Antioxidant Activity of Aqueous Extracts from Vitis vinifera’s Wastes
Kobra Alijanipoor1, Reza Haji Hosseini1, Reyhaneh Sariri2*, Atusa Vaziri1
1 Department of Biology, Faculty of Sciences, Payame Noor University, Tehran, Iran
2 Department of Biology, University of Guilan, Rasht, Iran
Submission: December 14, 2019; Published: January 22, 2019
*Corresponding author: Reyhaneh Sariri, Department of Biology, University of Guilan, Namjoo Street, Rasht, Iran
How to cite this article: Kobra A, Reza H H, Reyhaneh S, Atusa V. Antioxidant Activity of Aqueous Extracts from Vitis vinifera’s Wastes. Nov Appro Drug Des Dev 2019; 4(5): 555646. DOI: 10.19080/NAPDD.2019.04.555646
Abstract
Background: Antioxidants are biochemical compounds with ability to capture free radicals and reduce their harmful effects to biological molecules. The wide spectrum of side effects exhibited by synthetic compounds has attracted many researchers to replace synthetic compounds with natural counterparts. The use of plant wastes as possible sources of antioxidants could not only provide a nature based source, but it also is an environmental friendly research. Vitis vinifera (common grape) is one of the species from Vitis family native to many parts of the world including Iran.
Methods: The potential of wastes from the common varieties of grape, as a source of natural antioxidants, was studied in this research. The leaves, stems, branches and barks of home grown grape plants were collected in Rasht region. The extract were examined for antioxidant activity by measuring the ferric-reducing antioxidant power (FRAP) and testing the potential of 2,2-diphenyl-1-picrylhydrazyl (DPPH) to scavenge free radicals. The amount of total flavonoids, antocyanins and total phenols were also measured in the mixed waste extracts.
Results: The result of FRAP test was 0.224 ± 0.031 mmol/g DW and DPPH test showed 302.78 ± 5.21 mg/g dry weight (DW). Besides, the amount of flavonoids, anthocyanine and total phenols were 9.98 ± 0.31 mg/g DW, and 0.705 ± 0.014 mg/g DW and 14.56 ± 0.24 mg/g DW of gallic acid equivalent respectively. It was, therefore, concluded that the use of almost priceless residual as a source of antioxidant has beneficial effect not only for management of wastes it could also find possible applications in food and pharmaceutical industries.
Keywords: Grape, aqueous extracts, Vitis vinifera, waste management, secondary metabolites.
Abbrevations: FRAP: Ferric-Reducing Antioxidant Power; DPPH: 2,2-DiPhenyl-1-PicrylHydrazyl; ROS: Reactive Oxygen Species; DW: Dry Weight; DNA: Deoxyribonucleic Acid; TPTZ: TriPyridylTriaZine; GAE: Galic Acid Equivalent; BSA: Bovine Serum Albumin; UV-Vis: Ultra Violet-Visible (UV-Vis); SD: Standard Deviation; AEAC: Antioxidant activity Equivalent Ascorbic Acid; QEs: Quercetin equivalents; SAS: Statistical Analysis System; PPO: Poly Phenol Oxidase.
Introduction
Reactive oxygen species (ROS) are free radicals produced by various natural metabolic pathways in the human body. They are also the result of exogenous chemical reactions which eventually enter the body [1]. They can be beneficial as signal transducers and growth regulators, if produced or used with normal doses [2]. However, large amounts of ROS can be dangerous to the system as they are capable of oxidizing bio-molecules including proteins, lipids and deoxyribonucleic acid (DNA). The high amounts of ROS can initiate different degenerative diseases such as liver cirrhosis, various cancers, neurological disorders, atherosclerosis and arthritis [3]. The presence of natural antioxidants in cells such catalase, glutathione peroxidase and superoxide dismutase can reduce the effect of ROS. Their harm can also be overcome by using exogenous antioxidants including α-tocopherol, β-carotene, ascorbic acid, glutathione and uric acid [4,5].
Plant derived wastes including stems, leaves, barks, dead flowers as well as low quality fruit parts and leftovers from fruit processing industries are natural sources of secondary metabolites. If used in a proper way, they could be valuable sources of low price natural antioxidants. This could also provide a logical strategy to make best of them and control environmental pollution from their deterioration. Depending on the plant type, the part to be utilized and the way they are extracted the wastes may contain inorganic and organic secondary metabolites. For example, the content of iron can help to relieve anemia and have the power to counteract calcium loss and leading to bone strengthening. Considering the high volume of wastes produced in autumn from various plants and agricultural processes, the aim of this study was to investigate and compare the antioxidant activity of extracts from residual leaves, stems and barks of grape plant after its fruiting stage.
Vitis vinifera (common grape vine) is a member of Vitis that is grown in east and north parts of Iran. The plant could grow to about 32 m in length and its barks are flaky. The fruit is considered as a berry and best known as grape with a wide range of color and size. In cultivated plants, the fruit is quite large, up to 3 cm long, with red, green and purple (black) in color. Traditionally, the grape is used as a fresh fruit, processed to make wine or juice, or dried to produce raisins. Consumption of natural antioxidants including fresh fruits and vegetables has protective effects against various diseases. This effect is mostly related to the presence of several components such as vitamins, flavonoids, anthocyanins and other phenolic compounds [6,7].
Studies on natural antioxidants are mostly focused on activity of fruits and vegetables [8,9]. However, getting information on the antioxidant activity of their wastes has attracted researcher during the last few decades. Many agricultural activities and food processing industries produce a large body of wastes including leaves, barks, peels and residuals of food factories. These types of natural wastes are not used or recycled as they have not yet found a beneficial commercial application [10]. However, it has been reported that peel and seed fractions of some fruits have higher antioxidant activity than the pulp fractions [11]. For example, a higher antioxidant activity has been found for pomegranate peel than its pulp [12]. Grape seed is higher than its pulp in antioxidant capacity and is a rich source of proanthocyanin, which is very effective in scavenging various ROS [13].
Materials and Methods
All types of wastes from various grape plants were collected, with permission of owners, from houses and gardens in Rasht. After separation of the contaminants, they were washed and dried at 37°C and kept in sealed plastic bags until experiments. Buffers and solutions were freshly prepared in our laboratory. General chemical reagents including ethanol, acetone, sodium carbonate, acetic and hydrochloric acids were supplied by their local representatives. Folin-Ciocalteu’s phenol reagent, tripyridyltriazine (TPTZ), 2, 2-diphenyl-1-picrylhy-drazyl (DPPH), gallic acid, ferric and aluminum chlorides were obtained from Merck (Darmstadt, Germany). Bovine serum albumin (BSA) was purchased from Sigma (St. Louis, MO, USA).
Preparation of Aqueous Extracts
Various wastes of the grape plant were mixed and ground into a fine powder and incubated in distilled water for two days, at room temperature. The mixture was filtered using Whatman paper and the filtrate was concentrated by rotary evaporator at 40°C. The extract was then stored in a desiccator at 4°C until use.
Ferric Reducing Antioxidant Power (FRAP) Test
For this purpose, a procedure based on the ability of extracts to reduce ferric ions was used [13]. The test was started by preparing the FRAP reagent in sodium acetate buffer (300 mmol/L, pH 3.6), Tripyridyltriazine (TPTZ) (10 mmol/L in 40 mmol/L HCl) and FeCl3 solution (20 mmol/L) 10:1:1 (v:v:v) proportion. A 0.05 ml volume of mixed waste extracts prepared in distilled water was added to 1.5 ml of FRAP reagent. The mixture was stored at room temperature for 30 minutes and its absorbance measured at 593nm. The control solution was a mixture of 0.05ml distilled water and 1.5ml FRAP reagent. The antioxidant capacity was determined from the calibration curve in mmol FeSO4 equivalents per one gram of sample’s dry weight (μmol FeSO4/g DW).
DPPH Assay
A slightly modified method described in [14] was used to evaluate the antioxidant capacity of extracts on 1, 1-diphenyl-2- picrylhydrazyl (DPPH) radical. In practice, aliquots of extracts were mixed with 2ml of DPPH methanolic solution (0.004%). A control tube was also prepared containing 2ml DPPH and 2ml methanol. Both mixtures were mixed thoroughly and kept in the dark for 30 min. The absorbance at 517nm was measured against a blank of methanol without DPPH. The inhibition rate (%) of the DPPH radical was then calculated from the following equation.
Inhibition rate of DPPH (%) = (A control - A sample ) / A control ×100
A plot of extract concentrations versus inhibition rate (%) of the DPPH radical was used to evaluate IC50. The results were expressed as antioxidant activity equivalent ascorbic acid as standard (AEAC).
AEAC (mg AA/G DW) = IC50 ascorbate /IC50 sample×1000
Flavonoids
Flavonoid content was determined using a modified method of Chang [15]. In practice, 500μL from each extracted sample was mixed with 1.5ml methanol (%85), 100μL of %10 aluminum chloride methanolic solution, 100μL of 1 M potassium acetate solution and 2.8 ml distilled water. The resulting solution was incubated for 40 minutes at room temperature and its absorbance was measured at 415 nm. Total flavonoid content of mixed grape wastes was calculated using a standard curve of quercetin and expressed as mg quercetin equivalents (QEs)/g DW.
Anthocyanins
To measure total anthocyanin content of extracts, 0.02 grams of each sample was mixed with 4 ml 1% methanolic solution and stored at 4°C for 24. The mixture was then centrifuged at 13000 rpm for 10 min and the 530 and 675 nm absorbances of supernatant were read on UV-Vis spectrophotometer. The anthocyanin content for each extract was calculated from the following equation [16].
Anthocyanin content (mg/g DW) = A530 – (0.25 × A675)
Total Phenols
The colorimetric Folin-Ciocalteu method was used to measure the content of phenolic compounds. In brief, 100μL of extracts were added to 100μL of Folin-Ciocalteu reagent (%50) and mixed thoroughly. A mixture of 2ml of sodium carbonate (%2) and 2.8ml deionized water was then added and well mixed. The resulting solution was incubated at room temperature for 30 minute and its absorbance was read at 720 nm on a ultra violet- visible (UV-Vis) spectrophotometer. Total phenol content was calculated using a standard curve of aqueous solutions of gallic acid and expressed as mg gallic acid equivalents (GAE)/g DW [17].
Statistical analysis
Statistical analysis system (SAS) for Windows version 9.1software was used to statistically analyze the results. Each experiment was repeated at least three times and the data were reported as mean ± standard deviation (SD) of three replicates. Differences between means were determined by One-Way ANOVA using Tukey’s test. Statistical significance was defined at a level of P ≤ 0.05.
Results and Discussions
As mentioned above, FRAP test the ability of extracts is measure through the power to reduce the ferric ions. The results from FRAP experiment are shown in Table 1. As evident from these data, the antioxidant activity obtained by this assay was 0.224 ± 0.031 mmol/g DW. Similar to our experiments, the assay has been used to compare antioxidant power in peels, pulps and seeds of some fruits [17]. They have reported that fruit peels have stronger antioxidant power than seeds. The FRAP method has been slightly modified to measure the antioxidant power of celandine extracts [18]. They reported that the extract had a considerable power to reduce ferric ions, a result that agrees with our findings. We recommend the FRAP assay as a precise and straight forward method for measurement of antioxidant capacity in many natural sources including wastes from plants after harvesting.
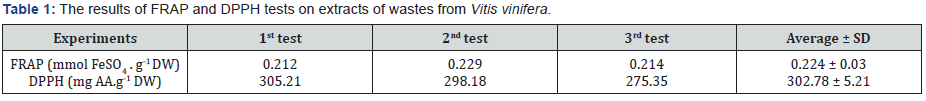
The DPPH assay is based on the ability of the extract to scavenge free radicals through donation of hydrogen. DPPH is a stable free radical that is reduced to diphenyl picryl hydrazine through a reaction with antioxidant compounds [14]. The initial reaction mixture has a deep violet color that is changed to light yellow after reduction of DPPH. As presented in Table 1, the aqueous extracts from grape plant leftovers had a considerable power to scavenge DPPH. The average of three tests for DPPH, showed an activity of 302.78 ± 5.21 mg/g dry weight. In support of our experiments, the antioxidant power to scavenge DPPH free radical has been used for determination of antioxidant capacity in a number of medicinal and aromatic plant extracts [19]. The effect of far infrared radiation on antioxidant activity of rice hulls has been investigated by this method [20]. In the present research, we found that the method was suitable for determining the antioxidant capacity in wastes extracts from leaves and various parts of grape plant. We have previously modified DPPH method to determine antioxidant capacity in human saliva [21,22].
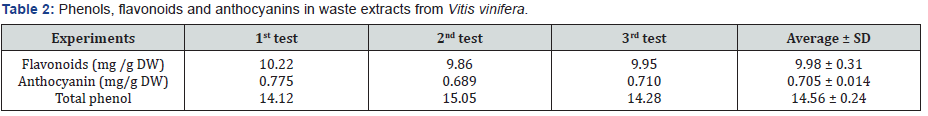
It is worth indicating that radical scavenging activity of plant extracts is partly due to the presence of flavo noids and total phenols. All experiments were repeated in triplicate and the results calculated as the average obtained from three test. In Table 2 amounts of flavonoids, anthocyanins and total phenols are presented. According to these data, flavonoids content was 9.98 ± 0.31 mg/g DW, anthocyanins 0.705 ± 0.014 and total phenol content 14.56 ± 0.24 mg/g dry weight of gallic acid equivalent. A number of research works have reported that leftovers of fruit juicing factories contain considerable amounts of flavonoids, anthocyanins and phenols. In support of our results, it has been found that pomegranate peels contain considerable antioxidant activity mostly due to the content of anthocyanins [11,23]. The results obtained in our research are also in agreement with a previous research within our team [24]. Other research groups have reported the effect of ultrasonic treatment on antioxidant capacity of citrus species [25]. Many beneficial effects are reported for flavonoids found in fresh fruits and vegetables. It is suggested that they could protect human heart when taken as a supplement [11]. That research has emphasized that when flavonoids are taken as drug supplements they highly reduce the mortality rate from coronary heart diseases. Interestingly, the presence of flavonoids plays a major role in the traditional benefits known for honey. It has been found that considerable amounts of flavonoids exist in propolis [15] and honey [17]. In the present research, the total anthocyanin content of water extracts from grape plant’s wastes was also evaluated. Although only a few reports were found on the use of agricultural wastes as a source of antioxidants, our results could be compared to other wastes. For example, the antioxidant activity in seeds of some fruits has been reported to be mostly due to total phenols [10]. The medicinal benefits of fruits and their natural juices is a well-known fact and have been studied specifically. For example the hepatoprotective effect of orange juice on the injuries caused by carbon dioxide has been studied in detail [8].
The extraction conditions used to obtain antioxidant capacity by plant wastes as well as the environmental conditions are determinant factors on results [26]. In most cases, methanol is reported as the solvent of choice for extraction of phenolic compounds. This is reported to be due to its ability to inhibit the reaction of poly phenol oxidase (PPO) that causes oxidation of phenolic and its ease of evaporation as compared to water [27]. On the other hand, when compared to a non-polar solvent such as acetone, both methanol and ethanol offer better ability for extraction of phenolic compounds [19]. Our flavonoid results are in contrast to what was found by other researchers who reported that value was very low compared to their other antioxidant experiments [19]. However, total flavonoid content of some fruit wastes has been studied by another research team and their results agree with our findings [15].
Conclusion
In this research, antioxidants were measured in aqueous extracts obtained from mixed wastes of various grapes grown in Rasht. The results indicated total phenol content of about 15 mg/g DW, total flavonoids of around 10mg/g DW and total anthocyanin in the range of 0.7mg/g DW which are considerable values when compared to the main parts of fruits and vegetable. Besides, considering the very low price of grape wastes, their high volume and the benefit this action provides for waste management, the design of a pilot plant seems necessary.
Acknowledgements
We would like to thank both University of Guilan and Tehran Sharjah Payame Noor University.
References
- Halliwell B, Gutteridge JM (1984) Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 219(1): 1-8.
- Henry J, Martine T (2002) Reactive oxygen species and cell signaling. Am J Respir Crit Care Med 166: S4-S8.
- Martinez-Cayuela M (1995) Oxygen free radicals and human disease. Biochimie 77: 147-161.
- Halliwell B, Gutteridge JM (1990) Role of free radical and catalytic metal ions in human disease: an overview. Methods Enzymol 186: 1-85.
- Cheeseman K, Slater T (1993) An introduction to free radical biochemistry. Br Med Bull 49: 481-493.
- Klimczak I, Małecka M, Szlachta M, Gliszczyńska-Świgło A (2007) Effect of storage on the content of polyphenols, vitamin C and the antioxidant activity of orange juices. J Food Compos/ Anal 20: 313-322.
- Knekt P, Jarvinen R, Reunanen A, Maatela J (1996) Flavonoid intake and coronary mortality in Finland: a cohort study. Brit Med J 312: 478-481.
- Valcheva-Kuzmanova S, Borisova P, Galunska B, Krasnaliev I, Belcheva A (2004) Hepatoprotective effect of the natural fruit juice from Aronia melanocarpa on carbon tetrachloride-induced acute liver damage in rats. Experi Toxicol Pathol 56: 195-201.
- Mokbel MS, Hashinaga F (2006) Evaluation of the antioxidant activity of extracts from buntan (Citrus grandis Osbeck) fruit tissues. Food Chem 94: 529-534.
- Soong YY, Barlow PJ (2004) Antioxidant activity and phenolic content of selected fruit seeds. Food Chem 88: 411-417.
- Jayaprakasha GK Singh RP, Sakariah KK (2001) Antioxidant activity of grape seed (Vitis Vinefera) extracts on peroxidation models In vitro. Food Chem 73: 285-290.
- Lim Y, Guo C, Yang J, Wei J, Xu J, Cheng S (2006). Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. Food Chem 96: 254-260.
- Guo C, Yang J, Wei J, Li Y, Xu J, Jiang Y (2003) Antioxidant activities of peel, pulp and seed fractions of common fruits as determined by FRAP assay. Nutri Res 23: 1719-1726.
- Miliauskas G, Venskutonis P, Van Beek T (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 85: 231-237.
- Chang CC, Yang MH, Wen HM Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 10: 178-182.
- Mita S, Murano N, Akaike M, Nakamura K (1997) Mutants of Arabidopsis thaliana with pleiotropic effects on the expression of the gene for β‐amylase and on the accumulation of anthocyanin that is inducible by sugars. Plant J 11: 841-851.
- Meda A, Lamien CE, Romito M, Millogo J Nacoulma OG (2005) Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem 91: 571-577.
- Then M, Szentmihályi K, Sarkozi A, Varga IS (2003) Examination on antioxidant activity in the greater celandine (Chelidonium majus L.) extracts by FRAP method. Acta Biologica Szegediensis 47: 115-117.
- Moure A, Cruz JM, Franco D, Dominguez JM, Sineiro J, et al (2001) Natural antioxidants from residual sources. Food Chem 72: 145-117.
- Lee SC, Kim JH, Jeong SM, Kim DR Nam KC (2003) Effect of far-infrared radiation on the antioxidant activity of rice hulls. J Agri Food Chem 51: 4400-4403.
- Azadbakht M, Sariri R, Molasoltani F, Ghafoori H, Aghamaali MR, et al. (2016) Salivary antioxidant power of passive smokers. J Nanomed Biotherap Discov 2: 142-148.
- Moori M, Ghafoori H, Sariri R (2016) Nonenzymatic antioxidants in saliva of patients with systemic lupus erythematosus. Lupus 3(25): 265-272.
- Negi PS, Jayaprakasha GK, Jena BS (2003) Antioxidant and Antimutagenic activities of Pomegranate Peel Extracts. Food Chem 80: 393-397.
- Nasuti S, Sariri R, Aghamaail MR, Ghafoori H, Shahmohamadi R (2011) In vitro antioxidant activity of extracts from wastes of five Iranian citrus species. Pharmacologyonline 3: 853-858.
- Ma YQ Chen JC, Liu DH, Ye XQ (2008). Effect of ultrasonic treatment on the total phenolic and antioxidant activity of extracts from citrus peel. J Food Sci 73: 115-120.
- Singh RP, Murthy KNC, Jayaprakasha GK (2002) Studies on the antioxidant activity of pomegranate (Punicagranatum) peel and seed extracts using in vitro J Agri Food Chem 50: 81-86.
- Yen GC, Chen HY (1994) Comparison of anti-mutagenic effect of various tea extracts (green, oolong, pouchong and black tea). J Food Protec 57: 54-58.






























