Electrochemical Determination of Resorcinol with Tartrazine at Non-Ionic Surfactant Modified Graphite Powder and Carbon Nanotube Composite Paste Electrode
Girish Tigari, JG Manjunatha*, C Raril, and N Hareesha
Department of Chemistry, FMKMC College, Madikeri, Mangalore University Constituent College, Karnataka, India
Submission: December 14, 2019; Published: January 22, 2019
*Corresponding author: JG Manjunatha, Department of Chemistry, FMKMC College, Madikeri, Mangalore University Constituent College, Karnataka, India
How to cite this article: Girish T, JG Manjunatha, C Raril, N Hareesha. Electrochemical Determination of Resorcinol with Tartrazine at Non-Ionic Surfactant Modified Graphite Powder and Carbon Nanotube Composite Paste Electrode. Nov Appro Drug Des Dev 2019; 4(5): 555647. DOI: 10.19080/NAPDD.2019.04.555647
Abstract
In this study, a sensitive and selective electrochemical sensor was developed for electrochemical determination of resorcinol (RC) using graphite powder and carbon nanotube composite paste electrode modified with TX-100 (TX-100/GPCNTCPE) in phosphate buffer solution (PBS), pH 7.0 using cyclic voltammetry (CV) and differential pulse voltammetry (DPV). The GPCNTCPE/TX-100 gives excellent current response for resorcinol as compared to bare graphite powder and carbon nanotube composite paste electrode (BGPCNTCPE). The impact of pH, Sweep rate and concentration of RC were evaluated. The anodic peak of RC was detected at 0.503 V vs. SCE with current response 11.40 μA. The proposed sensor offers low detection limit of order 3.41×10-6 M and limit of quantization (LOQ) is 11.42×10-6 M . This sensor is also adopted for simultaneous determination RC and tartrazine (TZ).
Keywords: Carbon Nanotubes; TX-100, Resorcinol; Electrochemical Determination; Tartrazine
Introduction
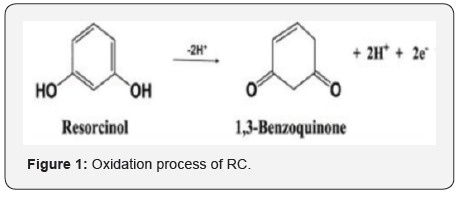
RC (Benzene-1, 3-diol) is a phenolic organic compound with high toxicity can cause dermatitis, catarrh, convulsion, cyanopathy, and even death. It has excellent medicinal value in treating skin disorders like Acne, seborrhoea dermatitis, eczema, psoriasis, and other skin infection. It is also used as a hair colouring agent, antiseptic and disinfectant [1-3]. So, it is too essential to detect and quantify the resorcinol. There were various methods reported regarding the estimation of RC such as Fluorescence [4] high- performance liquid chromatography [5] flow injection chemiluminescence [6] surface Plasmon resonance [7], but these are costly and time-consuming approaches. Also, some electrochemical methods were reported for the detection of resorcinol, the electrochemical methods were established due to its rapid sensitivity and low cost. SDS, TX-100, CTAB and polymer film Modified Carbon nanotubes (CNTs) and graphite paste electrode are wildly used in the electrochemical determination of vitamins, drugs, hormones, neurotransmitters, and food dyes [8-16], etc. due to their high mechanical strength and good electro- conductive property [17]. In this work graphite powder carbon nanotube composite paste modified with TX-100 is fabricated for voltammetric determination of RC and its simultaneous resolution with TZ. To the best of our knowledge, no voltammetric sensor built for concurrent resolution of RC with TZ based on TX-100/GPCNTCPE. The oxidation Process of RC is shown in Figure 1.
Materials and Methods
Apparatus
The CV and DPV were obtained by means of CHI-6038E electrochemical analyzer in combination with a typical three-electrode system and a personal computer for data storage and processing. The GPCNTCPE and TX-100/GPCNTCPE was used as the working electrode, saturated calomel electrode (SCE) and a platinum wire were used as a reference electrode and auxiliary electrode respectively. All the experiments carried out at room temperature.
Reagents and Chemicals
RC took from Himedia, India. CNTs were bought from Sisco Research Laboratories Pvt, Ltd, Mumbai. Silicon oil and TX-100 were brought from Nice Chemicals, India. Remaining chemicals were of analytical grade and used without refinement. The RC standard solution (25×10-4 M) was prepared using distilled water. 25×10-3 M TX-100 was prepared using distilled water. PBS (0.1 M) was prepared by intermixing the appropriate amount of 0.1 M monosodium hydrogen phosphate and 0.1 M disodium hydrogen phosphate and having pH 7.0 was used as supporting electrolyte. Data’s were taken at room temperature (25°C).
Fabrication of GPCNTCPE and TX-100/GPCNTCPE
The BGPCNTCPE was made by intermixing 35% graphite powder, 35% CNTs and 30% silicone oil in a mortar and grounded for about 15 min to get a homogeneous paste. Obtained composite paste was packed into the hole of a Teflon tube (3 mm in diameter), and the face of the electrode was made to uniform with the help of a tissue paper. TX-100/ GPCNTCPE was prepared by immobilizing 10 μL of TX-100 on the surface of the electrode for 5 min.
Results and Discussion
Electrochemical Analysis of RC at TX-100/GPCNTCPE
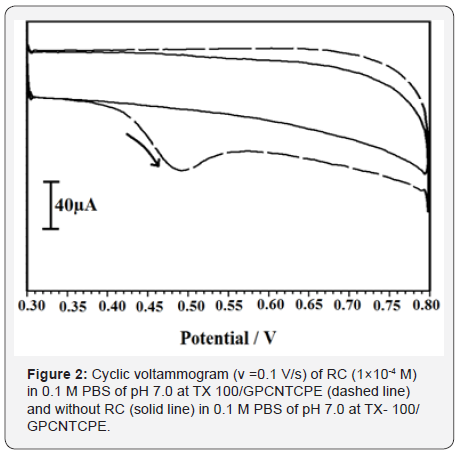
As shown Figure 2, in the absence (solid line) of RC which is characterized by CV with potential 0.3 to 0.8 V at a scan rate of 0.1 V/s (0.1 M PBS, pH 7.0) no peak appears for TX-100/GPCNTCPE. But under identical conditions and in presence of 1×10-4 M RC (dashed line) broader oxidation peak was observed at 0.503 V with the corresponding peak current of 11.40 μA, It shows that the electrochemical behaviour of RC at TX-100/GPCNTCPE.
Voltammetry Response of RC at TX-100/GPCNTPE
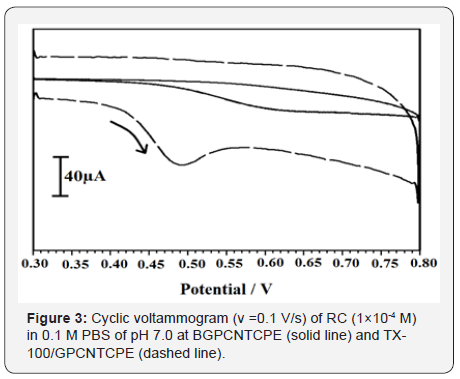
The electrochemical response of RC at TX-100/GPCNTCPE is shown in Figure 3, the electrochemical response of RC is not observed at BGPCNTCPE (solid line) in 0.1 M PBS, pH 7.0. But, the voltammetric response was achieved at TX-100/GPCNTCPE (dashed line). At TX-100/GPCNTCPE the anodic peak potential (Epa) for RC was found to be 0.503 V (vs. SCE) with a current response 11.40 μA, with irreversible behaviour.
Effect of Sweep Rate on Peak Current of RC
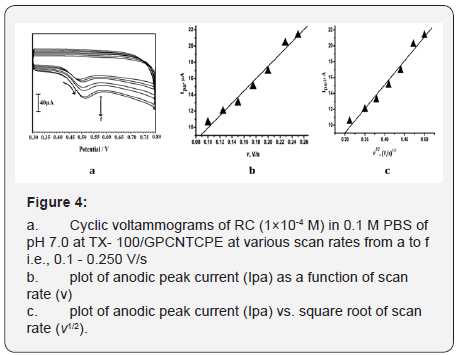
The impact of the sweep rate on electrochemical oxidation of RC was studied at TX-100/GPCNTCPE by cyclic voltammetry. Sweep rate studies were carried out to critic whether the process on TX-100/GPCNTCPE was under diffusion controlled. When the scan rate was varied from 0.1-0.250 V/s in 1×10-4 M solution of RC, the obtained cyclic voltammogram is shown Figure 4a. A plot of the oxidation peak current vs. scan rate (Figure 4b) gives a straight line with linear equation Ipa (A) = 78.04+1.65 v (V/s), and correlation coefficient R = 0.99 Here Ipa an increase with increase in the scan rate from 0.1 V/s to 0.250 V/s. The graph of anodic peak current (Ipa) vs. Square root of scan rate (v1/2) showed a linear relationship (Figure 4c) the correlation coefficient was found to be 0.99. This specifies the electrode process was diffusion controlled.
Electrochemical Analysis of RC at TX-100/GPCNTCPE
As shown Figure 2, in the absence (solid line) of RC which is characterized by CV with potential 0.3 to 0.8 V at a scan rate of 0.1 V/s (0.1 M PBS, pH 7.0) no peak appears for TX-100/GPCNTCPE. But under identical conditions and in presence of 1×10-4 M RC (dashed line) broader oxidation peak was observed at 0.503 V with the corresponding peak current of 11.40 μA, It shows that the electrochemical behaviour of RC at TX-100/GPCNTCPE.
Influence of pH
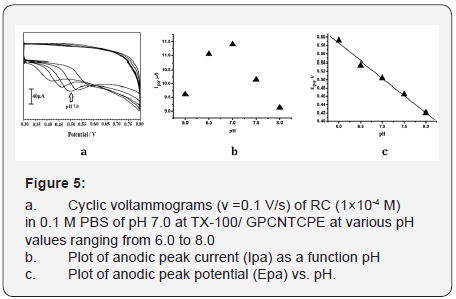
The effect of solution pH on the peak current and peak potential was analysed by increasing the pH. The cyclic voltammograms of RC documented from 6.0 to 8.0 pH at a scan rate of 0.1 V/s in 0.2 M PBS is revealed in Figure 5a, the plot of Ipa vs. pH (Figure 5b) shows the maximum peak current achieved at pH 7.0, this may be due to the faster electron transfer rate at neutral pH and is the physiological pH, hence this pH was preferred to bring out the electrochemical study
Also impact of pH on anodic peak potential of RC was investigated by cyclic voltammetry in the pH range 6.0 - 8.0 (Figure 5c), the Epa vs. pH graph evidently indicates that the catalytic peak shifts to a more negative potential with increasing pH with the corresponding equation Epa= -0.072 pH -1.08, the slope of 72 V/pH unit is near to the ideal value of 59 which might indicate that the number of protons and electrons involved in the electrochemical reaction is in the ratio 1:1[18]. The oxidation process of RC involves two electrons and two protons as shown in Figure 1.
Response of RC at SLSMCNTPE by DPV
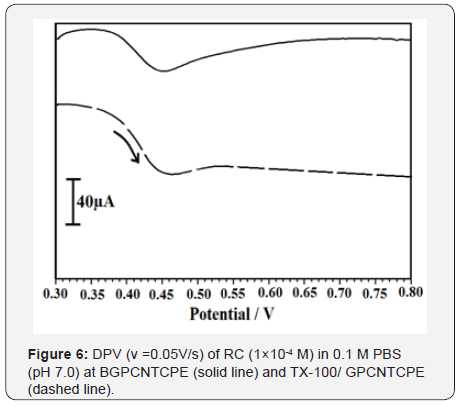
DPV is a sensitive technique than CV and it gives upper current density and superior resolution. DPV response for 1×10-4 M RC in 0.2 M PBS (pH 7.0) at BGPCNTCPE and TX-100/GPCNTCPE in the potential range 0.3 to 0.8 V and are shown in Figure 6, The peak potential for RC is detected at 0.503 V with good current sensitivity 12 μA and there was a small peak was detected at 0.452 V for GPCNTCPE, with current 2.5 μA which concludes catalytic oxidation of RC is amplified at TX-100/GPCNTCPE compared to BGPCNTCPE.
Calibration Plot and Limit of Detection for RC
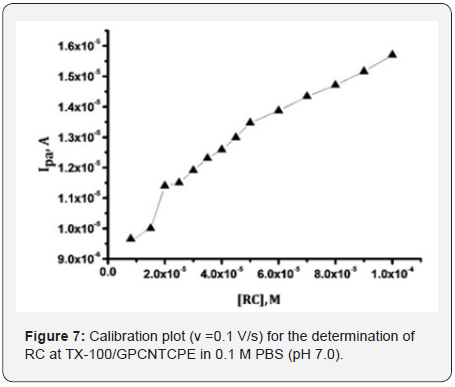
Figure 7 signifies the calibration plot, the anodic peak currents of CVs recorded at TX-100/GPCNTCPE in 0.1 M PBS (pH 7.0) for different concentrations of RC in the range 10-100 μM the oxidation peak current values amplified linearly with increase of RC concentration is expressed as Ipa (A) = 9.05 ×10-6 +0.063 C (M) with R=0.99. The limit of detection (LOD) and limit of quantization (LOQ) were assessed by applying the standard equations LOD = 3s\m and LOQ = 10s/m [19], where m is the slope of the calibration curve and s is the standard deviation of the peak currents of the blank solution (five replicates). The LOD and LOQ for determination of RC at TX-100/GPCNTCPE were evaluated as 3.41 ×10-6 M and 11.42×10-6 M. Table 1, [20- 23] displays a comparison of the Tx-100/GPCNTCPE with other electrodes described in the literature for determination of RC. This sensor gives satisfactory results for quantization of RC.
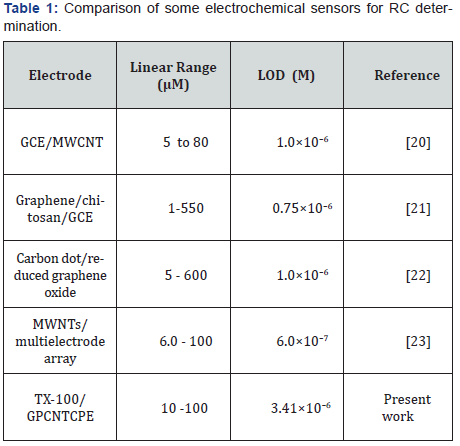
MWCNT: Multi Walled Carbon Nanotube, GCE: Glassy Carbon Electrode, TX-100/GPCNTCPE: Triton 100/Graphite Powder-Carbon Nanotube Composite Paste Electrode; LOD: Limit of Detection
Simultaneous Determination of RC and TZ
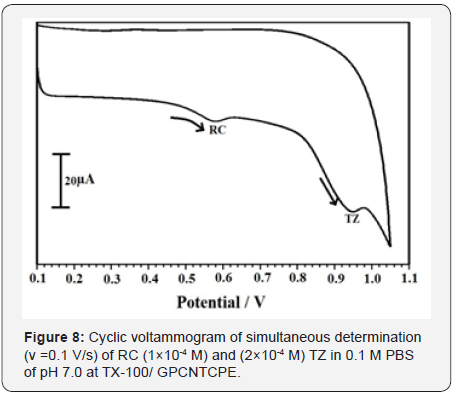
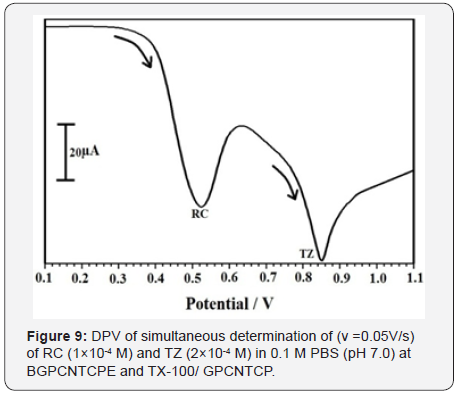
Tartrazine is used as a colorant in drug, food, and cosmetics so it is necessary to determine the TZ with RC by using both CV and DPV at TX-100/GPCNTCPE in 0.1M PBS, pH 7.0. The RC (1×10-4 M) and TZ (2×10-4 M) were detected at 0. 580 V and 0.951 respectively with enhanced current response using cyclic voltammetry as shown in Figure 8, The RC (1×10-4 M) and TZ (2×10-4 M) were resolved at potential 0.525 V and 0.850 V with good current response by means of DPV as shown in Figure 9.
Conclusion
In this study we developed sensitive sensor for resorcinol, the fabricated sensor having good stability, repeatability and reproducibility, Low detection limit of 3.41×10-4 M and LOQ is 11.42×10-6 M In concentration range 10-100 μM and also performed simultaneous resolution of RC and TZ using both CV and DVP with enhanced current sensitivity and this sensor also applicable RC determination in real sample. So, it a better choice for simultaneous resolution of RC and TZ.
Acknowledgements
We gratefully acknowledge the financial support from the VGST, Bangalore under Research Project No. KSTePS / VGST - KFIST (L1)2016-2017/GRD-559/2017-18/126/333, 21/11/2017.
References
- Manjunatha JG (2017) A new electrochemical sensor based on modified carbon nanotube-graphite mixture paste electrode for voltammetric determination of resorcinol. Asian J Pharm Clin Res 10(12): 295-300.
- Chenthattil Raril, Jamballi G Manjunatha (2018) Sensitive Electrochemical Analysis of Resorcinol using Polymer Modified Carbon Paste Electrode: A Cyclic Voltammetric Study. Anal Bioanal Electrochem 10 (4): 488-498.
- Khodari M, Mersal GAM, Rabie EM, Assaf HF (2018) Electrochemical Sensor based on Carbon Paste Electrode Modified by TiO2 nano-particles for the Voltammetric Determination of Resorcinol. Int J Electrochem Sci 13(4): 3460-3474.
- Pistonesi MF, Di Nezio MS, Centurión ME, Palomeque ME, Lista AG, et al. (2006) Determination of phenol, resorcinol and hydroquinone in air samples by synchronous fluorescence using partial least-squares (PLS). Talanta 69(5): 1265-1268.
- Yang L, Wang Z, Xu L (2006) Simultaneous determination of phenols (bibenzyl, phenanthrene and fluorenone) in Dendrobium species by high performance liquid chromatography with diode array detection. J Chromatogr A 1104(1-2): 230-237.
- Du J, Li Y, Lu J (2001) Flow injection chemiluminescence determination of polyhydroxy phenols using luminol-ferricyanide/ferrocyanide system. Talanta 55(6): 1055-1058.
- Zargar B, Hatamie (2012) A Colorimetric determination of resorcinol based on localized surface plasm on resonance of silver nanoparticles. Analyst 21(22): 5334-5338.
- Manjunatha JG (2018) A novel voltammetric method for the enhanced detection of the food additive tartrazine using an electrochemical sensor. Heliyon 4 (11): e00986
- Chenthattil Raril, Jamballi G Manjunatha (2018) Carbon Nanotube Paste Electrode for the Determination of Some Neurotransmitters: A Cyclic Voltammetric Study. Mod Chem Appl 6(3): 3.
- Manjunatha JG (2018) Highly Sensitive Polymer based Sensor for Determination of the Drug Mitoxantrone. J Surface Sci Technol 34(1-2): 74-80.
- Chenthattil Raril, Jamballi G Manjunatha, Lingappa Nanjundaswamy, Gurumallappa Siddaraju, Doddarasinakere K Ravishankar, et al. (2018) Surfactant Immobilized Electrochemical Sensor for the Detection of Indigotine. Anal Bioanal Electrochem 10 (11): 1479-1490
- Manjunatha JG, Kumara Swamy BE, Mamatha GP, Raril C, Nanjunda Swamy L,et al. (2018) Carbon Paste Electrode Modified with Boric Acid and TX-100 used for Electrochemical Determination of Dopamine. Mater Today Proc 5(10): 22368-22375.
- Manjunatha JG (2017) Electroanalysis of estriol hormone using electrochemical sensor. Sens Bio-Sensing Res 16: 79-84.
- Manjunatha JG (2018) A novel poly (glycine) biosensor towards the detection of indigo carmine: A voltammetric study. J Food Drug Anal 26(1): 292-299.
- Jamballi G Manjunatha, Bahaddurghatta E Kumara Swamy, Mohamad Deraman (2013) Electrochemical Studies of Dopamine, Ascorbic Acid and Their Simultaneous Determination at a Poly (Rosaniline) Modified Carbon Paste Electrode: A Cyclic Voltammetric Study. Anal Bioanal Electrochem 5(4): 426-438
- Periyalagan Alagarsamy , Navaneetha Krishnan , Shen Ming Chen , Thangavelu Kokulnathan , Tse-Wei Chen, et al. (2017) A Disposable Single-Use Electrochemical Sensor for Detection of Resorcinol Based on Electrochemically Activated Screen Printed Carbon Electrode in Hair Dyes. Int J Electrochem Sci 12: 6842-6852.
- J G Manjunatha (2017) Surfactant modified carbon nanotube paste electrode for the sensitive determination of mitoxantrone anticancer drug. J Electrochem Sci Eng 7(1): 39-49.
- Sayed Mehdi Ghoreishi, Mohsem Behpour, Elahe Hajisadeghian, Mahshid Golestaneh (2016) Voltammetric determination of resorcinol on the surface of a glassy carbon electrode modified with multi-walled carbon nanotube. Arabian J Chem 9(2): S1563-S1568
- C Raril, JG Manjunatha (2018) Cyclic Voltammetric Investigation of Caffeine at Methyl Orange Modified Carbon Paste Electrode. Biomed J Sci & Tech Res 9(3): XX
- Ya Ping Ding, Wei Li Liu, Qing Sheng Wu, Xiao-GangWang (2005) Direct simultaneous determination of dihydroxybenzene isomers at C-nanotube-modified electrodes by derivative voltammetry. J Electroanal Chem 575 (2): 275-280.
- Yin H, Zhang Q, Zhou Y, Ma Q, Liu T, et al. (2011) Electrochemical behaviour of catechol, resorcinol and hydroquinone at graphene-chitosan composite film modified glassy carbon electrode and their simultaneous determination in water samples. Electrochim Acta 56(6): 2748-2753.
- Zhang W, Zheng J, Lin Z, Zhong L, Shi J, et al. (2015) Highly sensitive simultaneous electrochemical determination of hydroquinone, catechol and resorcinol based on carbon dot/reduced graphene oxide composite modified electrodes. Anal Methods 7: 6089-6094.
- Zhang D, Peng Y, Qi H, Gao Q, Zhang C (2009) Application of multielectrode array modified with carbon nanotubes to simultaneous amperometric determination of dihydroxybenzene isomers. Sens Actuators B 136(1): 113-121.






























