Annexin 2 Protein Expression is Associated with Breast Cancer Subtypes in African American Women
Desta A Beyene1,5,7, Norma F Kanarek2, Tammey J Naab3, Santa L Ricks4 and Tamaro S Hudson5,6,7*
1Department of Biochemistry and Molecular Biology, Howard University, USA
2Department of Environmental Health and Engineering, Johns Hopkins Bloomberg School of Public Health, USA
3Department of Pathology, Howard University, USA
4Department of Biological Sciences, Hampton University, USA
5Department of Research, Veteran Affairs Medical Center, USA
6Department of Pharmacology, Howard University, USA
7Howard University Cancer Center, USA
Submission: January 23, 2019; Published: February 22, 2019
*Corresponding author: Tamaro S Hudson, Department of Research, Veteran Affairs Medical Center, College of Medicine, Howard University, Washington, DC, USA
How to cite this article: Desta A Beyene, Norma F Kanarek, Tammey J Naab, Santa L Ricks, Tamaro S Hudson. Annexin 2 Protein Expression is Associated with Breast Cancer Subtypes in African American Women. Nov Appro Drug Des Dev 2019; 4(5): 555650. DOI: 10.19080/NAPDD.2019.04.555650
Keywords
Immunohistochemistry; Breast cancer; Annexin 2; Overall survival; Recurrence-free survival; Triple Negative; HER2; Luminal A; Luminal B
Abbreviations: HER2: Human Epidermal Growth Factor Receptor 2; IDC: Invasive Breast Ductal Carcinomas; FFPE: Formalin-Fixed Paraffin Embedded; TMA: Preparation of Tissue Microarrays; IHC: Immunohistochemistry; ER: Estrogen Receptor; PR: Progesterone Receptor
Introduction
Breast cancer is the most common cancer diagnosis and the second leading cause of death among American women [1]. African American breast cancer patients of all ages are more likely to have advanced disease at diagnosis, an increased risk of recurrence and a poorer prognosis compared with their Caucasian counterparts [2,3]. Tumor stage, tumor grade and lymph node metastasis are commonly used as prognostic factors for breast cancer [4]. However, these are not sufficient for accurately predicting [4]. Therefore, biomarkers are needed to more accurately predict survival outcome. Considering the role of ANX2 in tumorigenesis and metastasis its use as prognostic marker for breast cancer needs to be investigated.
Annexin 2 (ANX2) belongs to a family of Ca2+ dependent phospholipid and membrane binding proteins called annexins [5] and contains a conserved repeating domain of approximately 70 amino acids. ANX2 (also called p36, annexin II, or ANXA2) is a 36 kDa protein [6] and is located on chromosome 15q22.2 [7]. ANX2 is highly conserved and ubiquitously distributed in various body cell types and accounts for about 0.5–2% of the total cell protein [8]. ANX2 plays a major role in tumorigenesis, drug resistance, and metastasis [9]. Overexpression of ANX2 is frequently observed in a broad spectrum of cancer cells, including breast cancer [10,11], colorectal carcinoma [12], and lung cancer [13] while under expressed in others, such as prostate cancer [14].
In breast cancer, ANX2 is undetectable in normal and hyperplastic ductal tissue samples but is consistently expressed in invasive breast cancer and ductal carcinoma in situ [15]. ANX2 gene expression is associated with tumors with selected poor prognostic characteristics such as grade characterized by poor differentiation, enriched expression of human epidermal growth factor receptor 2 (HER2), and TN subtypes [16]. According to Sortie et al. [17] Luminal A tumors have the more favorable prognosis and make up approximately 40% of all breast cancer cases. Luminal B tumors are very similar to Luminal A tumors but have higher expression of proliferative genes in comparison to Luminal A.
They make up 20% of breast cancer cases and tend to be diagnosed at higher tumor grades than Luminal A tumors. HER2 tumors are often aggressive and have poor prognosis. TN breast cancer makes up approximately 15-20% of breast cancer diagnoses. TN is the most aggressive breast cancer subtype [18] and is unresponsive to anti-hormonal and HER2-targeted therapies due to the absence of hormone receptors and HER2 expression. The upregulation of ANX2 in cancer has several clinical applications, including as a diagnostic marker for early detection, a predictive factor for prognosis, or a marker for drug resistance.
African American women continue to have high rates of breast cancer mortality compared with other ethnicities and have a limited number of predictive markers for the different subtypes. Therefore, the aim of the study was to examine the use of ANX2 as a potential prognostic marker for breast cancer subtypes in African American women.
Materials and Methods
Study design
This study was reviewed and formally exempted by the Howard University Institutional Review Board. Our cases originated primarily from the community population of women served who are predominantly minority and of low to moderate income. We analyzed 135 sequential invasive breast ductal carcinomas (IDC) from African-American women diagnosed and treated at the Howard University Hospital between 2000 and 2010 where adequate tumor tissue was available and obtained. Demographic and clinical information was case information from the Howard University Cancer Center Tumor Registry. The hormone receptor status of HER2, ER and PR were used to classify four breast cancer subtypes as described by others [16,19]. Breast cancer subtype was abstracted from hospital reports: Luminal A, Luminal B, HER2 positive, and TN. Tumor characteristics collected included grade (categorized as Grade (I-II/III-IV) and stage (I-II/ III-IV, tumor size, metastases (yes/no), and nodal involvement (yes/no)). Age at diagnosis and menopausal status (pre/post) were obtained from the medical record as well.
Tissue samples
Formalin-fixed paraffin embedded (FFPE) tissues in diagnosed women were assessed using tissue microarrays (TMAs, Pantomics, Inc, Richmond, CA). The TMAs consisted of 10 x 16 arrays of 1.0 mm tissue cores from well preserved morphologically representative tumor cells in archived FFPE surgical blocks from primary IDCs in 135 African-American women. A precision tissue arrayer (Beecher Instruments, Silver Spring, MD) with two separate core needles for punching the donor and recipient blocks was used. The device also had a micrometer-precise coordinate system for tissue assembly on a multi-tissue block. Two separate tissue cores of IDC represented each surgical case in the TMA. Each tissue core was assigned a unique TMA location number, which was subsequently linked to an Institutional Review Boardapproved database containing demographic and clinical data.
Preparation of tissue microarrays (TMA)
TMA is a useful tool for identifying the signature profiles of proteins in the different breast cancer subtypes. The TMA paraffin blocks were constructed in our lab as described by Hewitt SM [20]. The TMA blocks with core samples were tempered by placing in the incubator at 370C overnight, cut on microtome to obtain 5-μm thick slices and float mounted on super frost plus micro slides. Slides were stained with hematoxylin-eosin (H&E) and representative areas with invasive tumor were identified and marked on the H&E slide by the pathologist. The individually marked slides were placed on top of each donor paraffin block and carefully aligned to locate the corresponding tumor sites where the core samples are to be collected. Three separate 1.0mm tissue cores were obtained from each donor block and mounted in the recipient TMA (Beecher, Inc., Pathological Devices).
Immunohistochemistry
Immunohistochemistry (IHC) was performed on three TMA breast tumor sections of FFPE tissue. The TMA slides were deparaffinized in xylene twice for 5minutes each, rehydrated in absolute ethanol (2 times 5 minutes each) followed by 95% and 70% ethanol for 5minutes each. Deparaffination was completed manually with xylene washes and serial rehydration through alcohol-water series. Further deparaffination, rehydration and heat-induced antigen retrieval at pH 9.0 was performed on DAKO PT-Linker (Carpinteria, CA). Antibody detection was carried out using an anti-ANX2 (clone C-10, monoclonal mouse antihuman, 1:10k dilution). IHC was then completed using the DAKO Autostainer Link Chamber (Carpinteria, CA) according to manufacturer’s protocol. The binding of the primary antibody was visualized using the Avidin Biotin Complex method (ABC kit, Vector Lab). The chromogen substrate was diaminobenzidine (DAB kit, Invitrogen). Stained slides were counterstained with hematoxylin (Invitrogen) and finally treated with 70%, 95% and absolute ethanol and xylene. Slides were cover slipped with an automatic unit (Tissue-Tek SCA, Thermo- Fisher Scientific) and observed by a pathologist under the light microscope.
Evaluation of immunohistochemical staining
Tissue samples were analyzed as positive or negative for ANX2 antibody (mouse monoclonal antihuman, clone C-10, 1:10K dilution). Immunohistochemically stained sections were scored by two independent observers blinded to the clinical outcome using a laboratory grade binocular light microscope. Individual tissue cores were scored for intensity of reactivity (0, no staining; 1+, weak cytoplasmic and membrane staining; 2+, moderately intense staining; and 3+, strong staining) and the percentage of reactive cells. The results were entered into a secure research database. An H-score was derived from the results of these measurements by multiplying intensity score by extent/percentage of stained area. All samples were categorized based on the H-score [21,22]. H-score ≥200 cells stained with ANX2 were recorded as positive and those with H-score <200 cells stained as negative. All samples were scored while blinded to tissue phenotype. Estrogen Receptor (ER), Progesterone Receptor (PR) and Human Epidermal Growth Factor Receptor (HER2) scores were obtained from the Tumor Registry medical records. The specific DAKO antibodies used were: Er-alpha, rabbit monoclonal, cloneSP1, 1:200 dilution; PR, mouse monoclonal, clone PgR636, 1:100 dilution; HER2, rabbit polyclonal, clone e-erb-2, 1:500 dilution.
Statistical analysis
A case series of breast tumors with pathology specimens at Howard University Hospital were analyzed for ANX2 levels. A total of 135 samples with complete information were included. All statistical analyses were conducted in Stata 11 and included tests of significance by Chi Square, ANOVA, multinomial regression and Kaplan Meier analysis of survival.
Results
A visual comparison of the expression of ANX2 shows distinct features that distinguish three of the four breast cancer subtypes (Figure 1). ANX2 is highly expressed in HER2 (A) but is marginally expressed in TN (B) and shows limited expression in luminal (C). The demographics and pathological characteristics showed that approximately 75% (101/135) of the patients were older than 50 years. Positive expression status for ER, PR and HER2 was 39%, 48% and 90%, respectively. The percentages for luminal A, Luminal B, HER2+ and TN were 41%, 15%, 11% and 33%, respectively. Most of the cancers (77%) were classified as stage I-II. Sixty percent of tumors were of size greater than 20 mm. Ninety percent of the patients showed no distant metastasis but 57% had affected lymph nodes.
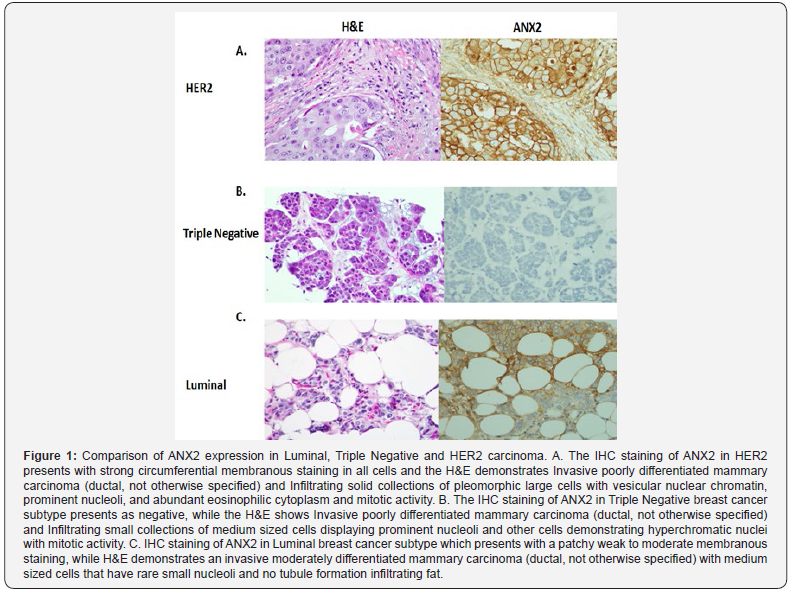
High expression of ANX2 was observed in 23% of the patients. Significant differences were observed for ANX2 expression (p=0.055) and age at diagnosis (p=0.024) by cancer subtype (Table1). Tumor size differences between less than 20mm and greater than 20mm within the subtypes were marginally significant (0.146). There were no significant differences in grade, disease stage, presence of affected nodes, or distant metastasis. (Table 2) shows t-test analysis results of ANX2 expression and clinical characteristics in relation to overall survival, disease-free and recurrence-free survival. A marginal significant difference (p = 0.173) was observed between the low and high expression of ANX2. The age difference between patients less than 50 years and 50 years or older was highly significant for all three survival outcomes (p<0.002). A significant difference was observed between pre- and post-menopausal status. Differences in stage (I-II vs III-IV), tumor size (<20mm vs >20mm) and lymph node (presence or absence) were significantly different for overall and disease-free survival. There was also a significant difference between patients that had metastasis and those that did not have any (<0.002) for all three-survival outcomes.
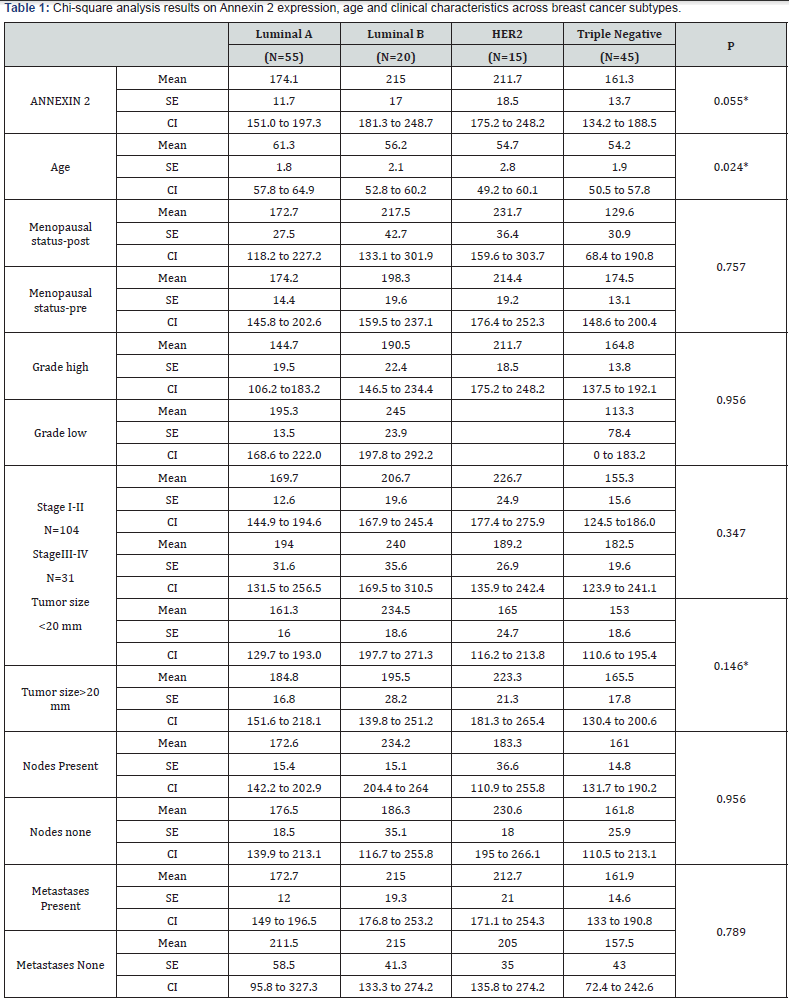
*Indicates significance at *p <0.05; **p<0.10
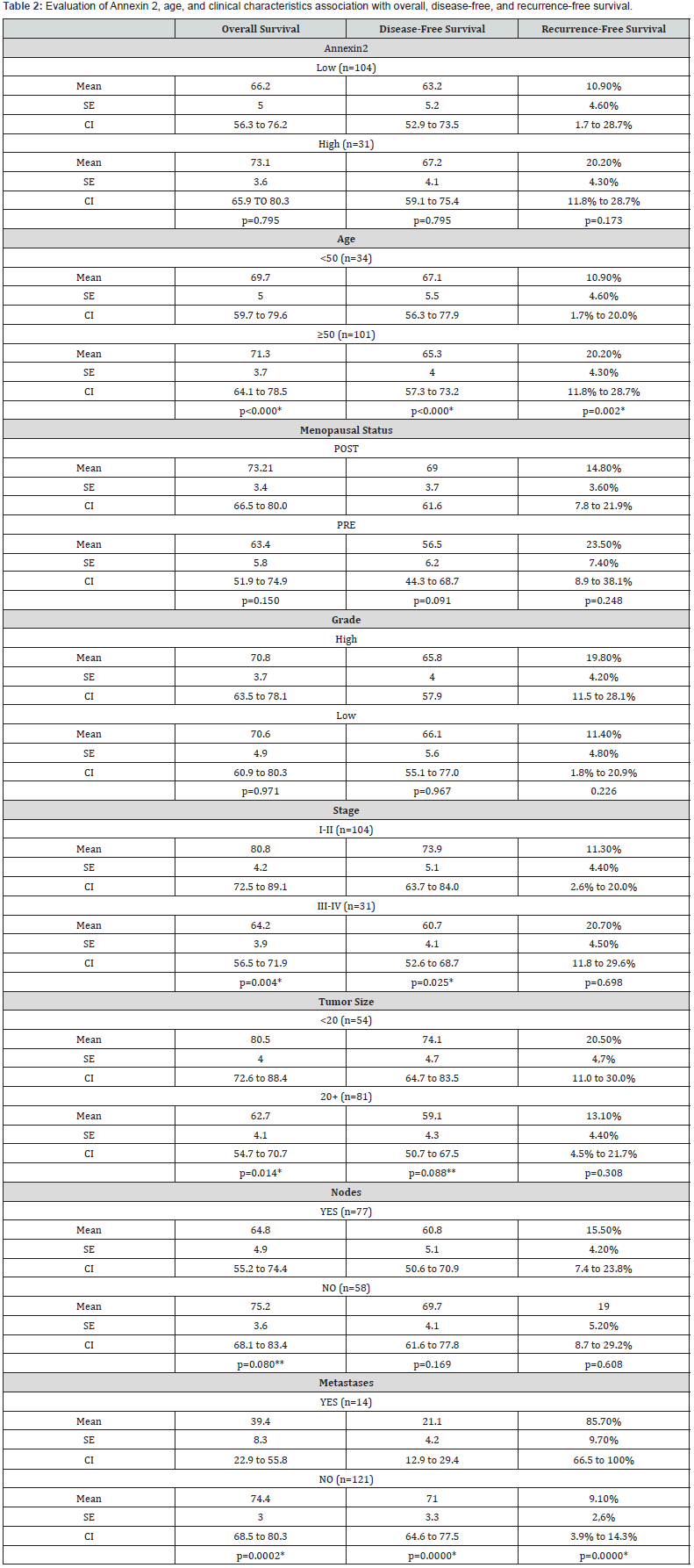
*Indicates significance at *p <0.05; **p<0.10
Multinomial regression analysis showed significant differences in ANX2 expression for HER2 and Luminal B but not for TN compared to Luminal A (Table 3). Age at diagnosis was a significant factor in HER2 and TN subtypes though of borderline significance in the Luminal B subtype. For high grade cancer, significant differences were found for TN and Luminal B.
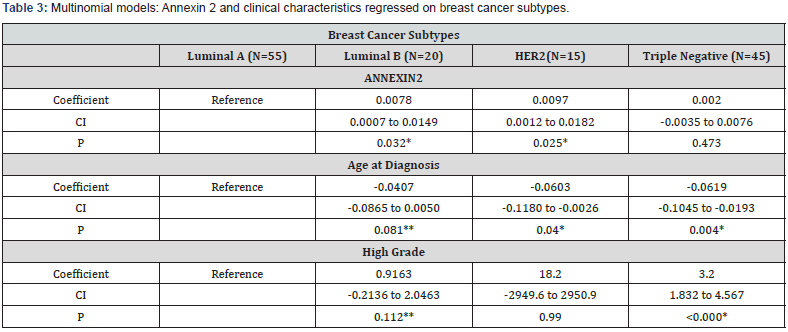
*Indicates correlation is significant at *p <0.05; **p<0.10
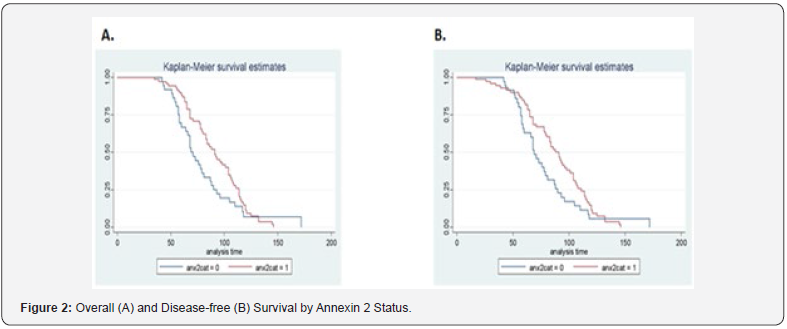
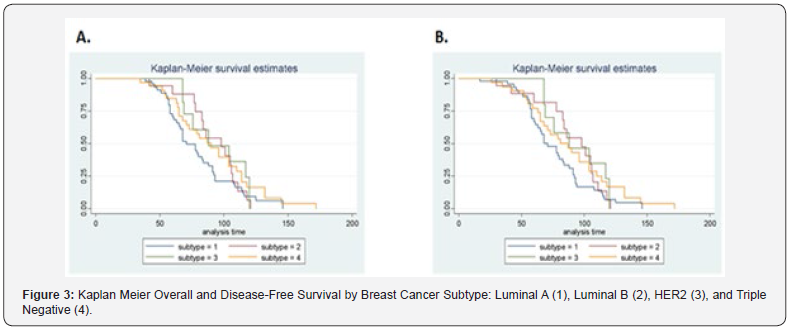
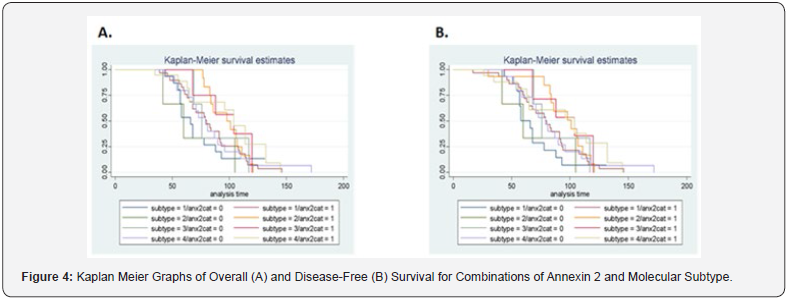
The Kaplan-Meir disease-free survival and overall survival by ANX2 expression are shown in (Figures 2-4). ANX2 expression was associated with statistically borderline (p=0.10) higher overall survival and disease-free survival time (Figure 2). The expression of ANX2 in relation to overall and disease-free survival for the four subtypes is shown in (Figure 3) and though not statistically significant, Luminal A appears to have the worse overall and disease-free survival. In these two figures, ANX2 expression was associated with higher overall and disease-free survival time for Luminal B, HER2 and TN. (Figure 4) shows ANX2 expression in relation to overall and disease-free survival for the four subtypes. ANX2 expression for HER2 subtype showed a higher overall and disease-free survival compared to the other three types, though across all combinations, there was no statistical significance.
Discussion
There is very limited research work on the association of ANX2 protein with the different breast cancer subtypes in African Americans. Most reported results address ANX2 gene expression in TN subtypes. In the present study, ANX2 protein expression was evaluated in relation to Luminal A, Luminal B, HER2 and TN breast cancer. ANX2 expression was a significant factor in the disease progression in all four breast cancer subtypes. Age at diagnosis and tumor size were also significant factors. The multinomial analysis using Luminal A as a reference showed significant increase in ANX2 expression for Luminal B and HER2. For the TN, significant ANX2 increase was observed in the high grade only. According to Noor & Gibbs et al. [9,18] increased ANX2 expression is related to higher levels of HER2 at mRNA and protein levels [14].
Reports from analysis of breast cancer subtypes and normal tissues have shown that ANX2 is highly expressed in TN compared to its low to negligible expression in the other subtypes and normal tumors [15,19]. It is also reported that ANX2 expression is associated with poor survival outcome and prognosis. This relates to the fact that ANX2 promotes TN progression through angiogenesis and metastasis [8]. Our results, on the other hand, showed a strong association between high grade and TN. Moreover, there was a strong association between ANX2 and Luminal B as well as HER2 indicating the specific association of ANX2 with the aggressive behavior of HER2. Generally, ANX2 expression is associated with an aggressive phenotype such as high stage, high grade and extensive lymph node metastasis. These features (stage, tumor size, number of nodes and metastasis) had shown significant differences in survival outcome.
In the literature, ANX2 overexpression is associated with racial variation and is a potential prognostic and diagnostic candidate for TN [16,18]. A gene expression study of African American women indicated that ANX2 expression was significantly elevated compared to Caucasian and Hispanic women. Furthermore, the elevated ANX2 gene expression was significantly associated with TN as well as with reduced overall survival and reduced recurrence-free survival [16,17]. Gibbs et al. [18] reported that ANX2 gene expression was correlated with poor survival in patients with TN but not with the other breast cancer types. This correlation between ANX2 expression and poor survival in TN was not found in our study. This is to be expected since significant ANX2 expression in TN was observed in the high-grade tumors only. The effect of disease stage and grade on survival may also have been confounded by age differences and the relatively few patients with high expression of ANX2.
A limitation in our study is that we did not have a large sample size, which may limit the assessments among breast cancer subtypes. Most reported gene studies of ANX2 displayed large sample sizes. In addition, we were unable to distinguish between basal-like breast tumors and TN. The number of patients who expressed ANX2 was much lower than among those who did not (23% vs 77%). Moreover, we note that a majority (75 %) of our patients were older than 50years and about 90% of them had high expression of HER2. Despite these limitations, this preliminary study has shown clearly that ANX2 expression is elevated in Luminal B and HER2 subtypes and in the case of TN, with high grade tumors only. In conclusion, the expression of ANX2 correlates with the aggressiveness of breast cancer and substantiates its prospect as a prognostic marker for molecular breast cancer subtypes.
Conclusion
This study highlights a significant association between ANX2 protein expression and the subtypes Luminal B (p=0.032) and HER2 (p=0.025). Our survival analysis showed that ANX2 protein expression in breast tumors might be a biomarker candidate for breast cancer outcome prediction in high risk groups such as Luminal B and Triple Negative cases (Table 1). Further confirmatory studies will be needed to examine the correlation between ANX2 expression and survival using a larger sample size. In conclusion, our results show that ANX2 protein expression alone is independently a poor prognostic factor of survival in African American women.
Clinical Practice Points
1. ANX2 protein expression can be used as a diagnostic marker for aggressiveness in breast cancer for African Americans.
2. From our results, a significant association between ANX2 protein expression and the breast cancer subtypes Luminal B and HER2 was observed.
3. Age at diagnosis for breast cancer plays an independent and critical role, in addition to ANX2 protein expression, for all breast cancer subtypes.
4. Tumor size, stage and number of nodes together had an influence on differences in overall survival and tumor size and stage together influenced breast cancer-free survival.
References
- Atlanta GA (2015) American Cancer Society. Cancer Facts & Figures. American Cancer Society, Inc, USA.
- Ademuyiwa FO, Olopade OI (2003) Racial differences in genetic factors associated with breast cancer. Cancer Metastasis Rev 22(1): 47-53.
- Chlebowski RT, Chen Z, Anderson GL, Rohan T, Aragaki A, et al. (2005) Ethnicity and breast cancer: Factors influencing differences in incidence and outcome. J Natl Cancer Inst 97(6): 439-448.
- Soerjomataram I, Louwman MW, Ribot JG, Roukema JA, Coebergh JW (2008) An overview of prognostic factors for long-term survivors of breast cancer. Breast Cancer Res Treat 107(3): 309-330.
- Moss SE, Morgan RO (2000) The annexins. Genome Biology 5(4): 219.
- Hajjar KA, Krishnan S (1999) Annexin II: a mediator of the plasmin/plasminogen activator system. Trends in Cardiovascular Medicine 9(5): 128-138.
- Huebner K, Cannizaro LA, Frey AZ, Hecht BK, Hecht F, et al. (1988) Chromosomal localization of the human genes for lipocortin I and Lipocortin II. Oncogene Research 2(4): 299-310.
- Schlaepfer DD, Haigler HT (1990) Expression of Annexins as a function of cellular growth state. Journal of Cell Biology 111(1): 229-238.
- Lokman NA, Ween MP, Oehler MK, Ricciardelli C (2011) The role of Annexin A2 in tumorigenesis and cancer progression. Cancer Microenvironment 4: 199-208.
- Sharma M, Ownbey RT, Sharma MC (2010) Breast cancer cell surface annexin II induces cell migration and neoangiogenic via tPA dependent plasmin generation. Exp Mol Pathol 88(2): 278-286.
- Deng S, Jing B, Xing T, Hou L, Yang Z (2012) Overexpression of annexin A2 is associated with abnormal ubiquitination in breast cancer. Genomics Proteomics Bioinformatics 10(3): 153-157.
- Duncan R, Carpenter B, Main LC, Telfer C, Murray GI (2008) Characterization and protein expression profiling of annexins in colorectal cancer. Br J Cancer 98(2): 426-433.
- Jia JW, Li KL, Wu JX, Guo SL (2013) Clinical significance of annexin II expression in non-small lung cancer. Tumor Biology 34(3): 1767-1771.
- Beyene DA, Naab TJ, Kanarek NF, Apprey V, Esnakula A, et al. (2018) Differential Expression of Annexin2, SPINK1 and Hsp60 predict progression of prostate cancer through bifurcated WHO Gleason score categories in African American Men. Prostate 78(11): 801-811.
- Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, et al. (2006) Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 295(21): 2492-2502.
- Onitilo AA, Engel JM, Greenlee R, Mukesh BN (2009) Breast cancer subtypes based on ER/PR and Her2 expression: Comparison of clinopathologic features and survival. Clin Med Res 7(1-2): 4-13.
- Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, et al. (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci 98(19): 10869-10874.
- Gibbs LD, Chaudhary P, Manshein K, Hare RJ, Mantsch RA, et al. (2018) ANAXA2 expression in African American triple-negative breast cancer patients. Breast Cancer Research and Treatment.
- Nguyen PL, Taghian AG, Katz MS, Niemierko A, Abi Raad RF, et al. (2008) Breast cancer subtype approximated by estrogen receptor, progesterone receptor, and Her-2 is associated with local and distant recurrence after breast-conserving therapy. J Clin Oncol 26(14): 2373-2378.
- Hewitt SM (2012) Tissue microarrays as a tool in the discovery and validation of predictive biomarkers. Methods Mol Biol 823: 201-214.
- Ma H, Lu Y, Marchbanks PA, Suzanne G Folger, Brian L Strom, et al. (2013) Quantitative measures of estrogen receptor expression in relation to breast cancer-specific mortality risk among white women and black women. Breast Cancer Research15(5): R90.
- Diallo-Danebrock R, Ting E, Gluz O, Herr A, Mohrmann S, et al. (2007) Protein expression profiling in high-risk breast cancer patients treated with high-dose or conventional dose-dense chemotherapy. Clin Cancer Res 13(2 Pt1):488-497.






























