An Update on Exosomes as Drug Delivery Vehicles: Obstacles to its Clinical Translation
Ahlam Ali*, Aaron Maguire and Fiona Furlong
School of Pharmacy, Queen’s University Belfast, UK
Submission:March 25, 2019; Published: April 02, 2019
*Corresponding author: Ahlam Ali, School of Pharmacy, Queen’s University, Belfast, Northern Ireland, UK
How to cite this article: Ahlam A, Aaron M, Fiona F. An Update on Exosomes as Drug Delivery Vehicles: Obstacles to its Clinical Translation. Nov Appro Drug Des Dev 2019; 5(1): 555655. DOI: 10.19080/NAPDD.2019.04.555655
Abstract
Exosomes are membrane vesicles with a diameter of 40-100 nm that are secreted by many cell types into the extracellular milieu. Exosomes have great potential to be drug delivery vehicles due to their natural material transportation properties, intrinsic long-term circulatory capability, and excellent biocompatibility, which are suitable for delivering a variety of chemicals, proteins, nucleic acids, and gene therapeutic agents. However, there are still some issues and challenges that need to be addressed including production of exosomes in large scale for clinical use, which cell type to use for exosome derivation, and determination of in vivo exosome potency and toxicology. The clinical translation of exosomal therapeutics warrants better understanding of exosome biology and function, and the development of nanotechnologies for the specific purification of well-characterised clinical-grade exosomes and their loading with a variety of therapeutic cargoes.
Keywords: Exosomes; Drug delivery; Vehicles; Protein; RNA; miRNA; siRNA
Introduction
Nanotechnology holds great promise for several new advances in targeted and controlled-release drug delivery platforms. Exosomes are nanosized biomolecules (40 to100nm) secreted by cells from multivesicular endosomes, with a density of 1.13 to 1.19g/ml in sucrose, and a sedimentation rate of 100,000g [1]. Exosomes contains an abundance of lipids (cholesterol, sphingomyelin, ceramide), proteins (tetracaines, Alix, TSG 101, HSPs) and other molecules involved in antigen presentation (MHC I and MHC II), signal transduction and targeting/adhesion. Exosomes have great ability to interact with target cells by a range of surface adhesion proteins and vector ligands such as tetracaines, integrins, CD11b and CD18 receptors, and deliver their cargo to target cells [2,3].
Exosomes transport various biological molecules, ranging from membrane receptors, proteins, to mRNA and microRNA for maintenance of biological homeostasis [4], as well as epigenetic reprogramming [5]. Exosomes as a natural liposome are ideal drug carriers used recently [6], which have many advantages over conventional nanocarriers for drug and gene delivery. Due to its nano-size, exosomes can avoid phagocytosis, fuse with the cell membrane, and bypass the engulfment by lysosomes. The fact that exosomes are a natural product of the body results in a low immune response [7]. Owning to their stability in the blood, exosomes are able to travel long distances within the body under both physiological and pathological conditions. Additionally, exosomes easily pass their contents across the cell membrane due to their unique characteristics and deliver their cargo in a biologically active form. Noteworthy, exosomes possess an intrinsic ability to cross biological barriers, including the most difficult to penetrate: the blood brain barrier (BBB) [8].
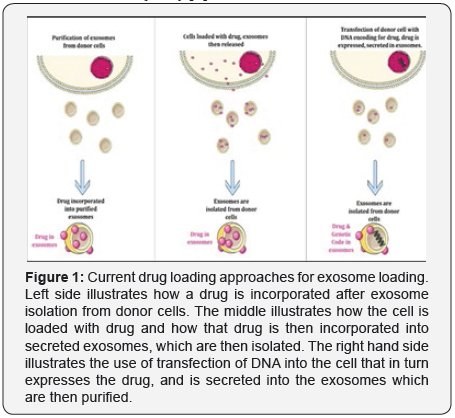
In addition, exosomes have a hydrophilic core, which makes them suitable to host water-soluble drugs. All the above makes exosomes an important tool for both diagnostic and therapeutic purposes. Several methods for exosome loading have been proposed to date, which can be classified into three different strategies (Figure 1) [9]. First strategy is loading naïve exosomes isolated from parental cells ex vitro; which is main method of loading. In this method, exosomes are first isolated from cell media by a variety of techniques. The current most widely used method, because of its lack of expense and time, is ultracentrifugation. Other methods include sucrose cushion density grade ultracentrifugation, filtration, size exclusion chromatography [10].
External loading of the EVs can now be attempted by several methodologies such as electroporation, sonication, and cell penetrating peptides [11-13]. Other two strategies include loading parental cells with a drug, which is then released in exosomes; and transfecting/infecting parental cells with DNA encoding therapeutically active compounds which are then released in exosomes. Each approach has its advantages and limitations and may be dictated by the type of therapeutic cargo, site of the disease, and conditions suitable for a specific type of exosome-encapsulated cargo.
Potential use as therapeutic vehicles-Pre-clinical studies
Exosomes should be able to carry a substantial amount of therapeutic cargo to qualify as drug delivery vehicles. A variety of cargos have now been shown to exhibit therapeutic effect after exosome-based delivery to particular tissues. Exosomes have been exploited as drug delivery vehicles for low molecularweight drugs in several investigations [14-21]. Exosomes loaded with an anti-inflammatory small molecule compound, curcumin, were shown to protect mice from lipopolysaccharides-induced brain inflammation [14,15]. The incorporation of curcumin in exosomes improved its solubility, increased circulation time, preserved drug therapeutic activity, and improved brain delivery.
Exosomes were used to load different chemotherapeutics such as Doxorubicin or Paclitaxel. Studies showed that exosome loaded chemotherapeutics accumulate in tumour tissues and reduce tumour growth in mice without the adverse effects observed with the equipotent free drug [16-18]. Paclitaxel treated MSCs mediated strong anti-tumorigenic effects because of their capacity to take up the drug and later release it in the exosomes [20]. Remarkably, the therapeutic effects of Doxorubicin-loaded exosome were shown to be greater than the commercially available Doxorubicin-loaded liposomes, Doxil; which was inefficient in reducing tumour growth in this model [16]. Exosomes derived from brain endothelial cell line, bEND.3, were loaded with anticancer drugs Doxorubicin and Paclitaxel and used for systemic delivery across the BBB to treat gliomas [19]. It has been demonstrated that exosomes can be used as vehicles for delivering siRNA [22-26].
Shtam et al. introduced two different exogenous siRNAs against RAD51 and RAD52 into exosomes derived from HeLa cells [24]. The exosome delivered siRNA against RAD51 was functional and caused massive reproductive cell death of recipient cancer cells. Since exosomes naturally carry miRNAs, a therapeutic application of this characteristic seems logical, as illustrated in the number of studies applying the approach for different disease models [27]. For example, exosome-delivered tumour suppressor miRNAs, miR-143 and let-7a, inhibited growth of prostate and breast cancer in vivo, respectively [28,29]. No adverse effects were observed in normal prostatic epithelial cells after treatment with exosome-encapsulated miR- 143 [28].
Obstacles of exosome-based therapy clinical translation
The above-mentioned studies provide an illustration as to how the exosome-based therapies may be valuable, especially for the future treatment of cancer. However, there is a number of limiting factors in the clinical translation of the findings. First of all, detailed understanding of the mechanisms about how exosomes cross biological barriers and contribute to pathology of disease, is lacking [30]. This critically limits the prospects of knowledge-based identification of tissue specific targeting peptides and identifying any endogenous cargos that may mediate potential unwanted side effects. Furthermore, it can’t be certain that siRNA cargo will affect gene expression without altering other signalling pathways. The efficiencies of drug loading into exosomes tend to be low, which plagues the expansion of these novel nanoparticles for drug delivery.
To address these challenges, the optimal methods for the encapsulation of drug/gene into exosomes should be carefully delineated in order to achieve maximum efficiency and reduce the need to administer large amounts of exosomes. Elucidating the factors (molecular weight, charge distribution, solubility, and so on) that influence drug entrapment efficiency is of fundamental importance in therapeutic applications. One major obstacle to clinical translation is the lack of suitable and scalable nanotechnologies for the purification and loading of exosomes. First, a major limitation in this field is the lack of standardized techniques for isolation and purification of exosomes. Exosomes are conventionally isolated by ultracentrifugation. However, these methods suffer from a number of limitations including poor reproducibility, tedious and time-consuming procedures, and low production yield. Additionally, the exosomes isolated by ultracentrifugation are usually contaminated with protein aggregates and other cell debris, which may affect quality attributes. Therefore, novel purification methods based on the use of specific, desired markers, such as the expression of the targeting moiety on the surface of exosomes, are required. In addition, siRNA loading into exosomes is relatively inefficient and cost-ineffective, highlighting the need for the development of novel transfection reagents tailored for nanoparticle applications. We envision that the resolution of these issues would lead to exosome-based drug delivery as a novel therapeutic strategy in the near future.
Conclusion
The safe and effective delivery of drug molecules to their target site is a field which has increasingly gained attention in drug design and development. At present, the study of exosomes has made great progress in drug carriers, chemotherapy and miRNA/siRNA. Despite these advances, there are still many challenges ahead. For example, it is necessary to further verify the safety of exosomes for clinical application, and to clarify the composition and mechanism of various substances in exosomes, and how to quickly obtain the high purity of exosomes and the dosage of clinical use. Therefore, to fulfil the clinical translation of exosomes, the main focus should be on the generation of scalable patient-derived exosomes, reliable purification and characterisation nanotechnologies, expansion of the exosome cargo repertoire and development of novel targeting strategies.
References
- Théry C, Amigorena S, Raposo G, Clayton A (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol 30(1): 3.22.
- Sousa D, Ferreira D, Rodrigues JL, Rodrigues LR (2019) Nanotechnology in Targeted Drug Delivery and Therapeutics. Applications of Targeted Nano Drugs and Delivery Systems pp. 357-409.
- Théry C, Ostrowski M, Segura E (2009) Membrane vesicles as conveyors of immune responses. Nat Rev Immunol 9(8): 581-593.
- Camussi G, Deregibus MC, Bruno S, Cantaluppi V, Biancone L (2010) Exosomes/microvesicles as a mechanism of cell-to-cell communication. Kidney Int 78(9): 838-848.
- Camussi G, Deregibus MC, Bruno S, Grange C, Fonsato V, et al. (2011) Exosome/microvesicle-mediated epigenetic reprogramming of cells. Am J Cancer Res 1(1): 98-110.
- Gulei D, Irimie AI, Cojocneanu-Petric R, Schultze JL, Berindan-Neagoe I, et al. (2018) Exosomes-Small Players, Big Sound. Bioconjug Chem 29(3): 635-648.
- Prokopi M, Kousparou CA, Epenetos AA (2015) The secret role of microRNAs in cancer stem cell development and potential therapy: a Notch-pathway approach. Front Oncol 4: 389.
- Zhao L, Liu W, Xiao J, Cao B (2015) The role of exosomes and “exosomal shuttle microRNA” in tumorigenesis and drug resistance. Cancer Lett 356(2): 339-346.
- Schorey JS, Cheng Y, Singh PP, Smith VL (2015) Exosomes and other extracellular vesicles in host-pathogen interactions. EMBO Rep 16(1): 24-43.
- Ren J, He W, Zheng L, Duan H (2016) From structures to functions: insights into exosomes as promising drug delivery vehicles. Biomaterials science 4(6): 910-921.
- Hood JL, Scott MJ, Wickline SA (2014) Maximizing exosome colloidal stability following electroporation. Anal Biochem 448: 41-49.
- Haney MJ, Klyachko NL, Zhao Y, Gupta R, Plotnikova EG, et al. (2015) Exosomes as drug delivery vehicles for Parkinson's disease therapy. J Controlled Release 207: 18-30.
- Keller A, Mussbach F, Breitling R, Hemmerich P, Schaefer B, et al. (2013) Relationships between cargo, cell penetrating peptides and cell type for uptake of non-covalent complexes into live cells. Pharmaceuticals 6(2): 184-203.
- Sun D, Zhuang X, Grizzle W, Miller D, Zhang H (2011) A novel nanoparticle drug delivery system: The anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol Ther 18(9): 1606-1614.
- Zhuang X, Xiang X, Grizzle W, Sun D, Zhang S, et al. (2011) Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain. Molecular Therapy 19(10): 1769-1779.
- Jang SC, Kim OY, Yoon CM, Choi D, Roh T, et al. (2013) Bioinspired exosome-mimetic nanovesicles for targeted delivery of chemotherapeutics to malignant tumors. ACS nano 7(9): 7698-7710.
- Tian Y, Li S, Song J, Ji T, Zhu M, et al. (2014) A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 35(7): 2383-2390.
- Rani S, Ryan AE, Griffin MD, Ritter T (2015) Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications. Mol Ther 23(5): 812-823.
- Yang T, Martin P, Fogarty B, Brown A, Schurman K, et al. (2015) Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio rerio. Pharm Res 32(6): 2003-2014.
- Pascucci L, Coccè V, Bonomi A, Ami D, Ceccarelli P, et al. (2014) Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Controlled Release 192: 262-270.
- Lee J, Kim J, Jeong M, Lee H, Goh U, et al. (2015) Liposome-based engineering of cells to package hydrophobic compounds in membrane vesicles for tumor penetration. Nano letters 15(5): 2938-2944.
- Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, et al. (2011) Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol 29(4): 341-345.
- Wahlgren J, Karlson TDL, Brisslert M, Vaziri Sani F, Telemo E, et al. (2012) Plasma exosomes can deliver exogenous short interfering RNA to monocytes and lymphocytes. Nucleic Acids Res 40(17): e130-e130.
- Shtam TA, Kovalev RA, Varfolomeeva EY, Makarov EM, Kil YV, et al. (2013) Exosomes are natural carriers of exogenous siRNA to human cells in vitro. Cell Commun Signal 11(1): 88.
- Pan Q, Ramakrishnaiah V, Henry S, Fouraschen S, Kwekkeboom J, et al. (2012) Hepatic cell-to-cell transmission of small silencing RNA can extend the therapeutic reach of RNA interference (RNAi). Gut 61(9): 1330-1339.
- Liu Y, Zhao L, Li D, Yin Y, Zhang C, et al. (2013) Microvesicle-delivery miR-150 promotes tumorigenesis by up-regulating VEGF, and the neutralization of miR-150 attenuate tumor development. Protein & cell 4(12): 932-941.
- Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, et al. (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9(6): 654-659.
- Kosaka N, Iguchi H, Yoshioka Y, Hagiwara K, Takeshita F, et al. (2012) Competitive interactions of cancer cells and normal cells via secretory microRNAs. J Biol Chem 287(2): 1397-1405.
- Ohno S, Takanashi M, Sudo K, Ueda S, Ishikawa A, et al. (2013) Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Molecular Therapy 21(1): 185-191.
- Schorey JS, Bhatnagar S (2008) Exosome function: from tumor immunology to pathogen biology. Traffic 9(6): 871-881.






























