Design and Synthesis and In-Vitro Assay Of Substituted 2-Aminobenzamide Derivatives
Vivekanand A Chatpalliwar1*, Ravindra R Patil2, Mansi D2 and Saurabh CK2
1SNJB's College of Pharmacy, Savitribai Phule Pune University, India
2RC Patel Institute of Pharmaceutical Education and Research, Shirpur, India
Submission: April 22, 2017; Published: November 22, 2017
*Corresponding author: Vivekanand A Chatpalliwar, SNJB's College of Pharmacy, Savitribai Phule Pune University, India, Email: vchatpalliwar@yahoo.co.in
How to cite this article: Vivekanand A C, Ravindra R P, Mansi D, Saurabh CK. Design and Synthesis and In-Vitro Assay Of Substituted 2-Aminobenzamide Derivatives. Nov Appro Drug Des Dev. 2017; 3(2): 555609. DOI: 10.19080/NAPDD.2017.03.555609
Abstract
Glucokinase has been unearthed in the past decade to be a strategic drug target for T2DM as it is implemented dually in increasing pancreatic insulin secretion and enhancing hepatic glucose metabolism. The research carried was to design, conduct in silico interaction studies, synthesize few of the accepted benzamide derivatives as GK activators (GKA) on the basis of the SAR studies and there in silico interactions with the enzyme. All molecular modeling studies were performed using the Molecular Design Suite (VLife MDS software package, version 3.5), and LigPrep (v2.2) module in the Schrodinger software molecular simulations. RCSB/PDB site was explored to study the simulated protein structures available and 1V4S-representing the simulated protein. Ligands from the studies were selected for synthesis, characterized, and subjected to in vitro glucokinase assay. Detail synthetic procedures, purifying methods, characterizations of the synthesized compounds, methodologies of in vitro assays, and the inferences drawn from the ensued observations are discussed at large. The results of the in silico studies and in vitro assay are presented.
Keywords: Glucokinase (GK); T2DM; GK activators (GKA)
Introduction
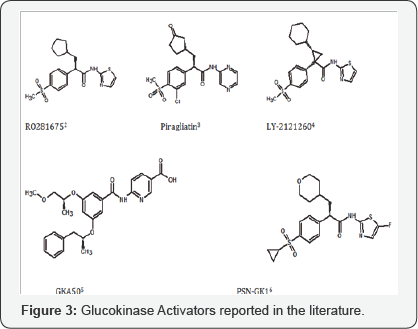
Maintenance of glucokinase enzyme (GK) in its active conformation by small molecule glucokinase activators has been the mainstay of anti-diabetic research in the last decade as an anticipation to produce compounds that would achieve severe glycemic control in diabetic patients. Serendipitous discovery of a phenylacetamide derivative to bind effectively with the glucose phosphorylating enzyme led to a research devoted to similar small molecules that could bind with the enzyme, in several in-vitro tests, and also lower the blood glucose levels in diabetic animal models. Owing to its important role in glucose homeostasis, several small molecule activators of GK were designed, synthesized and their anti-diabetic potential was widely reported [1].
Literature reveals reports on such trials carried with molecules chemically classified as benzamide derivatives. The co-crystallization, of ligand and the enzyme with co-activators, and its X-ray crystallography studies could establish the prime features of the ligand structure that interact with amino acid residues and bind in allosteric site of GK and maintain its conformation in the active mode. Accordingly, the carbonyl function and the imino nitrogen in the thiazole ring, present adjacent to amide group, are together involved in anchoring the ligand molecule to ARG63 residue of GK. The peptide linkage between ARG63 and VAL62 act as proton donor and proton acceptor, respectively, whereas, the amide oxygen acts as proton acceptor and the nitrogen atom in adjoining hetero cycle acts as proton acceptor.
The role of thiazole ring in binding with ARG63 of the hinge region of GK has been extensively studied [2-7]. Toxic metabolite, thiourea derivative, generated from thiazole ring was limiting further development of molecules containing thiazole moiety. Incorporation of a methyl group at position C-4 on the hetero cycle abolishes the possibility of generating toxic metabolite [8]. The methyl group was reported to provide certain rigidity to the ring against certain metabolic reactions that led to formation of thiourea derivative.
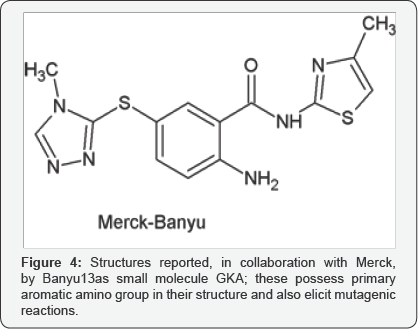
Screening of benzamide derivatives carrying 4-methylthiazole attribute further led to design and synthesis of several ortho-substituted benzamide derivatives that primarily contain amino function. Amino hydrogen was stated as being involved in hydrogen bonding interaction with TYR215, an amino acid residue in the active cavity responsible for allosteric binding of the ligand. Albeit such molecules have proven potent activators of GK in the in vitro assays, and also were reported [9,10] to be effective in lowering the blood-glucose levels in diabetic animal models, these could not progress further due to the mutagenic liabilities associated with them.
It is a well-known fact, since ages, that primary aromatic amine is extremely toxic, owing to its nucleo philic character. The linked mutagenic effects [11,12] can be attributed to the primary amino function in these molecules. Present paper describes the endeavours taken to find a suitable bio isosteric replacement of the primary amino group. Taking into consideration the successful trials carried world-wide with benzamide derivatives possessing 4-methylthiazole group, structures of new molecules were designed to contain these templates while substituting the primary amino function with some suitable group so as to interact and bind with amino acid residue in allosteric site of GK. Considering the process involved in catalytic activity of the enzyme during phosphorylation of glucose was thought imperative during designing new activators. Physiologically, at higher glucose in blood, the GK transforms into active conformation -the closed form, and at lower concentrations of glucose, the enzyme remains in less active form -in super open form. Respectively, the closed form converts glucose to glucoe- 6-phosphate at a higher rate than in the super-open form at lower concentrations of glucose [13-15]. Studies detailing crystal structure reveals the process of GK transformation from super-open (less active) to closed conformation. Accordingly, the transformation starts with binding of substrate, i.e. glucose, with few hydrophilic side chains of amino acid residues in the catalytic site. This induces the conformational change and enables it to bind the co-factors, ATP &Mg-2, finally leading to phosphorylation of glucose molecules, releasing glucose-6- phosphate and ADP.
Eventually, as the concentration of glucose decreases, the enzyme reverts back to its super-open form to operate at a much lower rate. The amino acid residues involved in the catalytic activity are ASP78, GLY80, SER151, ASN204, ASP231, GLU256, THR168, LYS169, GLY229, THR228, THR82, SER411, GLY295 and SER336 [16]. The small molecule glucokinase activators (GKA) are reported to bind with GK in its closed conformation, when amino acid residues in allosteric site become accessible for binding [17]; a site situated 20A away from the catalytic site. Binding of GKA is implemented in maintaining the enzyme in active conformation for sustained period. This results in decreasing intracellular glucose concentrations, thereby increasing the utilization of glucose.
The amino acid residues in each hydrophobic pocket [18] are enlisted below. The three different hydrophobic components of GKA bind in these three pockets [19]. Designing new GKA required concentrating on the cavity comprising these three hydrophobic pockets; also the interactions of components of molecular structure with the above mentioned amino acid residues was considered crucial. Possibility of substituting the amino hydrogen with non-polar alkyl group, nitro group, or a hydrogen atom was ruled out as it would abolish the hydrogen bonding interaction between the resultant ligands and TYR215. The same has been observed by some groups as indicated by the decline in potency of compounds with such modifications [20]. Hence, it was hypothesized to substitute the primary amino group with such an isostere that would interact with the hydroxyl group of TYR215.
Molecular Docking Studies
Molecular docking studies were performed on two series of compounds using two software's. For compounds in Figure 1, docking studies were initially carried Molecular Design Suite of VLife Sciences, later in Schrodinger software, whereas, for compounds in Figure 2 Schrodinger software was used. Crystal structure of GK (PDB entry 1V4S) was used in the simulations in both the software's, and was maintained in flexible mode.
As a part of SAR studies on new GKA without the o-amino substitution, structures were designed and simulated in soft as Mol2 files for compounds in Figure 1, and LigPrep (v2.2) module in the Schrodinger software molecular simulations, where low energy conformations of all ligands were prepared using OPLS force field for compounds in Figure 2.
Ligands were docked into the active site of 1V4S. The best suited conformations of few ligands, from both the software's, which were successful in reversing the protein in its original conformation, and produced maximum dock score were studied precisely.
Chemistry
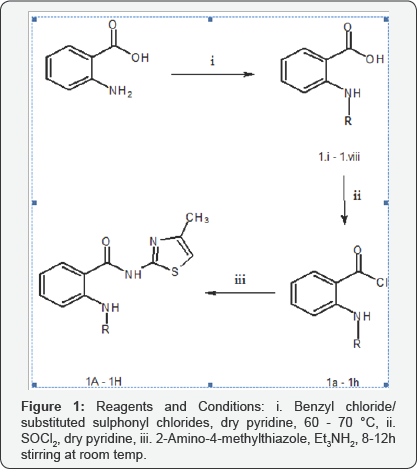
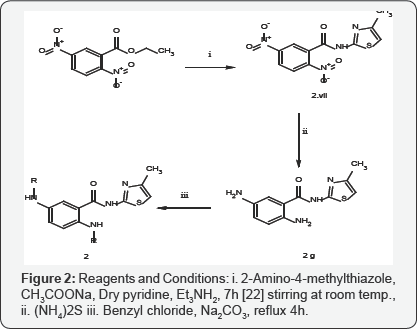
Target compounds were prepared by executing two schemes of reactions. Compounds in Series 1 were prepared from 2-aminobenzoic acid by amidation and acetylation of the amino group, followed by ammonolysis, via chlorination, of the carboxyl group, to yield compounds I.A-I.H. Compounds in Series 2 were obtained in three steps via ammonolysis, reduction, followed by mesylation/benzenesulphonation/tosylation/ aminosulphonation/benzylation/acetylation, respectively. Commercially available Ethyl 2, 5-dintrobenzoate was converted to N-(4-methyl-1, 3-thiazol-2-yl)-2, 5-dinitrobenzamide by treating the former ester with 2-amino-4-methylthiazole in cool conditions. The ammonolytic reaction resulted in the amide. Reducing the nitro groups with Fe, acetic acid yielded the expected diamino compound, which was treated with different acid chlorides in dry pyridine to yield compounds II.A- II.H Figures 3 & 4.
In vitro glucokinase assay
Few of the synthesized compounds were subjected to in-vitro glucokinase assay, studied at two different glucose concentrations, 2.5mM and 10mM, which simulated the low and high blood glucose conditions, respectively. The assay was carried spectrophotometrically (Spetra-Max 190; Molecular Devices, USA) by coupled reaction with glucose-6-phosphate dehydrogenase (G6PDH). The conversion of substrate glucose into glucose-6-phosphate is catalysed by glucokinase; the product is oxidized in-situ by G6PDH, simultaneously reducing nicotinamide adenine diphosphate (NADP) into NADPH. The increase product NADPH was monitored spectrometric ally at 340nm. The assay was performed in 96-well plate with a final volume of 100|iL. The assay fluid contained 50mM HEPES (pH 7.4), 5mM glucose, 25mM KCl, 2mM MgCl2, 1mM dithiothreitol (DTT), 1mM ATP, 1mM NADP, 2.5U/mL G6PD H, 0.5^g Glucokinase (HIS6). Different concentrations of the compounds (synthesized molecules and RO-281675 and PSN-GK1 as standards) were tested in the assay to determine their EC50 [21].
Discussion
The known fact about the toxicity of primary amino group, and the importance of this group in binding interactions with TYR215 in the allosteric site of GK, prompted to find some isosteric replacement to generate bioisosteres, without the amino function to generate safe GKA so that their usefulness would not be restricted owing to amino toxicity. Also, binding of those ligands were envisaged to be a result of binding interactions with same amino acid residues as the standard-the small molecule GKA RO-28-1675 and PSN-GKl-do.
The docking studies, carried in Biopredicta module, with compounds in first series (IA-IH) and standards structures RO281675 and PSK-GK1 provided results that indicated expected interactions of the ligands in the allosteric site of GK. The dock score was compare with the standards. Hence, the molecules were synthesized in synthetic laboratory and were subjected to in-vitro glucokinase assay. Surprisingly, the results of the assay were far from expectations. It ensured that the structures were lacking the third hydrophobic group to nestle in the third hydrophobic pocket (discussed earlier). On the contrary, docking studies of same molecules with the protein in Schrodinger demonstrated very little interaction with the protein in its allosteric site; the dock score was poor. Hence, the molecules were redesigned to contain three domains over benzamide scaffold keeping in mind the required substitution for primary amino group. Molecular simulations of the redesigned molecules with same protein in Schrodinger yielded proper dock score in comparison with the standard structures. Accordingly, the standard structure appeared to have interacted with polar side chains of amino acid residues present in the hinge region, quite reminiscent of the interactions reported for small molecule GKA (Figure 5). Apart from usual interactions with ARG63, the pyrazole nitrogen binds with the alcoholic hydrogen on THR65 and the hydrogen atom of -o-amino group interacts through hydrogen bond with the oxygen atom of TYR215. Surprisingly, none of the designed ligands in series II showed similar interactions with amino acid residues on GK, although dock score of one of the ligands, II. G., was quite close to the score of the standard (Table 1). This ligand displayed altogether different interactions (Figure 6). The much important hydrogen bond interactions, involving ARG63 and TYR215 were absent, but phenyl rings on benzyl groups on 2- and 5-positions of benzamide core were shown to be involved in hydrophobic interactions with TYR215, GLY448, TYR61 and PRO66. Probably, the bulkiness of both the phenyl groups prohibits the amide linkage and the ortho-amino hydrogen to reach closer to the interacting amino acid residues, ARG63 and TYR215, respectively. However, owing to its high dock score, it was thought worth to synthesize the ligand (II G.) only and subject it to in vitro glucokinase assay [22].
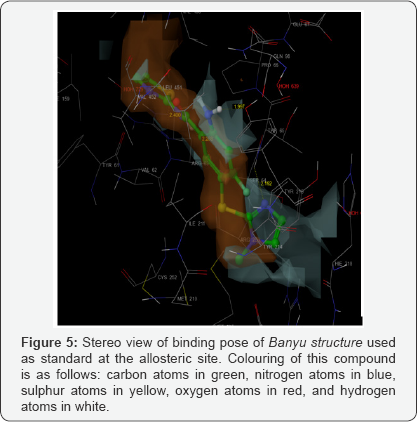

*It is this pocket that activator molecules are pivoted in the allosteric site through hydrogen bonding with the side chain of ARG63.
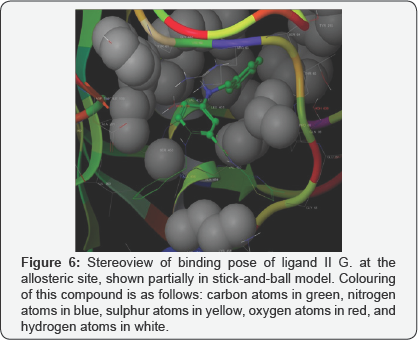
Substituting one hydrogen atom on the o-amino function with a benzyl group appeared worthful, as indicated by the comparable dock score (Table 1) and EC50 nearing equal to that of PSN-GK1 (Table 2). The same cannot be said for the sulphamoyl and acetyl substitution for the amino hydrogen, as obtained dock score for compounds with these substitutions were way far different. These findings were evocative of tolerance for benzyl group on o-amino group. These observations suggest that the groups at position 5 on benzamide scaffold are required to be aromatic and hydrophobic, and be spaced enough from the benzamide- aromatic ring so as to reach and interact with the hydrophobic clouds of PRO66.
Few of the designed ligands were rejected -was decided not to proceed for their laboratory synthesis- on the basis of poor interaction with the amino acid residues in allosteric site of the enzyme, and also on the basis of poor dock score. A criterion for such rejection was set at -7.0 dock score. Structure of ligands that show scores higher than the cut-off were synthesized.
As depicted in the figure below, Ligand 2.1 -designed to replace one amino hydrogen atom with methyl suphonyl substituent on the o-amino function of benzamide, showed no interaction with the hydroxyl group on Tyr215, resulting in poor dock score (Glide Score -3.76588).
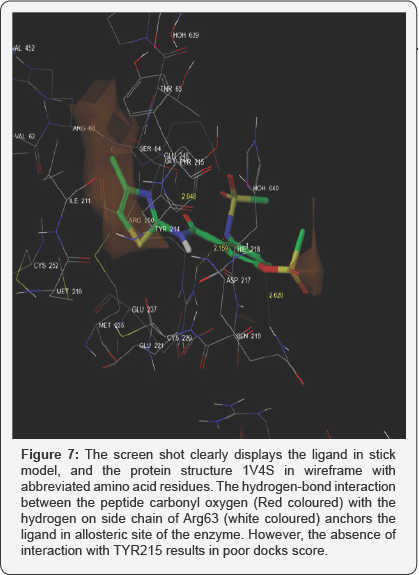
All commercially purchased solvents and reagents were purified and dried [23,24] prior to their use. Characterizations of the synthesized compounds are mentioned in individual description. Melting points reported here in are uncorrected. Silica gel G plates activated at 110°C for 30min were used for TLC, developed using solvent system: (a) chloroform : ethyl acetate: 1 drop of acetic acid [3:7v/v], and are mentioned herein individual characterization of the compound. The developed TLC plates were examined for colored spots that were apparent on exposing them to iodine vapours. The Rf values of purified compounds are represented here in individual characterization. Single spot TLC, using various solvent systems, ascertained purity of the compounds Figure 7.
Percentage yields are reported in brackets along with melting point (m.p.) reported in °C. UV spectra were recorded on Chemito Spectrascan UV-2600. IR spectra were recorded in ATR on JASCO 4100 FTIR from sophisticated instrument laboratory of our institute and are reported here in cm-1. NMR spectra of compounds were obtained from SAIF, Manipal University, Manipal, on Bruker NMR Spectrometer, as well as from SAIF, CIL, Punjab University, Chandigarh, and are reported herein as ppm. Data of elemental analyses were obtained from IIT, Pawai, Mumbai, on Thermo Finnigan and are expressed as percentage of respective molecular weights in the individual description of the compounds mentioning the calculated value in parentheses following the practical value.
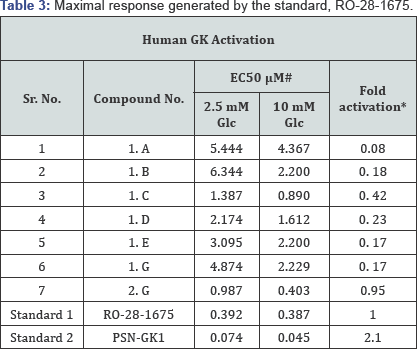
# Values are mean of three assays
*Calculated considering the maximal response generated by the standard, RO-28-1675, as 1
2-[(Methylsulfonyl) amino] benzoic acid [25]: A solution of 2-aminobenzoic acid (0.01 mol) in aqueous NaOH (10 %, 60 mL), was treated with sulphonyl chloride (0.015 mol) under reflux in a water bath for 15-40 min with stirring. The solution was heated under an air condenser to remove the excess of acid chloride, and was neutralized with sufficient dilute hydrochloric acid to yield the sulphonated product as a precipitate. The product was collected at pump, washed with ample water and crystallized from rectified spirit. m. p.: 186-90 °C (yield: 65 %); Rf: 0.68; λmax.: 299 nm (MeOH), IR: 3208.00cm-1 (N-H str.), 2934.16 cm-1 (C-H str.), 1660.41cm-1 (C=O str.), 1325.82cm-1 (assym. str. S=O), 1254.47cm-1(C-N str.), 1173.54cm-1 (S=O sym. str.), 1143.58cm-1 (C-O) (Table 3).
2-[(Phenylsulphonyl) amino] benzoic acid: A solution of 2-aminobenzoic acid (0.01 mol) in aqueous NaOH (10 %, 60 mL), was treated with benzenesulphonyl chloride (0.015 mol) under reflux in a water bath for 45 min with stirring. The solution was heated under an air condenser to remove the excess of acid chloride, and was neutralized with sufficient dilute hydrochloric acid to yield the sulphonated product as a precipitate. The product was collected at pump, washed with ample water and crystallized from rectified spirit. m. p.: 218-22 °C (yield:70 %); Rf: 0.66; λmax: 293nm (MeOH), IR: 3629.37 cm-1 (O-H str.) 3108.69cm-1 (N-H str.), 2931.27cm-1 (C-H str.), 1701.84cm-1 (C=O str.), 1357.64cm-1 (asym. str. S=O), 1297.86cm-1 (C-N str.) 1169.62cm-1 (S=O sym. str.), 1123.33cm-1 (C-O str.) [26].
2-{[(4-Methylphenyl)sulfonyl]aminob}benzoic acid [26]: A solution of 2-aminobenzoic acid (0.01mol) in aqueous NaOH (10 %, 60mL), was treated with 4-methylphenylsulphonyl chloride (0.015 mol) under reflux in a water bath for 45min with stirring. The solution was heated under an air condenser to remove the excess of acid chloride, and was neutralized with sufficient dilute hydrochloric acid to yield the sulphonated product as a precipitate. The product was collected at pump, washed with ample water and crystallized from rectified spirit.
m. p.: 209-12 °C (yield:56 %), Rf 0.58; λmax: 293nm (MeOH), IR: 3634.63cm-1 (O-H str.) 3108.6cm-1 (N-H str.), 2931.07cm- 1 (C-H str.), 1701.8cm-1 (C=O str.), 1350.6cm-1 (asym. str. S=O), 1297.86cm-1 (C-N str.)1169.62cm-1(S=O sym. str.), 1123.33cm-1 (C-O str.)
2-[(Morpholin-4-ylsulfonyl)amino]benzoic acid: A solution of 2-aminobenzoic acid (0.01mol) in aqueous NaOH (10 %, 60mL), was treated with morpholinesulphonyl chloride (0.015mol) under reflux in a water bath for 45min with stirring. The solution was heated under an air condenser to remove the excess of acid chloride, and was neutralized with sufficient dilute hydrochloric acid to yield the sulphonated product as a precipitate. The product was collected at pump, washed with ample water and crystallized from rectified spirit. m. p.: 198°C (yield: 83 %), Rf: 0.76; λmax: 319nm (MeOH), IR: 3629.37cm-1 (O-H str.)3383.50cm-1 (N-H str.), 2939.95cm-1 (C-H str.), 1699.94cm- 1 (C=O str.), 1526.38cm-1 (N-H bend.), 1323.89cm-1 (asym. str. S=O), 1351.86cm-1 (Ar.C-N str.), 1168.72cm-1 (S=O sym. str.), 1122.37cm-1 (C-O) [26].
2-(Sulfamoylamino) benzoic acid: A solution of 2-aminobenzoic acid (0.01mol) in aqueous NaOH (10%, 60mL), was treated with sulphonyl chloride (0.015mol) under reflux in a water bath for 45min with stirring. The solution was heated under an air condenser to remove the excess of acid chloride, and was neutralized with sufficient dilute hydrochloric acid to yield the sulphonated product as a precipitate. The product was transferred to a conical flask and 20mL of conc. ammonia solution was added to it. The flask was allowed to stand at room temperature overnight. The precipitated compound thus formed was collected at pump, washed with ample water and crystallized from rectified spirit m. p.: 223-25 °C (yield:75%), Rf: 0. 47; λmx. 289nm (MeOH), IR: 3629.37 cm-1 (O-H str.) 3108.69 cm= (N-H str.), 3027cm-1 (aromatic C-H str.), 2931.27cm-1 (aliphatic C-H str.), 1701.84cm-1 (C=O str.), 1343cm-1 (asym. str. S=O), 1699.94cm-1 (C=O str.), 1169.62cm-1(S=O sym. str.), 1123.33cm-1 (C-O str.) [25].
2-(Benzylamino) benzoic acid: A solution of 2-aminobenzoic acid (0.01mol) and sodium carbonate (3.5g) in water was heated in a water bath. Freshly distilled benzyl chloride (0.02mol) was run into the solution for 1 hour, along with vigorous stirring. The heating was continued for 5 hours, cooled and a liter of water added with stirring. The mixture was filtered and the residue was washed with salt water. The crude benzylated acid was crystallized using rectified spirit [27].
m. p.: 178-81°C (yield: 54%), Rf: 0.73; λmax: 281nm (MeOH), IR: 3629.37cm-1 (O-H str.) 3147.69cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2931.27cm-1 (aliphatic C-H str.), 1701.84cm-1 (C=O str.), 1297.86cm-1 (C-N str.), 1123.33cm= (C-O str.)
Conversion into acid chloride
In a perfectly dry atmosphere 0.01mol of the acids (I. i., I. ii.,I. iii., I. iv., I. v. I. vii.) were dissolved in dry toluene (about 20mL) and the assembly was set for reflux under protection against atmospheric moisture. To this mixture, redistilled thionyl chloride (0.02mol) was slowly added, and the mixture was heated for 30-45min. The mixture was subjected to distillation at reduced pressure to remove the unreacted thionyl chloride and the solvent. The residue was dried under vacuum [28].
2-[(Methylsulphonyl)amino]benzoyl chloride: m. p.: 136- 38°C (yield: 46 %) Rf :0.64; λmax: 306nm, IR: 3273.57cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2990.16cm-1(C-H str.), 1764.76cm-1 (C=O str.), 1488.78cm-1 (N-H bend.), 1341.96cm-1 (assym. str. S=O), 1329.68cm-1 (C-N str.), 1339.01cm-1 (S=O sym. str.), 1119.48cm-1 (C-O str.), 749.20cm-1 (C-Cl str.).
2-[(Phenylsulphonyl) amino] benzoyl chloride: m. p.: 187-88°C (yield: 45 %) Rf :0.61; λmax : 306nm; IR: 3273.57cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2990.16cm-1 (C-H str.), 1764.76cm-1 (C=O str.), 1488.78cm-1 (N-H bend.), 1343.08cm-1 (asym. str. S=O), 1329.68cm-1 (C-N str.), 1314.90cm-1 (S=O sym. str.), 1319.48cm-1 (C-O str.), 749.16cm-1 (C-Cl str.).
2-[{(4-Methylphenyl)sulphonyl}amino] benzoyl chloride: m. p.: 191-93 °C (yield: 50%) Rf :0.56; λmax: 306nm; IR: 3273.57cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2990.16cm-1 (C-H str.), 1764.76cm-1 (C=O str.), 1488.78cm-1 (N-H bend.), 1343.08cm-1 (asym. str. S=O), 1329.68cm-1 (C-N str.), 1314.90cm-1 (S=O sym. str.), 1319.48cm-1 (C-O str.), 749.16cm-1 (C-Cl str.).
2-[(Morpholin-4-ylsulfonyl) amino] benzoyl chloride: m. p.: 116-120°C (yield: 48 %) Rf :0.67; Xmax : 305nm; IR: 3383.50cm- 1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1769.94cm-1 (C=O str.), 1526.38cm-1 (N-H bend.), 1323.89cm-1 (asym. str. S=O), 1351.86cm-1 (aromatic C-N str.), 1168.72cm-1 (S=O sym. str.), 1122.37cm-1 (C-O), 749.16cm-1 (C-Cl str.).
2-(Sulfamoylamino) benzoyl chloride: m. p.:172-73°C (yield: 57 %) Rf:0.45; λmax: 324nm; IR: 3383.50cm-1 & 337.89cm- 1(N-H str.), 3027.45cm-1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1769.94cm-1 (C=O str.), 1526.38cm-1 (N-H bend.), 1323.89cm-1 (asym. str. S=O), 1351.86cm-1 (aromatic C-N str.), 1168.72cm-1 (S=O sym. str.), 1122.37 cm-1 (C-O), 749.16cm-1 (C-Cl str.).
2-(Benzylamino)benzoyl chloride: m. p.: 172-73°C (yield:48 % ) Rf :0.40; λmax;. 294nm; IR: 3383.50cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1769.94cm-1 (C=O str.), 1526.38cm-1 (N-H bend.), 1351.86 cm-1 (aromatic C-N str.), 1122.37cm-1 (C-O), 749.16cm-1 (C-Cl str.).
Conversion into amide [22]
Ice cold ethanolic solution of the acid chloride was drop-wise added to a mixture of 2-amino-4-methyl-1, 3-thiazole (0.01mol) and sodium acetate (0.01mol) in dry pyridine (10-15mL), and the mixture was stirred for 7-10h, maintaining the temperature between 0-5°C. The solution was then poured over crushed ice in a beaker, which was previously acidified with few drops of 5 % hydrochloric acid. Stirring the resultant product yielded pale yellow colored precipitate. The crude benzamide derivatives were dried, crystallized using ethyl acetate, and their melting points were determined.
2-[(Methylsulfonyl) amino]-N-(4-methyl-1, 3-thiazol-2-yl) benzamide: m. p.: 213-16°C (yield: 72%) Rf :0.45; λmax: 319nm,IR: 3273.57cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2990.16cm-1 (C-H str.), 1653.27cm-1 (C=O str.),1488.78cm-1 (N-H bend.),1341.96cm-1 (assym. str. S=O), 1329.68cm-1 (C-N str.), 1339.01cm-1 (S=O sym. str.), 1149.45cm-1 (C=S str.),1119.48cm-1 (C-O str.).
Elemental analysis (C, H ,N, & S)
C (54.07)/(54.53), H (3.16)/(3.77) N (7.93)/(7.48), S(17.83)/(17.31) H1 NMR: 2.28(3H,s),2.93(3H,s) ,4(1H,s), ,6.48(1H,s),6.98-7.64(4H,m),9.15(1H,s).
N-(4-Methyl-1, 3-thiazol-2-yl)-2-[(phenylsulfonyl) amino] benzamide: m. p.:237-38°C (yield: 68 %) Rf :0.41;λmax: 310nm; IR: 3273.57cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2990.16cm-1 (C-H str.), 1654.76cm-1 (C=O str.), 1488.78cm-1 (N-H bend.), 1343.08cm-1 (asym. str. S=O), 1329.68cm-1 (C-N str.), 1314.90cm-1 (S=O sym. str.), 1149.45cm-1;(C=S str.), 1319.48cm-1 (C-O str.);
Elemental analysis (C, H ,N & S)
C (54.00)/(54.53), H (391)/(3.77) N (7.62)/(7.48), S(16.63)/(17.13) H1NMR: 2.28(3H,s), 4(1H,s),6.48(1H,s),6.98- 7.86(9H,m),9.15(1H,s).
2-{[(4-Methylphenyl)sulfonyl]amino}-N-(4-methyl-1,3- thiazol-2-yl)benzamide: m. p.:241-42°C (yield:73 %) Rf :0.51;λmax :305nm; IR: 3273.57cm-1 (N-H str.), 3027.45cm- 1 (aromatic C-H str.), 2990.16cm-1 (C-H str.), 1654.73cm- 1 (C=O str.), 1488.78cm-1 (N-H bend.), 1343.08cm-1 (asym, str. S=O),1329.68cm-1(C-Nstr.), 1314.90cm-1 (S=O sym.str.), 1319.48cm-1(C-Ostr.);
Elemental analysis (C, H, N, & S)
C (55.65)/(55.65), H (4.10)/(4.15) N (7.42)/(7.21), S(16.93)/ (16.51) H1 NMR: 2.28(3H,s),2.34(3H,s), 4(1H,s),6.48(1H,s),6.98- 7.74(8H,m),9.15(1H,s).
N-(4-Methyl-1, 3-thiazol-2-yl)-2-[(morpholin-4-ylsulfonyl) amino] benzamide: m. p.: 147-150°C (yield: 72 %) Rf :0.67; λmax: 305nm; IR: 3383.50cm-1 (N-H str.), 3027.45cm- 1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1674.89cm- 1 (C=O str.), 1526.38cm-1 (N-H bend.), 1323.89cm-1 (asym. str. S=O), 1351.86cm-1 (aromatic C-N str.), 1168.72cm-1 (S=O sym. str.), 1122.37cm-1 (C-O).
Elemental analysis (C, ,H, N & S)
C (46.02)/(46.99), H (3.12)/(3.54), N (13.02)/(13.41), S(19.98)/(2 0.47)H1NMR:2.2 8(3H,s),2.8 7-3.67(8H,t), 4(1H,s),6.48(1H,s),6.98-7.64(4H,m),9.15(1H,s).
E. N-(4-Methyl-1, 3-thiazol-2-yl)-2-(sulfamoylamino) benzamide: m. p.:224-25 °C (yield: 70 %) Rf :0.45; λmax: 324nm; IR: 3383.50cm-1 & 3374cm-1(N-H str.), 3027.45cm-1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1669.40cm-1 (C=O str.), 1526.38cm-1 (N-H bend.), 1323.89cm-1 (asym. str. S=O), 1351.86cm-1 (aromatic C-N str.), 1168.72cm-1 (S=O sym. str.), 1122.37cm-1 (C-O).
Elemental analysis (C, H, N, & S)
C (42.88)/(42.16), H (3.39)/(3.54), N (13.44)/(13.41), S(19.99)/(20.47).
H1NMR: 2.28(3H,s),2(2H,s), 4(1H,s),6.48(1H,s),6.98- 7.64(4H,m), 9.15(1H,s).
G. 2-(Benzylamino)-N-(4-methyl-1,3-thiazol-2-yl) benzamide: m. p.: 239-42°C (yield:73 %) Rf :0.40; λmax: 294nm; IR: 3383.50cm-1 (N-H str.), 3027.45cm-1 (aromatic C-H str.), 2939.95cm-1 (aliphatic C-H str.), 1669.94cm-1 (C=O str.),1526.38cm-1(N-H bend.),1351.86cm-1(aromatic C-N str.),1122.35cm-1(C-O);1526.38 cm-1 (N-H bend.), 1351.86 cm-1 (aromatic C-N str.), 1122.35 cm-1 (C-O).
Elemental analysis (C, H, N, & S)
C (66.78)/(66.64), H (4.13)/(4.97) N (8.99)/(8.64), S(9.78)/ (9.88). H1NMR: 2.28(3H,s), 4(1H,s),4.35(2H,s),6.48(1H,s),6.98- 7.67(9H,m), 9.15(1H,s).
Compounds in Scheme 2 were prepared by adopting following procedures.
Ammonolysis of Ester [22]: Ice cold ethanolic solution of the ester, ethyl 2, 5-Dinitrobenzoate was drop-wise added to a mixture of 2-amino-4-methyl-1, 3-thiazole (0.01 mol) and sodium acetate (0.01 mol) in dry pyridine (10-15mL), and the mixture was stirred for 7-10h, maintaining the temperature between 0-5°C. The solution was then poured over crushed ice in a beaker, which was previously acidified with few drops of 5% hydrochloric acid. Stirring the resultant product yielded pale yellow colored precipitate. The crude benzamide derivative was dried, crystallized using chloroform-ethanol, and its melting point was determined.
Reduction to the amino group [29]: To an ice cold solution of ammonia (6 N, 25mL) held in a conical flask equipped with a gas tube, II. vii (0.01 mol) was slowly added while stirring the resultant mixture. The flask was then kept in an ice-bath and the hydrogen sulphide gas was passed in the flask, about 3h. The temperature of the flask was raised to 50°C, after which the reaction mixture was gently boiled for 5h under the hood. This pale yellow solution was filtered and glacial acetic acid was rapidly introduced into the solution. The precipitate so obtained was filtered at pump, washed with ample water and crystallized from rectified spirit.
Benzylation [30]: To a mixture of pure sodium carbonate (3g) and II. g (0.01mol) in a three necked round bottomed flask, freshly distilled benzyl chloride (0.02mol) was added while stirring, and the mixture was heated under reflux was 4 h and allowed to cool. The mixture was filtered on cooling washed with salt water, and distilled under vacuum to remove unreacted benzyl chloride. The residue was washed with water and crystallized from acetone-n-hexane.
1-(2, 5-Dinitrophenyl)-2-(4-methyl-1, 3-thiazol-2- yl) ethanone: m. p.: 134-36°C (yield: 62%), Rf: 0.71; λmax: 395nm,IR:3029cm-1(C-H str. aromatic), 2986cm-1 (C-H str. aliphatic), 1695cm-1 (C=O str.), 1361cm-1 (C-N str.), 1125cm-1 (CO).
1-(2, 5-Diaminophenyl)-2-(4-methyl-1, 3-thiazol-2-yl) ethanone: m. p. 181-83°C (yield 50 %); Rf: 0.52; λmax: 324nm, IR:3427.57cm-1 (N-H str.), 3187.74cm-1(N-H str.) 3030.47cm- 1(C-H str. aromatic) 2985.54cm-1 (C-H str. aliphatic), 1692.23cm- 1 (C=O str.), 1554.34cm-1 (N-H bend.), 1348.96cm-1 (C-N str.), 1123.37cm-1 (C-O).
5- Bis(benzylamino)-N-(4-methyl-1, 3-thiazol-2-yl) benzamide: m. p:254-55°C(yield: 74 %g); Rf: 0.76; λmax: 324nm, IR:3427.57cm-1 (N-H str.), 3187.74cm-1(N-H str.) 3030.47cm- 1(C-H str. aromatic) 2985.54cm-1 (C-H str. aliphatic), 1692.23cm- 1 (C=O str.), 1554.34cm-1 (N-H bend.), 1348.96cm-1 (C-N str.), 1123.37cm-1 (C-O).
Elemental analysis(C,H,N,& S)
C (69.85)/(69.91), H (5.29)/(5.40) N (9.86)/ (9.78), S (7.13)/ (7.47) H1NMR: 2.28(3H,s), 4(2H,s),6.48(1H,s),4.35(4H,s),6.56(1 H, s), 6.63-6.84(2H,d),7.23-7.33(10H,m), 9.15(1H,s).
Conclusion
In summary, the toxic aromatic amino function can be replaced by benzyl, or aryl, group carrying aromatic ring to produce probably non-toxic small molecule GKA. However, pre-clinical studies for assessing mutagenic liabilities of the synthesized compounds were not within the scope of our studies, hence no claim can be made regarding complete safety.
Acknowledgement
The authors are thankful to B. C. U. D. of Savitribai Phule Pune University, Pune, for providing financial assistance to pursue a minor research project resulting in the manuscript. Dr. C. D. Upasani, Principal of S. S. D. J. College of Pharmacy, and Dr. S. J. Surana, Principal, R. C. Patel Institute of Pharmaceutical Education and Research, are also thanked for the research facilities they provided to pursue research.
References
- Pal M (2009) Recent advances in glucokinase activators for the treatment of type 2 diabetes. Drug Discov Today 14(15-16): 784.
- Sarabu R, Berthel SJ, Kester RF, Tilley JW (2008) Novel glucokinase activators: a patent review (2008 - 2010). Exp Opin Ther Patents 21(1): 13-33.
- Daniewski AR, Liu W, Radinov RN, WO Patent (2007) 115968.
- Efanov AM, Barnett DG, Brenner BM, Briggs SL, Delaunois A, et al. (2005) J Endocrinolgy 136: 2696.
- Mckerrecher D, Allen JV, Caulkett PWR, Donald CS, Fen wick ML, et al. (2006) Bioorg Med Chem Lett 16: 2705.
- Bertram LS, Black D, Briner PH, Chatfield R, Cooke A, et al. (2008) Med Chem 51: 4340.
- Nishimura T, Lino T, Mitsuya M, Bamba M, Watanbe H, et al. (2009) Bioorg Med Chem 19: 1357.
- Li F, Zhu Q, Zhang Y, Feng Y, Leng Y, et al. (2010) Bioorg Med Chem 18: 3875.
- Lino T, Sasaki Y, Bamba M, Mitsuya M, Kamata K, et al. (2009) Bioorg Med Chem Lett 19: 5531.
- Kamata K, Mitsuya M, Eiki J, Nagata Y (2004) Structure 12: 429.
- Kugler-Steigmeimer ME, Friederich U, Graf U, Lutz WK, Maier P, et al. (1989) Mutat Res 211: 279.
- Munoz ER, Barnett BM (1998) Mutat Res 413: 15.
- Grimbsy J, Sarabu R, Corbett WL, Haynes NE, Bizzarro FT, et al. (2003) Science 301: 370.
- Dunten P, Swain A, Kammlott U, Crowther R, Lukacs CM, et al. (2004) In Glucokinase and Glycemic Disease- From Basics to Therapeutics Matschinsky FM, Magnuson MA Krager AG (Eds,) Basel, Germany pp 145.
- Efanov AM, Barrett DG, Brenner MB, Briggs SL, Delaunois JD, et al. (2005) J Endocrinology 146: 3696.
- Marotta DE, Anand GR, Anderson TA, Miller SP, Okar DA, et al. (2005) Arch Biochem Biophy 436: 23.
- Sarabu R, Taub R, Grimsby J (2007) Drug Discovery Today: Therapeputic Strategies 4: 101.
- Kumari V, Li C (2008) Current Chemical Genomics 2: 76.
- Johnson TO, Humphries PS (2006) Annual Reports in Medicinal Chemistry 41: 141.
- Nishimura T, Lino T, Mitsuya M, Bamba M, Watanabe H, et al. (2009) Bioorg J Med Chem Lett 19: 1357.
- Lino T, Tsukahara D, Kamat K, Sasaki K, Ohyama S, et al. (2009) Bioorg M Chem 17: 2733.
- Khadse SC, Chatpalliwar VA (2012) Synthesis of Benzamides by Microwave Assisted Ring Opening of Less Reactive Dimethyl amino benzylidene Oxazolone. Arabian Journal of Chemistry.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India, (PVT.) Ltd pp. 413.
- Paquette LA (2009) Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons, USA, 1- 6.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India, pp. 883.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India, pp. 1275.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India, pp. 780.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India pp. 1073.
- Blatt AH, Gilman H (1932) Organic Syntheses. John Wiley and Sons, New York 1(2): 52-53.
- Furniss BS, Hannaford A, Smith PWG, Tatchell AR (1989) Vogel's Textbook of Practical Organic Chemistry, V (ed.), Saurabh Printers India, pp. 902.






























