Theoretical Approaches for Treatment of Neglected Tropical Diseases by Drug Designed Gold Complexes with Thiosemicarbazone
Ronaldo Junio de Oliveira1
1Department of Physics, Institute of Exact, Natural Sciences and Education, Federal University of Triângulo Mineiro, Brazil
Submission: March 01, 2019; Published: March 15, 2019
*Corresponding author: Ronaldo Junio de Oliveira, Department of Physics, Institute of Exact, Natural Sciences and Education, Federal University of Triângulo Mineiro, Brazil
How to cite this article: Ronaldo J O. Theoretical Approaches for Treatment of Neglected Tropical Diseases by Drug Designed Gold Complexes with Thiosemicarbazone. Nov Appro Drug Des Dev 2019; 5(1): 555651. DOI: 10.19080/NAPDD.2019.04.555651
Abstract
Theoretical studies have been increasing as a pivotal step in the design of novel compounds with biological activity. In special, molecular modeling and docking algorithms are computational tools used in the early stages of development and evaluation of drug candidates that may act in a specific biomolecule target. Regarding Chagas that is a neglected tropical disease, it has two enzymes which is used as major drug target: cruzain and Trypanosoma cruzi old yellow enzyme (TcOYE). In this manuscript, it will be reviewed the latest metal-based thiosemicarbazone compounds, specially gold(III), evaluated by theoretical tecniques for the discovery of new treatments for Chagas disease.
Keywords: Docking; Molecular modeling; Chagas; Trypanosoma cruzi; Cruzain; Old yellow enzyme; Gold; Leishmaniasis
Introduction
The use of computational tools in the early stages of development and evaluation of drug candidates has increased enormously in the recent decades, especially with the increase in performance and accuracy by docking algorithms [1]. In addition, this decade was marked with the 2013 Nobel Prize in chemistry awarded to three computational scientists in which their contributions to the area were enormously. Theoretical studies are now an obligatory routine in experimental laboratories and pharmaceutical companies due to the fact that it increases efficiency and reduces production costs by filtering most part of wetlab experiments. Specially, the molecular modeling and docking techniques have been employed to better understand the binding mode of action of many chemical compounds against active site of disease-related biomolecular targets [1-6].
Neglected tropical diseases are a special case of infirmities that, although they are present in 11% of world incidency, they represent a small fraction of the medicines developed worldwide. In the last 8 years, more than 250 new pharmaceutical compounds hitted the marked, yet only 3% are related to neglected diseases [7]. There are more than 20 neglected tropical diseases recognized by World Health Organization (WHO), and Chagas disease is among them one important subject to drug discovery research. Chagas disease is endemic mainly in Latin America, and other continents as well, infecting more than 8 milion people worldwide accordingly to WHO.
Chagas is caused by the flagellate protozoan Trypanosoma cruzi that is currently treated with benznidazole, the only drug used for chemotherapeutic treatment of infected patients, which is not effective in the chronic phase of the illness and causes many undesirable side effects [8]. One strategy for the treatment of Chagas disease is the design of compounds that selectively inhibit enzymes essential for survival of the parasite within the host cell. In the case of T. cruzy, cysteine protease cruzain and old yellow (TcOYE) enzymes are very attractive drug targets [9,10], (Figure 1).
Cruzain enzyme is by far the most studied protease of T. cruzy due to its role as a virulence factor, replication and survival of the parasite [11]. In this context, an alternative for pharmacological treatment of Chagas disease is to design compounds that inhibit the biological function of the parasite enzymes. Gold (III) complexes with thiosemicabazone have been receiving special attention due to their anti-cancer and antiparasitic effects such as against T. cruzi [12-15]. Our research group identified, in a recent paper, the compound 4-CL of formula [AuIII(Hdamp)(L1)]Cl (Hdamp = dimethylammoniummethylphenyl), which showed low cytotoxicity in spleen cells and high selectivity index [16].
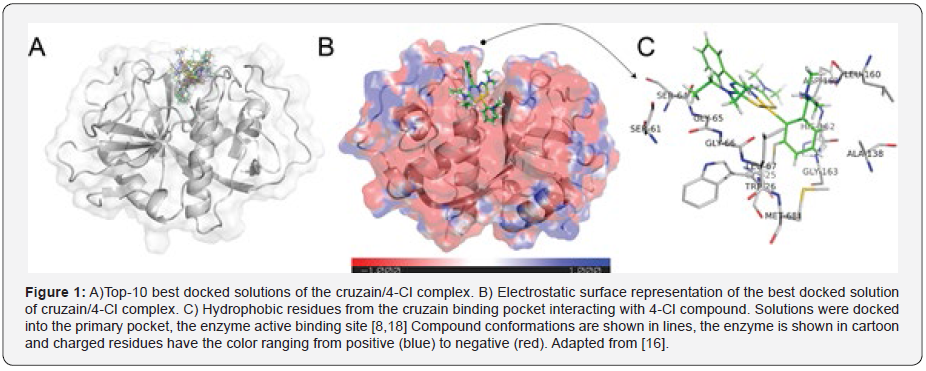
Compound 4-Cl was subjected to in vitro and in vivo experimental trials in a more recently work where it was shown that 4-Cl is effective in directly killing T. cruzi parasites in both trypomastigote and amastigote forms [17]. 4-Cl reduces parasitaemia and tissue parasitism at a very low dose in addition to protecting the liver and heart from tissue damage, resulting in survival of 100% of the 4-Cl-treated mice during the acute phase [17]. 4-Cl was avaluated by the in silico molecular docking simulations with the predicted binding mode with cruzain shown in (Figure 1). The ligand interacts with thirteen cruzain hydrophobic residues in the binding pocket and with residue Cys25 via a hydrogen bond (Figure 2).
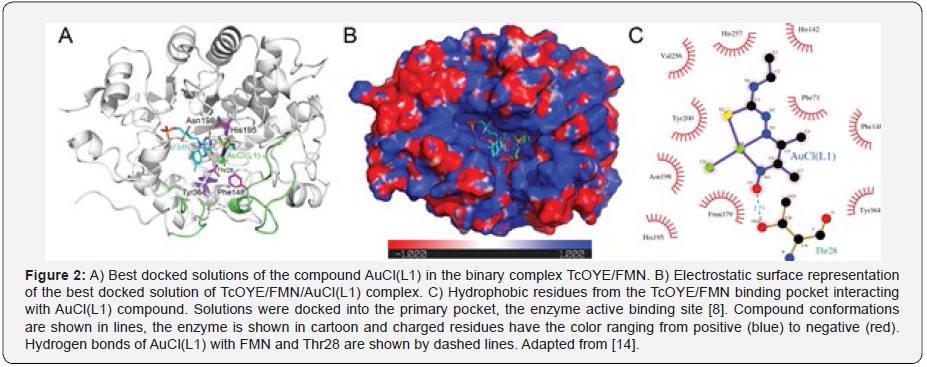
Another enzyme that has been used for Chagas disease as a drug target is TcOYE. Metal-derived compounds complexed with thiosemicarbazone were designed to act against T. cruzy and both, in silico study and in vitro trypanocidal activity showed that, within the studied metal compound series, gold(III) complex presented the greatest potential, being the most selective compound with a similar selectivity index to that of the standard drug benznidazole [14]. Figure 2 shows compound AuCl(L1) binded to the binary complex TcOYE/FMN, in a close distance that an electron/proton transfer might occur. In this study [14], semiempirical calculations were executed in order to characterize the variation in the interaction enthalpy in two different gold oxidative states of the compound (AuIII and AuI). Semiempirical results suggested that the ligand complex formed by AuI in reduced oxidation state is more stable (lower enthalpy variation) them the ligand complexed with AuIII, which would explain a hydride transfer observed in the enzymatic activity assays performed in the same study [14].
Conclusion
In silico protocols are applied in the pipeline of drug discovery in order to select best-ranked compounds that might interact specially via hydrophobic and hydrogen bond interactions. This theoretical step has been proved to be important in the development of gold(III)-thiosemicarbazone compounds with biological activity against the T. cruzi parasite. Future studies in structure-based development of novel anti-chagasic drugs based in gold(III)–thiosemicarbazone complexes and biochemical/ metabolic pathways are necessary to better uncover the mechanism of action of this drug candidate in the parasite, yet we already developed promising candidates. Another important characteristic of gold(III)–thiosemicarbazone complexes is that they not only act against Chagas disease and cancer, yet an on-going work suggests that slightely modified compounds may also act against others neglected tropical diseases such as Leishmaniasis.
Acknowledgments
This research was supported by the brazilian funding agencies, Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG, No. APQ-00941-14) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, No. 438316/2018-5). The author thanks the facilities Núcleo de Computação Científica da Universidade do Estado de São Paulo (NCC/GridUNESP) and Centro Nacional de Processamento de Alto Desempenho em São Paulo (CENAPAD-SP) for the computational resources.
References
- Verdonk ML, Cole JC, Hartshorn MJ, Murray CW, Taylor RD (2003) Improved protein-ligand docking using GOLD. Proteins 52(4): 609-623.
- Mandelli F, Cairo JPLF, Citadini APS, Büchli F, Alvarez TM, et al. (2013) The characterization of a thermostable and cambialistic superoxide dismutase from Thermus filiformis. Lett Appl Microbiol 57(1): 40-46.
- Allen WJ, Balius TE, Mukherjee S, Brozell SR, Moustakas DT, et al. (2015) DOCK 6: Impact of new features and current docking performance. J Comput Chem 36(15): 1132-1156.
- Chen YC (2015) Beware of docking! Trends Pharmacol Sci 36(2): 78-95.
- Lacerda RBM, Freitas TR, Martins MM, Teixeira TL, Candido PA, et al. (2018) Isolation, leishmanicidal evaluation and molecular docking simulations of piperidine alkaloids from Senna spectabilis. Bioorg Med Chem 26(22): 5816-5823.
- Cavatão de Freitas T, Oliveira RJ de Mendonça RJ de, Candido PA, Silva Pereira LL, Devienne KF, et al. (2019) Identification of Bioactive Compounds and Analysis of Inhibitory Potential of the Digestive Enzymes from Syzygium sp. Extracts. Journal of Chemistry 19(2): 125-127.
- Ferreira LLG, Andricopulo AD (2019) Drugs and vaccines in the 21st century for neglected diseases. Lancet Infect Dis 19(2): 125-127.
- Ferreira RS, Simeonov A, Jadhav A, Eidam O, Mott BT, et al. (2010) Complementarity Between a Docking and a High-Throughput Screen in Discovering New Cruzain Inhibitors. J Med Chem 53(13): 4891-4905.
- Kubata BK, Kabututu Z, Nozaki T, Munday CJ, Fukuzumi S, et al. (2002) A Key Role for Old Yellow Enzyme in the Metabolism of Drugs by Trypanosoma cruzi. J Exp Med 196(9): 1241-1252.
- Espíndola JWP, Cardoso MV de O, Filho GB de O, Oliveira e Silva DA, Moreira DRM, et al. (2015) Synthesis and structure–activity relationship study of a new series of antiparasitic aryloxyl thiosemicarbazones inhibiting Trypanosoma cruzi cruzain. Eur J Med Chem 10: 818-835.
- Doyle PS, Zhou YM, Hsieh I, Greenbaum DC, McKerrow JH, et al. (2011) The Trypanosoma cruzi Protease Cruzain Mediates Immune Evasion. PLoS Pathog 7(9): e1002139.
- Ferreira FB, Pereira TM, Souza DLN, Lopes DS, Freitas V, et al. (2017) Structure-Based Discovery of Thiosemicarbazone Metalloproteinase Inhibitors for Hemorrhage Treatment in Snakebites. ACS Med Chem Lett 8(11): 1136-1141.
- Rettondin AR, Carneiro ZA, Gonçalves ACR, Ferreira VF, Oliveira CG, et al. (2016) Gold(III) complexes with ONS-Tridentate thiosemicarbazones: Toward selective trypanocidal drugs. Eur J Med Chem 120: 217-226.
- Gonçalves ACR, Carneiro ZA, Oliveira CG, Danuello A, Guerra W, et al. (2017) PtII, PdII and AuIII complexes with a thiosemicarbazone derived from diacethylmonooxime: Structural analysis, trypanocidal activity, cytotoxicity and first insight into the antiparasitic mechanism of action. European Journal of Medicinal Chemistry 141(1): 615-631.
- Paixão DA, Lopes CD, Carneiro ZA, Sousa LM, de Oliveira LP, et al. (2019) In vitro anti-Trypanosoma cruzi activity of ternary copper (II) complexes and in vivo evaluation of the most promising complex. Biomed Pharmacother 109: 157-166.
- Lopes CD, Gaspari APS, Oliveira RJ, Abram U, Almeida JPA, et al. (2018) Organometallic gold (III) [Au(Hdamp)(L14)]Cl (L1 = SNS-donating thiosemicarbazone) complex protects mice against acute cruzi infection.
- Gillmor SA, Craik CS, and Fletterick RJ (1997) Structural determinants of specificity in the cysteine protease cruzain. Protein Sci 6(8): 1603-1611.






























