procedure was utilized to diminish the foundation sign of UCNPs-ICA. Utilizing dexamethasone subsidiary as a covering antigen, the ideal UCNPs-ICA displays great unique straight location for TAM in the range 1.0-100 ng mL-1 with a middle inhibitory grouping of 9.8 ng mL-1. As far as possible for TAM in a corrective example are 20 μg kg-1. The pretreatment samples need dilution with water, suggesting the assay can quantitate TAM on-site using a portable upconversion luminescence reader with a cumulative analysis time of only 10 min. [19] (Table 4-6).
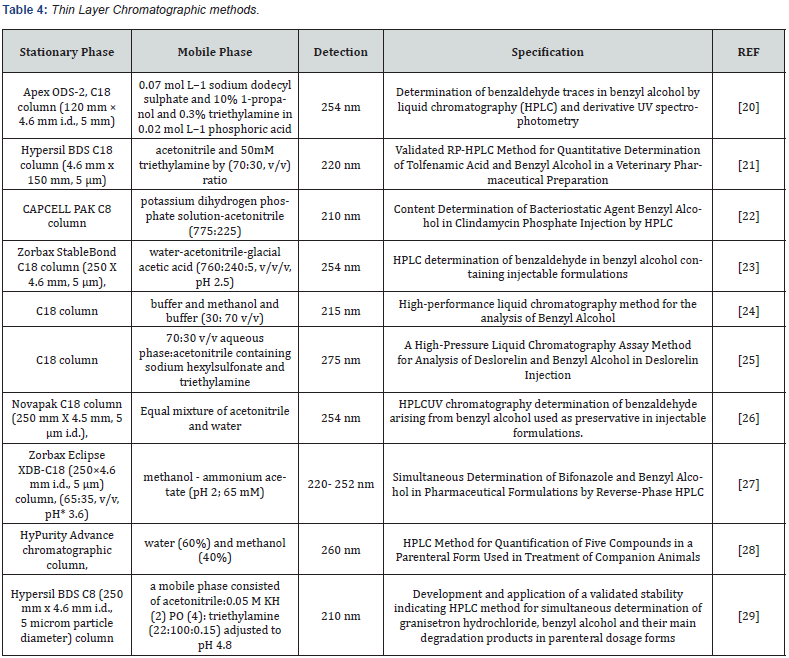
Methods for Analysis of Benzyl Alcohol
Chromatographic methods:
HPLC methods:
Conclusion
This literature review represents an up to date survey about all reported methods that have been developed for determination of triamcinolone acetonide and benzyl alcohol in their pure form, combined form with other drugs, combined form with degradation products, and in biological samples such as liquid chromatography, spectrophotometry, spectroflourimetry, etc.
References
- Van Heugten A J P, de Boer W, de Vries W S, Markesteijn C M A,Vromans H (2018) Development and validation of a stability-indicating HPLC-UV method for the determination of triamcinolone acetonide and its degradation products in an ointment formulation. Journal of Pharmaceutical and Biomedical Analysis 149: 265-270.
- Baker M M, Belal T S, Mahrous M S, Ahmed H M, Daabees H G (2016) A validated stability-indicating HPLC-DAD method for simultaneous determination of econazole nitrate, triamcinolone acetonide, benzoic acid and butylated hydroxyanisole in cream dosage form. Analytical Methods 8(10): 2185-2200.
- Abasi N, Sohrabi M R, Motiee F, Davallo M(2020)Enhancement spectra resolution for spectrophotometric simultaneous determination of triamcinolone, neomycin, and nystatin based on continuous wavelet transform and first derivative transform in commercial ointment formulation.Optik: 165315.
- EL-Zaher A A, Elkady E F, Al-Tahami A A (2019) RP-HPLC-UV Detection Method for the Simultaneous Determinat ion of Econazole Nitrate, Triamcinolone Acetonide and Benzoic Acid in Ternary Mixture. Current Pharmaceutical Analysis 15(7): 738-744.
- Xiao L, Ding Z, Shigui X U, Guo Y, Wang Y, Luo M (2016) Simultaneous Determination of Triamcinolone Acetonide Acetate and Tretinoin in Dimensional Ointment by HPLC. China Pharmacy 27(3): 381-383.
- Abbas S S, Hegazy M A, Hendawy H A, Weshahy S A,Abdelwahab M H (2018) Resolution and Quantitation of Triamcinolone Acetonide and Its Coformulated Drug in the Presence of Its Impurities and Degradation Products by HPTLC and HPLC. Journal of AOAC International, 101(4), 981-991.
- Yang P, Zeng W, Sun L (2016) Simultaneous Determination of Triamcinolone Acetonide Acetate and Chloramphenicol in Chloramphenicol and Triamcinolone Acetonide Acetate Cream by HPLC-DAD. China Pharmacist 19(3): 591-593.
- Muralidharan S, Venugopal V, Kumar J, PARASURAMAN S (2016) Development and Validation of a New RP-HPLC Method for the Analysis of Triamcinolone in Human Plasma. Turk J Pharm Sci 13(1): 9-16.
- Sheshala R, Hong G C, Yee W P, Meka V S, Thakur R R S (2019) In situ forming phase-inversion implants for sustained ocular delivery of triamcinolone acetonide. Drug Delivery and Translational Research: 9(2): 534-542.
- Al-Tahami A A (2018) Development and Validation of Analytical Methods for the Determination of Some Dermatological Drugs CU Theses.
- Wu Y, Xiao X (2017) Evaluation of Percutaneous Absorption of Triamcinolone Acetonide Acetate and Econazole Nitrate Cream in vitro. Journal of Hubei University of Chinese Medicine (2): 10.
- Trinh H M, Cholkar K, Joseph M, Yang X, Mitra A K (2017) Clear, aqueous topical drop of triamcinolone acetonide.AapsPharmscitech 18(7): 2466-2478.
- El-Saharty YS, Hassan, NY,Metwally, FH (2002) Simultaneous determination of Terbinafine HCl and Triamcinolone acetonide by UV derivative spectrophotometry and spectrodensitometry. J Pharm Biomed Anal 28(3): 569-580.
- Hartmann F,Ude, P (1974) Quantitative thin-layer chromatography determination of the urinary excretion of free triamcinolone-acetonide during external treatment with occlusive dressings in patients with psoriasis.VerhDtschGes Inn Med 80:1548-51.
- Danica A Zivanov-Stakić D, Vladimirov S, Zubac K (1990) Determination of triamcinolone, triamcinolone acetonide and fluocinonide in dosage forms. Acta poloniaepharmaceutica 47(1): 15-7.
- Kedor-Hackmann, E R M, Gianotto, E A S and Santoro, M I R M (1997) Determination of Triamcinolone Acetonide in Ointment by UV Derivative Spectrophotometry and High Performance Liquid Chromatography. Analytical Letters 30(10):1861-1871.
- Aquino G, Stopilha R, Fernandes-Pedrosa M, Santos K, Egito S, et al. (2011) Validation of quantitative analysis method for triamcinolone in ternary complexes by UV-Vis spectrophotometry.Revista de CienciasFarmaceuticasBasica e Aplicada 32(1).
- Zu-ting P, Jun-ping Y (2004) Spectrofluorimetric method for the determination of triamcinolone acetonide. Wuhan Univ J Nat Sci 9: 353–358.
- Zhang S, Yao T, Wang S, Feng R, Chen L,et al. (2019) Upconversion luminescence nanoparticles-based immunochromatographic assay for quantitative detection of triamcinolone acetonide in cosmetics Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 214: 302-308.
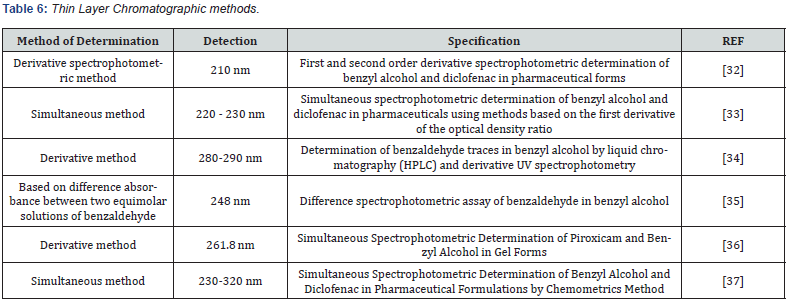
- Di Pietra A M, Cavrini V, Raggi M A (1987) Determination of benzaldehyde traces in benzyl alcohol by liquid chromatography (HPLC) and derivative UV spectrophotometry. International journal of pharmaceutics 35(1-2): 13-20.
- Mahgoub, S (2017) Validated RP-HPLC Method for Quantitative Determination of Tolfenamic Acid and Benzyl Alcohol in a Veterinary Pharmaceutical Preparation. Austin Chromatogr 4(1): 1046.
- Ge H S, Liu Y R, Zhao K H (2010) Content Determination of Bacteriostatic Agent Benzyl Alcohol in Clindamycin Phosphate Injection by HPLC. [J] China Pharmacy 25.
- Uzunovic A, Pilipovic S, Tubić B, Sapcanin A,Tahirovic I (2017) HPLC determination of benzaldehyde in benzyl alcohol containing injectable formulations. Acta pharmaceuticaHungarica 87: 200-201.
- Khade, V and Mirgane, S (2014) High-performance liquid chromatography method for the analysis of Benzyl Alcohol. International Journal of Scientific & Engineering Research5(11): 887-889.
- Wood GC, Iyer MR, Geller AM, Fleischner AM,Sheth, BB (1998) A High Pressure Liquid Chromatography Assay Method for Analysis of Deslorelin and Benzyl Alcohol in Deslorelin Injection, Journal of Liquid Chromatography & Related Technologies, 21(14): 2183-2190.
- Kashani H N, Moghaddam F A, Mehramizi A (2012) HPLC–UV chromatography determination of benzaldehyde arising from benzyl alcohol used as preservative in injectable formulations. Asian Journal of Pharmaceutical and Clinical Research 5(2): 98-100.
- Čudina OA, Čomor MI, Janković IA (2005) Simultaneous Determination of Bifonazole and Benzyl Alcohol in Pharmaceutical Formulations by Reverse-Phase. HPLC Chroma 61: 415-418.
- Stan GS, Badea IA,Aboul-Enein HY (2016) HPLC Method for Quantification of Five Compounds in a Parenteral Form Used in Treatment of Companion Animals. Journal of Chromatographic Science 54(9): 1567-1572.
- Hewala I, El-Fatatre H, Emam E,Mubrouk M (2010) Development and application of a validated stability-indicating HPLC method for simultaneous determination of granisetron hydrochloride, benzyl alcohol and their main degradation products in parenteral dosage forms.Talanta 82(1): 184-195.
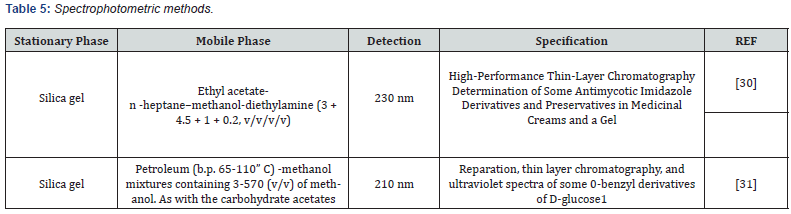
- Čakar M, Popović G,Agbaba, D (2005) High-performance thin-layer chromatography determination of some antimycotic imidazole derivatives and preservatives in medicinal creams and a gel. Journal of AOAC International 88(5): 1544-1548.
- Tate M E, Bishop C T (1963) Preparation, thin layer chromatography, and ultraviolet spectra of some O-benzyl derivatives of D-glucose. Canadian Journal of Chemistry 41(7): 1801-1806.
- De Micalizzi Y C, Pappano N B, Debattista N B (1998) First and second order derivative spectrophotometric determination of benzyl alcohol and diclofenac in pharmaceutical forms.Talanta 47(3): 525-530.
- Ghasemi J, Niazi A, Ghobadi S (2005) Simultaneous spectrophotometric determination of benzyl alcohol and diclofenac in pharmaceuticals using methods based on the first derivative of the optical density ratio. Pharmaceutical Chemistry Journal 39: 671-675.
- Di Pietra A M, Cavrini V, Raggi M A (1987) Determination of benzaldehyde traces in benzyl alcohol by liquid chromatography (HPLC) and derivative UV spectrophotometry. International journal of pharmaceutics 35(1-2): 13-20.
- Hewala I I (1993) Difference spectrophotometric assay of benzaldehyde in benzyl alcohol.Talanta 40(6): 919-923.
- Goger N G, Zahtep L M, Berkkan A,Senturk Z (2005) Simultaneous spectrophotometric determination of piroxicam and benzyl alcohol in gel forms.Chemiaanalityczna 50(6): 1059.
- Ghasemi J, Niazi A, Ghobadi S (2005) Simultaneous spectrophotometric determination of benzyl alcohol and diclofenac in pharmaceutical formulations by chemometrics method. Journal of the Chinese Chemical Society 52(5): 1049-1054.






























