A New Look at the Malignant Growth in Regenerating Animal Tissue and Cells
Karel Sláma*
Laboratory of Insect Physiology, Czech Republic, Europe
Submission:March 12, 2019; Published: March 25, 2019
*Corresponding author: Karel Slama, Laboratory of Insect Physiology, Czech Republic, Europe
How to cite this article: Karel Slama. A New Look at the Malignant Growth in Regenerating Animal Tissue and Cells. Nov Appro Drug Des Dev 2019; 5(1): 555654. DOI: 10.19080/NAPDD.2019.04.555654
Abstract
Regeneration of the naturally dead or artificially removed insect epidermal cells is executed by induction of mitotic divisions at the margin of the wound. The regenerated cells unite with the old epithelium and restore the tissue integrity, which gives a signal for arresting further mitotic divisions. It was found that proliferaton of insect epidermal cells required the presence of polyhydroxylated derivatives of 6-keto, 7-dehydrochoolesterol (ecdysone, ecdysteroids, Ecd). These compounds were accidentally discovered in the search for insect moulting hormone. Extensive investigations revealed, however, that Ecd were not animal hormones. According toi their wide distribution in plants and strong anabolic, growth promoting effects in domestic animals and humans, the biological status of Ecd has been identified as the previously neglected and incorrectly identified vitamin D1.
In contrast to vitamins D2 and D3, vitamin D1 is partly soluble in water, occurs in plant food and is biologically active without UV-radiation. In higher animals and humans, the drugs containing vitamin D1 produce a plethora of beneficial pharmacological effects. The deficiency of vitamin D1 during cell regeneration causes formation of malignant syncytia with defective cell membranes and constantly dividing nuclei, which is characteristic for malignant tumors. Plants containing vitamin D1 are considered to be the new, naturally occurring drugs exhibiting rejuvenating, growth promoting, anti-malignant and other beneficial pharmacological effects. A new hypothesis suggesting that the malignant growth may be caused by the deficiency of vitamin D1 in human blood has been proposed
Keywords: Ecdysteroid; Cell Regeneration; Mitotic Divisions; Polynuclear Cell
Introduction
Malignant growth is not a regular illness, it is deficiency in regulation of tissue and cell regeneration. In principle, natural death of the outlived cells stimulates mitotic divisions among the neighbouring cells. The dividing cells regenerating the missing part need essential compounds, vitamins for construction of the perfect structural and functional properties of the newly regenerated cells. The well-developed hydro/lipophilic cell membranes are responsible for healing of the wound and restoration tissue integrity, which stops further cell divisions. Investigations on regeneration of insect epidrmal cells revealed that deficiency of essential vitamin (vitamin D1) prevented restoration of tissue integrity [1]. The regenerating cells formed defective cell membranes, produced syncytia of malignant, tumor-like structures containing endomitotically dividing nuclei.
The neglected vitamin D1
There are still some old problems in the human health care that are difficult to resolve. An example are the malignant tumors of unknown origin, which are treated only by chemotherapy preventing all kinds of regeneration associated with the mitotic divisions. Under physiological conditions, somatic growth associated with cell proliferation depends the antirachitic (preventing ricket) vitamin D. As has been already mentioned above, the recent study on regeneration assumes that the problems of growth associated with the malignant tumors may be related to a 100-years old story of the neglected vitamin D1. Pioneering studies on the antirachitic vitamin D suggested some relationships with the exposure of tested materials to sunlight [2].
It was also found that chemical structure of the vitamin D should be somehow related to that of 7-dehydrocholesterol, which could be converted in chemical laboratory from cholesterol by UV radiation [2, 3]. After 1930, scientists isolated and identified compounds related to 7-dehydrocholesterol (Figure 1A), from pig, rat and human skin, as well as from animal sources, such as whole milk or liver [4,5]. The compounds were isolated and named vitamin D2 (ergocalciferol) and D3 (cholecalciferol) (Figure 1B) and [6].
Vitamins D2 and D3 need to be activated by UV-radiation of the skin [2,3]. Like other derivatives of cholesterol, these vitamins were defined as the purely lipid soluble animal sterols [4,6]. Investigations of the vitamin D were thus directed to the lipid soluble animal fats, like milk or codfish oils. The polar, partly water soluble sterolic fractions were ignored and disposed of without being tested for antirachitic properties of vitamin D. The plants were also excluded from the search, because 7-dehydrocholesterol was considered to be exclusively animal sterol (zoosterol) [3,4,6].This old definition of chemical and biological status of vitamin D, as the purely lipid soluble animal sterol activated by UV-radiation, persisted practically until this time. The commercially available preparations of vitamin D3 are Vigantol and the product of D3 hydroxylation in liver or kidneys is known as the vitamin D3-triol (Rocaltrol) [6].
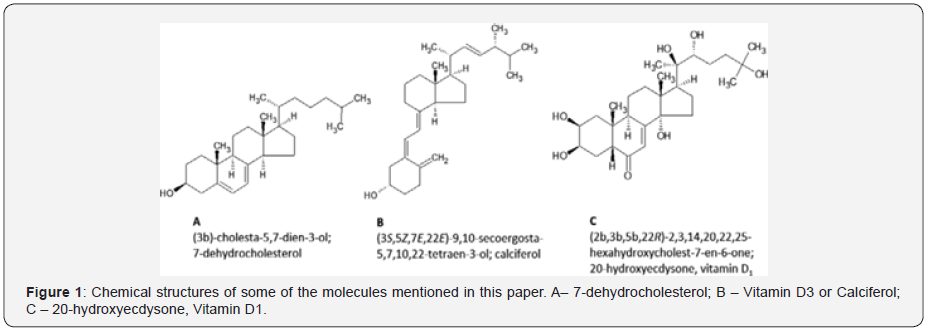
Ecdysteroids (ECD; here vitamin D1)
In the old times, vitamin D1 was identified as UV-irradiation product of vitamin D2 [5,7]. Due to rather low technical stage of mass spectrometry in the 1990ies, chemical structure of the vitamin D1 has never been correctly identified and used in pharmacology [6,7]. A great progress in triterpenoid chemistry occurred in 1965, when german chemist, P. Karlson, accidentally discovered in his search for insect moulting hormones, a polyhydroxylated derivative of 6-keto, 7-dehydrocholessterol, which was named ecdysone [7,8]. From 500 kg of silkworm pupae he obtained 25 mg of crystalline ecdysone (ecdysis means shedding of the old insect cuticle). A sample of ecdysone was sent to Prof. C. M. Williams of Harvard University, who was she respected authority in insect hormones.
He found that ecdysone stimulated secretion of a new cuticle in diapausing pupae of Cecropia silkworms. This effect was previously ascribed to the hormone of the insect prothoracic gland [9,10]. Based on this information, Karlson [8] defined the biological status of ecdysone as the prothoracic gland hormone [11-20]. Ironically, the respected vitamin expert, Prof. P. Kaslson [9] and other outstanding chemists (ecdysonists, [11,18-21], were unable to figure out close structural similarities between 7-hydroxycholesterol of ecdysone and vitamin D.
Soon after disclosure of crystallographic structure of ecdysone by Karlson [7,8], phytochemists in different countries promptly announced surprising findings of ecdysone, the “insect moulting hormone”, in a number of various plants [13-21], reviews [11,12,18-21]. Certain plants, like a fern Polypodoum vulgare, contained in 1 g of rhizomes as much ecdysone as did 500 kg of the silkworm pupae used by Karlson [7]. The distribution of ecdysone and similar compounds (Ecd) in plants was intensively studied [13-17]. The most abundant compound both in animals and plants was 20-hydroxyecdysone containing 6 hydroxyl groups, also known as ecdysterone [12]. Extensive search for new positions of hydroxyl groups in these “insect hormones” became a fashionable topic of phytochemistry for several decades [11,12,18-21]. Scientists of western countries are engaged in identification of the ecdysteroid receptor [22,23], while Soviet and Ukrainian scientists described a plethora of practically more important anabolic growth effects, or the associated rejuvenating, tonic, neurogenic, immunogenic and other beneficial pharmacological effects of Ecd in domestic animals and the human body [24-38].
Many recent authors still adhere strictly to the old concept of Karlson [8] and Williams [10], based on the hormone of insect prothoracic gland. Little is known that C. M.Williams himself refrained from the original hypothesis, when he found that the disinte1grating pupal intestine represented a large depot of the sequestered Ecd [25]. This important confession of Williams remained mostly unnoticed, because his influential students forced him to publish the results in almost unknown peripheral journal [25]. In a similar way, alternative views on Ecd action were also ignored. Conclusion that Ecd could represent a reserve material for growth in plants [16], was considered insane. In insects, biological status of Ecd was determined as “ the homeostatic factors synchronizing the developmental cycles [26]. In 1993, biological status of Ecd was defined as an essential D-vitamin, with great potential use in pharmacology and medicine [27]. In general, Ecd were not an insect hormone and the actual raison dˇ etre of these compounds in plants was not physiological activity of any of the known phytohormones [28].
Ecdysteroids (Ecd) are vitamins or hormones?
Investigations in insects showed that in spite of the widespread belief Ecd were not the prothoracic gland (PG) hormone [1,12,33-37]. This statement is corroborated by the following facts:
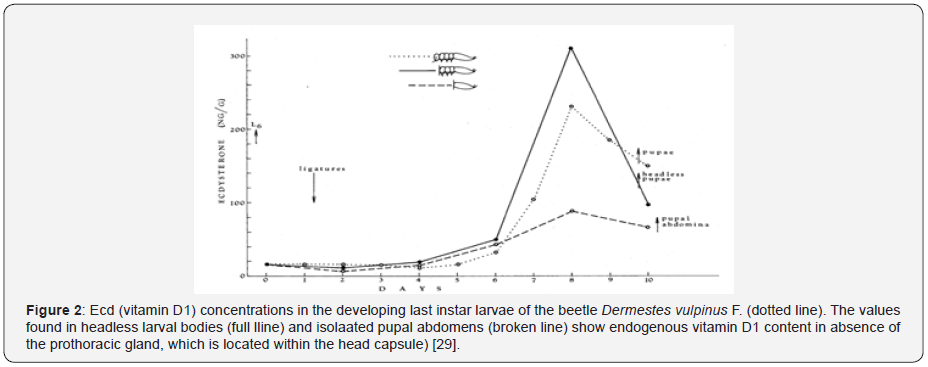
a) Severa groups of insects (Coleoptera, Hymenoptera) show autonomic development [29], manifested by sequence of the moulting cycles and endogenous Ecd peaks in complete absence of the PG (Figure 2) [30].
b) Larvae of the wax moth (Galleria mellonella) develop regularly into pupae and adults in spite of the experimentally removed PG [31,32].
c) Endogenous peaks in Ecd concentration have their origin in disintegrating pupal intestine [25], and in other disintegrating tissues [33].
d) The prepupal Ecd peaks in Lepidoptera exactly coincide with total disintegration of the PG [34].
e) The correct physiological function of PG depends on hypermetabolic production of water, not on the production of a moulting hormone [34]. In conclusion, the dietary origin of Ecd from plants (not a biosynthesis) and the release of Ecd from various peripheral, not endocrine tissues [33], represent serious breach of the definition of animal hormone [34-37].
Another acute terminological conflict of Ecd is related to stimulation of insect ecdysis. The neuromuscular act of shedding of the old insect cuticle (ecdysis) does not correspond with the old terminology of Karlson and other chemists [11,35,36]. The injections of exogenous ecdysone cause precocious secretion of the new cuticle followed by a dose-dependent inhibition of ecdysis [26,37]. The terms of science should correspond with the facts in nature. Thus, in order to restrain the terminological misunderstandings, it has been recently proposed to call the polyhydroxylated derivatives of 6-keto, 7-dehydrocholesterol (Ecd) by the generic name of the previously neglected vitamin D1 [1,24,35-38]. There are apparent similarities between the anabolic, vitamin D-like effects of the vitamin D1 in domestic animals and in the human body [24,38]. The old view of prothoracic gland hormone hinders the potential study and use of the perspective beneficial drug (Vitamin D1) in pharmacology and medicine.
Practical testing of physiologically active drugs found in insects is well substantiated by copious data on similarities between insect and human physiological systems [27,35,36,38]. The similarities in this respect apply to specific role of vitamin E in insect reproduction [39]. Important arguments suggesting that Ecd did not represent insect hormones but essential vitamins, received by animal body from plants, were actually published a long time ago, in 1993 [27]. The inverse proportionality between endogenous Ecd peaks and minimum total body metabolism [40], provided presumptive evidence that vitamin D1 was received from plants or intestinal symbionts during the larval feeding period and later it was reutilised from disintegrating larval tissues [1,33] without its de novo biosynthesis.
The effects of vitamin D1 on regeneration of insect tissues
In mammals, somatic growth involved enlargement of internal endoskeleton composed chiefly from bones and muscles. In insects, however, somatic growth deepens on the enlargement of an exoskeleton composed from epidermal epithelium and integumental cuticular cover. Internal transformations of tissues and organs during nonfeeding stages of insect metamorphosis (histolysis/histogenesis) [12], are characterised by endogenous peaks in the content of vitamin D1 (Ecd) in the body [26,27,30,33,35,37,41], see Figure 2. As has been already mentioned, the endogenous peaks in vitamin D1 content coincide exactly with the minimum metabolic intensity [40], which are synchronized with the periods of maximum proliferation of cells in the newly developing organs (histogenesis). Reutilization of vitamin D1 during the nonfeeding period supports earlier conclusions [41], that insects, and probably other animals as well, do not synthesize the steroid nucleus de novo . Sterol derivatives are abundant and can be easily obtained from plants or intestinal flora, without the burden of steroidogenic enzymes in the animal cells.
Serious disadvantage of this evolutionary adaptgation is that a deficiency of the essential vitamin may cause serious derrangements of growth and metabolism [41]. The problem is less acute in insects, which can reutilise the structurally bound, insoluble vitamin D1 from the histolysing tissues [33]. However, so far, I was unable to find out whether simialr reutilisation of sterol for synthesis of vitamin D could exist also in the human body.
Recently there appeared a detailed analysis of the effects of vitamin D1 and juvenile hormone on regeneratioin of epidermal cells in larvae and pupae of the tobacco hornworm (Manduca sexta) [1]. During the endogenous peak of vitamin D1, the excised patches of epidermal cells regenerated a homochronic (identical) epidermal structures. The disconnected epidermal cells on the margin of the wound started to divide mitotically until the cell-to-cell integrity of the regenerating cells became restored. The living cells communicated with each other and immediately arrested the mitotic divisions as soon as the tissue integrity was re-established. The presence of vitamin D1 in haemolymph during regeneration was the most important factor for giving the signal for tissue integrity, required for arresting further mitotic divisions [1]. The excised patches of epidermal cells, forced to regenerate in absence of the vitamin D1 (ligatured Dauer-larvae, diapausing pupae), showed irregular, tumor like regenerates, with the defective cellular membranes and polynucleated syncytia [1,35-37].
The described results show that the presence of vitamin D1 is essential for cell proliferation, which also explains the evolutionary coordination of cell proliferation with the endogenous peaks in concentration of the vitamin [1,33,35- 37,40]. Application of juvenile hormone analogue caused the regenerating cells to develop morphological structures of the previous ontogenetic stage (larval pataches on pupal body, known as metathetely) [1]. Alternatively, the application of vitamin D1 caused the regenerating cells to develop prematurely the structural characters of the future ontogenetic stage (pupal regeneration patches on larval body, known as prothetely). In all cases, however, the regenerating cells required for reaching with vitamin D1 the tissue integrity [1].
The sources of vitamin D1
Like other essential vitamins, vitamin D1 occurs in a number of plant species [1,12,18-2]. In addition to ferns [14], it can be found in vascular plants of different families, especially in Asteraceae [1,16 -21,42]. Certain plants contain relatively large amounts of vitamin D1, usually as growth reserves accumulated in winter roots and seeds (up to 3% of dry mass). Biosynthesis of the vitamin occurs in green leaves during the vegetation season (0.005 to 0.01%) [1,12,21,35,37]. Larger or smaller quantities of vitamin D1 were found in mushrooms [43], crustaceans [43], insects [8,11,12,20,23,30,38], marine animals (Pycnogonids) [44], Japanese Quails [45,46], mice [20,24,47,48], domestic animals [24,38,48-50] and humans [24,38,49, 51-53]. In the case of phytophagous animals and the humans, the vitamin has been certainly obtained from the diet, similarly like in the case of other essential vitamins.
Obviously, newborn babies should receive the growth promoting, antirachitic vitamin D1 from milk. In cattle, which is the main commercial source of milk and butter, the vitamin is obtained from fodder plants and tentatively also from extensively developed intestinal symbiotic flora. The gap in our knowledge concerning the distribution of vitamin D1 in the human body has been related to the misleading belief into the insect hormone. Nevertheless, the first alternative explanation of biological status of Ecd, e. g. reserve growth factors of plants, was publůished as early as before 40 years [16]. Moreover, convincing evidence for the vitaminic nature of Ecd was provided in 1993 [27,29,35,36].
In spite of the above alternative data, the old concept based on insect moulting hormone is still advocated by major part of the main stream authors [18-21,23,38]. Exceptionally, prothoracic glands have been considered as the sole biosynthetic organ of Ecd in insects [54]. Moreover, extensive studies on ecdysteroid receptor [22,23], have dominated in the literature. Phytochemists speculate about the distinction of phytoecdysteroids and zooecdysteroids [21], unaware that insects do not synthesize sterol [41], which is received from plants. Before discovery of Ecd in plants [13,14,17], actually the derivatives of 7-dehydrocholesterol, they believed that cholesterol was exclusively a zoosterol. The old dogmatic view that cholesterol was not a phytosterol has been challengesd when the polyhydroxylated derivative of 7-dehydrocholesterol (vitamin D1 ,Ecd, zoosterol?) was found in the plant Leuzea carthamoides 700-fold in excess over the true phytosterols, ergosterol and β-sitosterol. The indistinctive presence of cholesterol was due to preferential hydroxylation of cholesterol which disappeared from the pool of free lipid soluble phytosterols [55].
In addition to plants containing relatively substantial amounts of vitamin D1, like the fern Polypodium vulgare L., the daisies Leuzea carthamoides (Willd.) and Serratula coronata L. as well as the mint, Ajuga turkestanica [11,12,14,18-21,38], vitamin D1 can be found as a common constituent of vegetable foods, like Quinoa (Chenopodium quinoa; 0.03% [21]; spinach (Spinacia oleracea L., 0.01% [42], or Suma root (Hebanthe eriantha, 0.66% [42]. A more recent pharmacological preparation, SERPISTEN registered in Russia, used purified extracts of vitamin D1 from Serratula coronata L. [18]. The preparation replaced earlier anabolic and rejuvenating preparation ECDISTEN, registered in former Soviet Union already in 1980 [18-21,24,38]. In conclusion, the most important pharmacological properties of vitamin D1 are the vitamin D-like, anabolic growth effects, stimulation of muscular functions, removal of antifatigue, improvement of physical conditions and other beneficial pharmacological effects of an ideal natural drug [24,38,52].
Hypervitaminosis D1,vitamins D2 and D3
With the discovery of insect juvenile hormone analogues in plants [56], we speculated that by using these compounds, plants evolved a self defence mechanism against insect herbivores. The presence of large amounts of Ecd in plants [12,16,19] could be associated with severe pathophysiological syndromes of ”hyperecdysonism”, which are quite common after Ecd injections [37,57]. The real ecological significance of the hypothesis has been criticised, although some Ecd-containing plants were found to be deterrent to insect feeders [58].
The possibility of Ecd-mediated insect-plant interactions were studied in more detail by entomocenotic analysis of insects feeding on plants containing rather high Ecd content (Leuzea carthamoides) [59]. It appeared that certain insects were well adapted to feed on this plůant. Especially, polyphagous Noctuid larvae appeared to be virtually resistant against the dietary supply of the vitamin D1. The resistance against hyperecdysonism was practically investigated in larvae of Manduca sexta, which tolerated injections of up to 1000 μg of vitamin D1 per g of larval mass. In contrast to this, however, the nonfeeding pupae of this species, unable to excrete, suffered from serious hypervitaminosis syndromes after injections of as little as 2 μg [37].
Obviously, detoxication of large amounts of the dietary vitamin D1 was achieved by its excretion out of the body. In most instances, these injections result into complete mortality, due to disturbances of the moulting cycles [36]. The premature, out of the regular schedule induction of new cuticle is connected with profound neuromuscular paralysis, decreased metabolism and inhibited ecdysis [12,35-37 ]. We are all consuming vitamin D1 with the food, for it is contained in more than 200 different species of plants [11,12,16,19,24,38,42,48]. Possible consequences of D1 hypervitaminosis in the human body are unknown. There has never been enough of the drug (Ecd) required to produce hypervitaminosis in large animals [47,49,60].
The existence of D1 hypervitaminosis in insects provides evidence that this compound is a real vitamin, not a hormone. Just like in the case of other vitamins, its excess gets restrained by excretion. Its deficiency, however, cannot be repaired by biosynthesis. The true animal hormone, like the juvenile hormone and a number of its bio analogues [60], show qualitatively different mode of action. It acts according to the rule all-ornone, because it is used to activate morphogenetic instructions coded in the genome. A minimum effective concentration, as well as a 500000-fold overshooting of the dose, produce the same biological response [60].
Insect genome (Derosophila) contains 37% of the homologous genes also functioning in the human genome [61]. This may explain an apparent similarity in execution of elementary physiological systems both in insect and human body [62]. These similarities led me to conclude [1], that the role of vitamin D1 in prevention of the malignant growth in insects could be valid for human systems as well. Inability of the contemporary medicine to elucidate the cause of the malignant growth suggests that there may be something wrong in understanding the regulation of growth among tissue and cell growth. I feel that the error occurred already a hundred years ago, during identification of antirachitic vitamin D. The chemists knew that the active compound could be related to 7-dehydrocholesterol [2-4]. They were searching for a highly nonpolar, purely lipid soluble animal sterol.
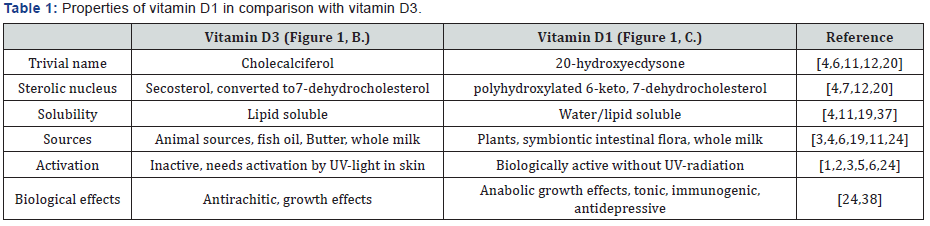
This explains why they ended up with the biologically little active vitamins D2 and D3, while missing the existence of a partly water soluble vitamin D1 occurring in plants. General properties of vitamins D2 , D3 and D1 have been summrised in (Table I). The data show substantial differences in solubility, occurrence, biologival activity and necessity of UV-radiation. Vitamin D1 has a sweet taste due to the sugar-like presence of 6 or 7 hydroxylic groups. A recipe for rejuvenating drug recommends extraction of the roots or seeds of Leuzea carthamoides in 50% alcohol.
The D1 avitaminosis theory of malignant growth
It is difficult to argue about the incorrect assignment of vitamins D2 and D3.They are known for 100 years and their description can be found in all textbooks of medicine. Recent study of regeneration and malignant growth in insects [1], however, throws some doubts about their biological activity and the necessity of UV-light for their activation. The new concept comes out with an assumption that the true, growth promoting vitamin D is the polyhydroxylated, 6-keto, 7-dehydrocholesterol (neglected vitamin D1, which was later discovered in the search for insect moulting hormone, ecdysone.
In insects, intensive periods of cell proliferation (histolysis /histogenesis) are always synchronized with increased endogenous concentrations of vitamin D1 (Figure 2). Evidently, the dividing insect cells need the vitamin for the construction of morphologically perfect daughter cells. The determined hydro/lipophilic structure of cell membranes is essential for restoration of tissue integrity that is needed for arresting further mitotic divisions. The dividing cells of insects and probably all animals require vitamin D1 for termination of regeneration and restoration of uninjured tissue integrity. In absence of vitamin D1 in the blood, the regenerating cells produced polynucleated, malignant syncytia with defective cell membranes and endomitotically dividing nuclei [1].
Due to the mentioned similarity between insect and human physiological systems [61,62], it has been reasonably assumed that a cause of the malignant growth in the human body could be also related to deficiency of the vitamin D1.This fact can be supported by previous evidence about the vitamin D-like, antirachitic, growth promoting effects of vitamin D1 (Ecd) in domestic animals and in the human body [11, 12,16,21,24,35,38,45,50,52 ].
There exist numbers of theories how to stop malignant growth by the destruction of polynuclear tumor cells. These structures are not a cause but consequences of the malignant growth. It may be more important to prevent the normal regenerating cells from the formation of malignant tumors [1]. In analogy with insects, it might be perhaps better to supply the dividing cells with the essential vitamin D1. Unfortunately, the concept of vitamin D1 deficiency comes too late, after many people suffered from oncological problems. It was due to notorious belief that Ecd (vitamin D1) were prothoracic gland hormone of insects [7,8]. Like many other theories for explaining cancer, the proposed theory of vitamin D1 deficiency may also turn out not to be true. It neds approval by clinical assays. However, it may be at least one of the few possibilities for alternative substitution of chemotherapy by stimulation of regular cell growth.
Vitamin D1 (Ecd) is a well-documented anabolic, growth promoting natural drug, with a number of beneficial pharmacological effects [24,38]. The scope of these effects ranges from stimulation of growth of the whole body in different animals [1,11,12,23,35,38], through enhanced milk and meat production in cattle [24], increased muscular performance of human athletes [49], ending with antimalignant growth effects [1,63]. This shows that natural or synthetic drugs, containing vitamin D1 as the active ingredient, could constitute an ideal drug with beneficial pharmacological properties. The problem is that testing of the drug on the human body requires to use relatively large quantities of the very expensive, purified vitamin D1 (20-hydroxyecdysone). The proposed theory of vitamin D1 deficiency is the first attempt to throw some light into the origin of malignant growth.
References
- Sláma K (2019) Vitamin D1 versus ecdysteroids: Growth effects on cell regeneration and malignant growth in insects are similar to those in humans. Eur J Entomol 116: 16-32.
- Hess AF, Unger RJ (1921) The cure of infantile rickets by artificial light and by sunlight. Proc Soc Exp Biol Med 18: 298-299.
- Hess AF, Weinstock M, Heelman FD (1925) The antirachitic value of irradiated phytosterol and cholesterol. J Biol Chem 63: 305-309.
- Windaus AF, Bock F (1937) Über das provitamin aus dem Sterin der Schweineschwarte. Hoppe-Seyler’s Z. Physiol Chem 245(3-4): 168-170.
- Windaus AF, Luttringhaus V, Deppe M (1931) Über das komplizierte Vitamin D1. Justus Liebig Anal Chem 489: 262-269.
- Wolf G (2004) The discovery of Vitamin D: The contribution of Adolf Windaus. J Nutrit 134(6): 1299-1302.
- Karlson P, Hoffmeister H, Hummel P, Hocke P, Spitteler P (1965) Zur Chemie der Ecdysons. VI. Reaktionen des Ecdysonmolekü Chem Ber 98(7): 2394-2402.
- Karlson P (1996) Ecdyson, das Häutungshormon der Insekten. Naturwissenschaften 53: 445-453.
- Karlson P (1981) Vitamin D1 and D2. Trends Biochem Sci 6: 29-30.
- Williams CM (1952) Physiology of insect diapause. IV. The brain and prothoracic glands as an endocrine system of Cecropia Biol Bull 103(1): 120-138.
- Koolman J (1989) Ecdysone-From Chemistry to Mode of Action. Georg Thieme Veralag, Stuttgart, New York, USA, pp. 482.
- Sláma K, Romaňuk M, Šorm F (1974) Insect Hormones and Bioanalogues. Wien, New York, USA, pp. 477.
- Nakanishi K, Koreeda M, Sasaki S, Chang ML, Hsu HY (1966) Insect hormones. The structure of ponasterone A, insect-moulting hormone from the leaves of Podocarpus nakaii Chem Commun 24: 915-917.
- Jizba J, Herout V, Šorm F (1967) Isolation of ecdysterone (crustecdysone) from Polypodium vulgare rhizomes. Tetrahedron Lett 18(8): 1689-1691.
- Sláma K (1969) Plants as a source of materials with insect hormone activity. Ent exp appl 12: 721-728.
- Sláma K (1979) Insect hormones and antihormones in plants. In: Rosenthal GA, Janzen DH (Edt.), Herbivores, their Interaction with Secondary Plant Metabolites. Academic Press, New York, London, pp. 673-700.
- Hikino H, Hikino Y, Nomoto K, Takemoto T (1968) Cyasterone, an insect metamorphosing substance from Cyathula capitata: Structure. Tetrahedron 24(13): 4895-4906.
- Volodin VV (2003) Phytoecdysteroids. Nauka St Petersburg, Russia, pp. 293.
- Dinan L, Harmatha J, Volodin V, Lafont R (2009) Phytoecdysteroids: diversity, biosynthesis and distribution. In: Smagghe G, (edn.): Ecdysone: Structures and Functions. Dordrecht, UK, p. 3-44.
- Lafont R, Dauphin-Villemant C, Warren JT (2011) Ecdysteroid chemistry and biochemistry. In: Gilbert L.I. (edn.): Insect Endocrinology. Elsevier BV, Amsterdam, UK, pp. 106-177.
- Kumpun S, Maria A, Crouzet S, Eurard-Todesch N, Girault JP, et al. (2011) Ecdysteroids from Chenopodium quinoa Willd, an ancient Andean crop of high nutritional value. Food Chem 125(4): 1126-1234.
- Tan A, Palli SR (2008) Ecdysone receptor isoforms play distinct roles in controlling molting and metamorphosisin the red flour beetle Tribolium castaneum. Mol Cell Endocrinology 291(1-2): 42-49.
- Gilbert LI (2012) Insect Endocrinology.UK, pp. 577.
- Sláma K, Lafont R (1995) Insect hormones-ecdysteroids: Their presence and actions in vertebrates. Eur J Entomol 92: 355-378.
- Williams CM (1987) Midgut of lepidopteran pupae is a major depot of sequestered, mobilizable ecdysteroids. Memo Inst Oswaldo Cruz 82(3): 47-49.
- Sláma K (1980) Homeostatic function of ecdysteroids in ecdysis and oviposition. Acta Entomol Bohemoslov 77: 145-168.
- Sláma K (1993) Ecdysteroids: insect hormones, plant defensive factors or human medicine? Phytoparasitica 21(1): 3-8.
- Macháčková I, Vágner M, Sláma K (1995) Comparison between the effects of 20-hydroxyecdysone and phytohormones on growth and development in plants. Eur J Entomol 92(1): 309-316.
- Sláma K ( 2015) A new look at the nature of insect juvenile hormone with particular reference to studies carried out in the Czech Republic. Eur J Entomol 112(4): 567-590.
- Delbecque JP, Sláma K (1980) Ecdysteroid Titres During Autonomous metamorphosis in a dermestid beetle. Z Naturforsch 35(11-12): 1066-1080.
- Sláma K (1983) Illusive functions of the prothoracic gland in Galleria. Acta ent bohemoslov 80: 161-176.
- Sláma K (1988) The mysterious thoracic hormonal centre in insects. In: Sehnal F, ZabzaA, Denlinger DL (Edn.) Endo-crinological Frontiers in Physiological Insect Ecology. Wroclaw Technical University Press, Wroclaw, pp. 663-675.
- Sláma K (1998) The prothoracic gland revisited. Ann Entomol Soc Am 91: 168-174.
- Sláma K, Lukáš J (2016) Hypermetabolic Conversion of Plant Oil into Water: Endothermic Biochemical Process Stimulated by Juvenile Hormone in the European Firebug, Pyrrhocoris apterus Int J Insect Sci 8: 81-93.
- Sláma K (2015) An alternative look at insect hormones. Life Excit Biol 3: 188-204.
- Sláma K (2016) Are ecdysteroids insect hormones? Atlas of Science, p. 5.
- Sláma K Zhylitskaya H (2016) Comprehensive Physiology and Toxicology of Ecdysogens: The Metabolically Activated Porphyrin-Ecdysteroid Complexes in Insects. Compar Biochem Physiol 181-182: 56–67.
- Lafont R, Dinan L (2003) Practical uses for ecdysteroids in mammals including humans an update. J Insect Sci, p. 37-37.
- Jedlička P, Cvačka J, Sláma K (2009) Juvenile hormone-stimulatedsynthesis of acyl-glycerols and vitamin E in female accessory sexual glands of the fire bug, Pyrrhocoris apterus L. Arch Insect Biochem Physiol 72(1): 48-59.
- Sláma K (1982) Inverse relationships between ecdysteroid titres and total body metabolism in insects. Z Naturforsch 37(9): 839-844.
- Svoboda JA, Thompson MJ (1985) Steroids. Comp. Insect Physiol Biochem Pharm 10: 137-175.
- Hunyadi A, Herke I, Lengyel K, Báthory M, Kele Y, et al. (2016) Ecdysteroid-containing food supplements from Cyanotis Arachnoidea on the European marketevidence for spinach product counterfeiting. Sci Rep 6: 313-322.
- Horn DHS, Fabbri S, Hampshire F, Lowe MJ (1968) Isolation of crustecdysone (20R-hydroxyecdysone) from a crayfish (Jasus lalandei, H. Milne Edwards). Biochem J 109(3): 399-406.
- Tomaschko K, Bückman D (1992) Excessive abundance and dynamics of unusual ecdysteroids in Pynogonum litorale Ström (Arthropoda, Pantopoda). Gener Compar Endocrinol 90(3): 296-305.
- Koudela K, Tenora J, Bajer J, Mathová A, Sláma K (1995) Stimulation of growth and development in Japanese quails after oral administration of ecdysteroid-containing diet. Eur J Entomol 92: 339-354.
- Sláma K, Koudela K, Tenora J, Mathová A, (1996) Insectormones in vertebrates; anabolic effects of 20-hydroxyecdysone in Japanese quail. Experientia 52(7): 702-706.
- Stopka P, Štancl J, Sláma K (199) Effect of insect hormone, 20-hydroxyecdysone on growth and reproduction in mice. Acta Soc Zool Bohem 63: 367-378.
- Smagghe G (2009) Ecdysone: Structures and Function. Dordrecht, London, pp. 583.
- Syrov VN (1984) On the mechanism of anabolic action of phytoecdysteroids. Nauch Dokl Vyssh Shk Biol Nauk 9: 37-39.
- Krátký F, Opletal L, Hejhalek J, Kuchařová S (1997) Effect of 20-hydroxyecdysone on the protein synthesis of pigs. Zivocisna Vyroba 42: 445-451.
- Koolman J, Moeller H (1986) Major helminth infections detected by RIA of serum and urine. Insect Biochem. 16(1): 287-291.
- Kholodova YD (2001) Phytoecdysteroids: biological effects, application in agriculture and complementary medicine. Ukrain Biokhim Zh 73(3): 21-29.
- Gharib B, Baswaid S, Quilici M, De Reggi M (1991) Ecdysteroid- like compounds in human urine: they can occur in the absence of any parasitic infection. Clin Chim Acta 199(2): 150-166.
- Sehnal F, Fonágy A, Akai H, Kallenborn HG (1988) Prothoracic Glands And Ecdysteroid Titre In Galleria Mellonella J Insect Physiol 34(7): 609-614.
- Stránský K, Němec V, Sláma K (1998) Lipid composition of the seeds of an ecdysteroid containing plant, Leuzea cathamoides (Willd.) DC (Asteraceae). Russ J Plant Physiol 45: 333-338.
- Sláma K, Williams CM (1965) The juvenile hormone. V. The sensitivity of the bug Pyrrhocoris apterus, to a hormonally active factor in American paper-pulp. Biol Bull 130: 235-246.
- Williams CM (1970) Hormonal interactions between plants and insects. Chem Ecol, pp. 105-132.
- Ufimtsev KG, Shirshova TI, Volodin V (2009) Ecdysteroids-Deterrents of Phytophagous Insects. Russian Academy of Sciences, p. 88.
- Zelený, Havelka J, Sláma K (1997) Hormonally mediated insect-plant relationships: Arthropod populations associated with ecdysteroid-containing plant, Leuzea carthamoides (Asteraceae). Eur J Entomol 94(2): 183-198.
- Sláma K (2013) Insect hormones: more than 50 years after the discovery of insect juvenile hormone analogues (JHA, juvenoids). Terrest Arthr Rev 6: 257-333.
- Devillers J (2013) Juvenile Hormones and Juvenoids. Modeling Biological Effects and Environmental Fate. Boca Raton, pp. 387.
- Sláma K, Santiago Blay JA (2017) Terrestrial Insects with Tracheae Breath by Actively Regulating Ventilatory Movements: Physiological Similarities to Humans. The Excitement of Biology 5: 4-70.
- Sláma K (2018) Pharmacological preparation, its use and determination of the risk of malignant tumours. Czech Patent Application, pp. 719.






























