Drug Synthesis Using Ionic Liquids Through Green Approch
Amit Gosar1, Sayyed Hussain2, Tabrez Shaikh1 and Amit Joglekar1
1Department ARD, Indoco Remedies Ltd, India
1Department of Chemistry, Sir Sayyed College, India
Submission:January 25, 2019; Published: March 20, 2019
*Corresponding author: Tabrez Shaikh, Department of Chemistry ,Sir Sayyed College, India
How to cite this article: Amit G, Sayyed H, Tabrez S, Amit J. Drug Synthesis Using Ionic Liquids Through Green Approch. Nov Appro Drug Des Dev 2019; 5(1): 555653. DOI: 10.19080/NAPDD.2019.04.555653
Abstract
Environmental pollution is majorly caused by the chemical and pharmaceutical industries which have increased multifold time in the last several decades. It is the responsibility of science to design the process which will generate the less hazardous chemicals and much environmentally friendly waste. Ionic liquid finds a source of hope in reducing the organic solvent waste in coming years. These Ionic liquids may replace the organic solvents which are hazardous, toxic and non-environmentally friendly used in the synthesis of chemical compounds. This review article describes the Introduction to Ionic liquids, their types and classification, the advantages of their use in replacement of organic solvent in the synthesis of chemicals compounds and drug substances.
Keywords: Ionic Liquids; Synthesis; Properties; Application; Environmental; Pollutants; Chemistry; Recycling
Introduction
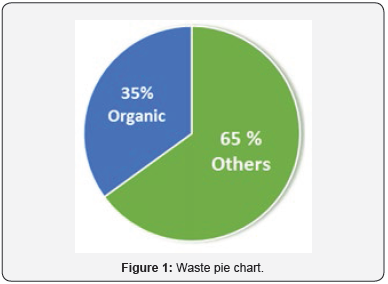
Waste is any unwanted material in all the forms that can cause harm by being inhaled, swallowed, and absorbed through the skin or damage the environmental life cycle. The major contribution to environmental waste is organic chemicals which come from various industries. Pharmaceutical industries comprise the largest source of organic pollutants among all the industries while organic solvent waste is major waste which is expelled by these industries. As the world is facing serious environmental challenges, many environmental protection agencies suggested that green chemistry should be used by chemical and pharmaceutical industries in order to reduce toxic, undesired waste, and environmental waste. If the processes can be implemented right, green chemistry can afford to reduce waste product and decrease the resource consumption. Presently the organic waste is the major waste generated in all the manufacturing industries which compromise upto 35 % (Figure 1) of total waste [1-2].
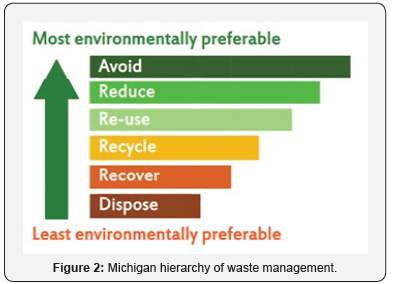
Looking at the Michigan hierarchy waste management chart (Figure 2), avoiding and reducing the amount of chemical waste generated is more preferable and cost-effective when compared to adopting waste management practices such as reducing, recycling, recovering and disposing to the environment. Considering this, the use of Ionic liquid in the manufacturing of pharmaceutical was incorporated in the 1990s in replace of conventional organic solvents. Thus ionic liquids began to be considered as environmentally benign compared to organic solvent [3].
Ionic liquids:
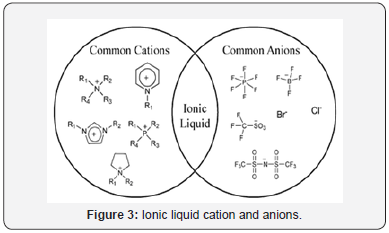
What are ionic liquids (ILs)? The very answer is in this question only. Thus Ionic liquids are liquids that consist ion of an organic cation and organic or inorganic polyatomic anion (Figure 3). Since there are many known and potential cations and anions, the potential number of ionic liquids compared to organic solvents is very huge. Thus discovering a new ionic liquid is easy. The ion liquid discovery is attributed to Paul Walden, who made the ionic liquid and studied their physical-chemical properties. These ionic liquids have a very vast application, but for our subject, it is limited to its use in synthesis in replace of organic solvents which are hazardous, toxic and not environmental friendly [4].
Cations
The ionic liquid cation generally consists of an organic structure with positively charged. The most common cations in ionic liquids are nitrogen or phosphorous containing organic ions. Physical and chemical properties of ionic liquids are influenced by the cation present in them.
Anions
The ionic liquid anion generally consists of weakly basic organic or inorganic compounds which are negatively charged. The most common anion in ionic liquids are acetate, nitrate, borate or sulphate ions.
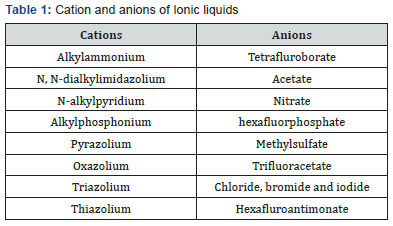
Some examples of cation and anion present in ionic liquids are given in (Table 1).
Properties of Ionic Liquids
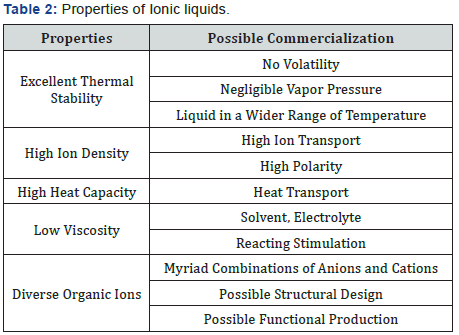
Properties of Ionic liquid such as melting point, viscosity, and solubility of materials depends on the substituents on the organic component and by the counterion. Their physical and chemical properties can be adjusted by the variation of ions. The fine adjustment of properties is possible by the variation of the length and branching of the alkyl groups incorporated into the cation. Many ionic liquids have even been developed for specific synthetic problems. For this reason, ionic liquids have been termed “designer solvents” [5] (Table 2).
Melting point
As Ionic liquids are used in replace of organic solvents which are liquid at room temperature, hence their melting point should be that of water in order to work with them at room temperature. The magnitude of the melting point is found related to the structure and composition of ionic liquids. Hence, selecting cation and anion for ionic liquid determines the melting point of ionic liquids.
Density
Cation and anion present in ionic liquid decide its density. Practically the density observed of ionic liquids are between 1.0 to 1.35 gcm-3. The bulkiness of organic anion present in ionic liquid determines the density of ionic liquids. Density tends to decrease with an increase in bulkiness.
Thermal stability
Solvent use in the synthesis should be thermally stable at all the working temperature. Ionic liquids are found to be much stable at or above 400°C when compared to organic solvents. Anion part of ionic liquid contributes greatly toward the thermostability of liquids as compared to cations. Hydrophilicity of anion decrease the thermal stability of ionic liquids.
Viscosity
Ionic liquid should have mid-viscosity when compared to organic solvents. It should be neither highly viscous nor low viscous which make unable to handle it or hard to mix with the starting material for synthesis.
Volatility
One major problem with organic solvent is that they are highly volatile, which make them to reproduce the process which can be overcome by the use of Ionic liquids, as most of these are non-volatile hence repeatability of the process is very high.
Synthesis of Ionic Liquids
The synthesis of ionic liquids can be described in two steps.
1. The Formation of the Desired Cation. The desired cation can be synthesized either by the protonation of the amine by an acid or through quaternization reactions of the amine with a haloalkane and heating the mixture.
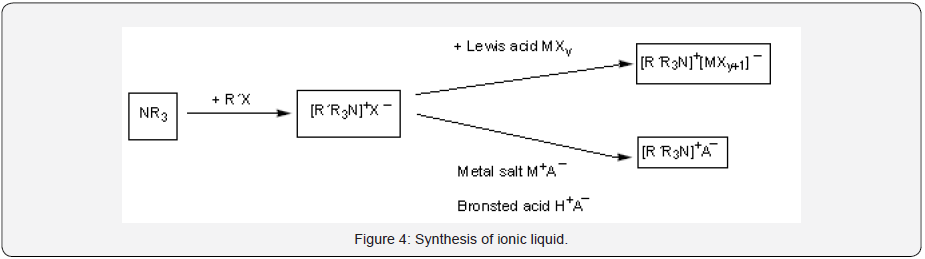
2. Anion Exchange. Anion exchange reactions can be carried out by treatment of halide salts with Lewis acids to form Lewis acid-based ionic liquids or by anion metathesis. As ionic liquid is formed by combination of two or more ions, Due to combination of two or more ion there are many possiblity of Ionic liquids can be synthesied and designed [6] (Figure 4).
Advantages Of Ionic Liquid
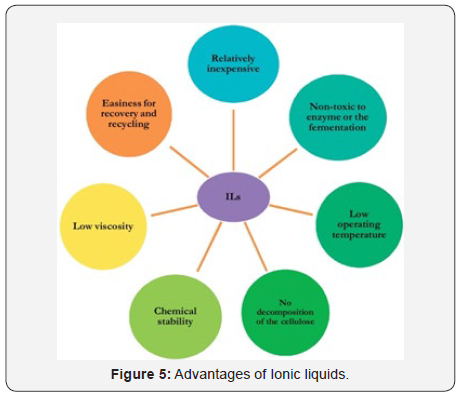
Ionic liquids (ILs) have very good properties as a reaction medium for chemical reactions, generally, they are non-volatile, non-flammable, have low toxicity and good solubility for many organic and inorganic materials. QSAR toxicity assessment of their structure confirms the nontoxic nature of most of the ionic liquids. They can easily adapt the process scale without any major changes in the process (Figure 5).
Application of Ionic Liquid
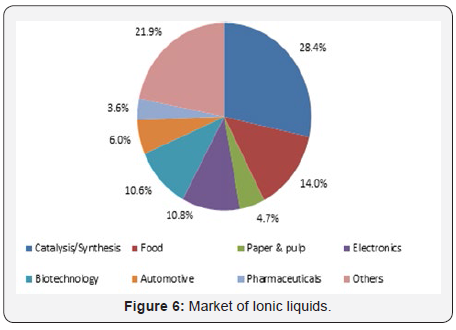
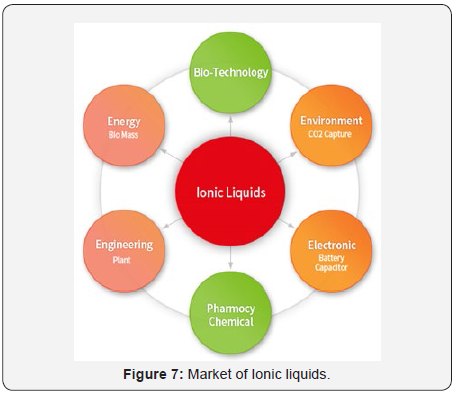
Ionic liquids find very vast and versatile application as compared to organic solvents (Figure 6&7). Some of these applications are illustrated and discuss as below [7-8].
1. Due to versatile properties of ionic liquids, they can be used as solvents in chemical and pharmaceutical industry.
2. Ionic liquids find good application in separation technology for metal extraction for both radioactive and rare earth metals.
3. Ionic liquids can be used for desulfurization and carbon dioxide capture.
4. Another use of ionic liquids includes electrolyte media in the condenser, secondary cell, fuel cell, and solar cell in the electrical and electronic industry.
5. In the nano chemical sector, ionic liquids are used as stable catalysts.
6. Ionic liquids, can absorb air pollutants such as sulfur or nitrogen oxides associated with fossil fuel combustion and serve to separate carbon dioxide, a culprit of global warming.
7. More recently, ionic liquids are used in the development of biopolymer (bioplastic)-based films and coating materials, suggesting their wider use in the future.
Conclusion
The need for green chemistry will increase in the future. Hence replacement or limiting the use of solvents for chemicals processing will remain a dynamic area for research. Ionic liquid dues to its properties such as low volatility, non-flammability, low melting point, high thermal stability, controlled miscibility, recyclability and no toxicity, finds a hope for the green chemistry. Due to high selectivity and activity of ionic liquids, there is growing interest in developing applications for them in a wide range of synthetic reactions. Further, its application is found in environmental chemistry, engineering, biotechnology and electronic industries. Ionic liquids have all potential to replace conventional solvent in both laboratories and industry.
References
- Petkovic, Marija, Kenneth R Seddon, Luis Paulo N Rebelo, Cristina Silva Pereira (2011) "Ionic liquids: a pathway to environmental acceptability." Chem Soc Rev 40(3): 1383-1403.
- Earle, Martyn J, Kenneth R Seddon(2000) "Ionic liquids. Green solvents for the future." Pure and applied chemistry 72(7): 1391-1398.
- Cleary JA (2014) life cycle assessment of residential waste management and prevention. The International Journal of Life Cycle Assessment 19(9): 1607-1622.
- Wasserscheid P, Welton T (2008) Ionic liquids in synthesis. John Wiley & Sons; 2008 Jun 25.
- Wilkes JS (2004) Properties of ionic liquid solvents for catalysis. Journal of Molecular Catalysis A: Chemical 214(1): 11-17.
- Dai S, Luo H (2008) Synthesis of ionic liquids. United States patent 7: 423.
- Patel DD, Lee JM (2012) Applications of ionic liquids. The Chemical Record 12(3): 329-355.
- Plechkova NV, Seddon KR (2008) Applications of ionic liquids in the chemical industry. Chem Soc Rev 37(1):123-150.






























