Synthesis and Characterization of New Triazinane-2-Thione and Oxadiazinane 4-Thiones from Benzimidazole
Kumaraswamy G1*, Ravichander M1, Uma K3, Jagadeesh BK2 and Brahmeshwari G2
1Department of Chemistry, Mahatma Gandhi Institute of Technology, India
2Department of Chemistry, Kakatiya University, India
3Excelra Knowledge Solutions Private Limited, India
Submission: April 02, 2017; Published: Jue 28, 2017
*Corresponding author: Kumaraswamy G, Department of Chemistry, Mahatma Gandhi Institute of Technology, India, Email: kumargullapelli001@gmail.com
How to cite this article: Kumaraswamy G, Ravichander M, Uma K, Jagadeesh BK, Brahmeshwari G. Synthesis and Characterization of New Triazinane-2-Thione and Oxadiazinane 4-Thiones from Benzimidazole. Nov Appro Drug Des Dev. 2017; 2(2): 555581. DOI: 10.19080/NAPDD.2017.02.555581
Abstract
The present study a new strategy for the synthesis of most potent nitrogen heterocycles i.e., triazinane-2-thiones and oxadiazinanes-4- thiones (3-4) were efficiently synthesized by cyclic condensation based on classical Mannich amino methylation of N, N' unsymmetrical Thio ureas. The synthesized compounds have been characterized by analytical and spectral (IR, 1HNMR and Mass) data.
Keywords: Synthesis; Characterization; Triazinane-2-thione; Benzimidazole
Introduction
Demand for higher efficiency, economy and selectivity in the synthesis of novel molecular scaffolds drives organic chemistry, particularly nitrogen-containing molecules has been of longstanding interest to organic chemists due to their great importance in chemistry and biology [1]. Many of these heterocyclic compounds exhibit various biological activities [2,5]. Little work has been published in the area of 1,3,5 triazinane- 2-phones showing antimicrobial activity [6], Biocidal [7] and enantio-differentiating coupling reagents [8]. The potent Heterocycles having, structural unit has a significant place among pharmaceutically important synthetic and natural materials [9].
Materials and Methods
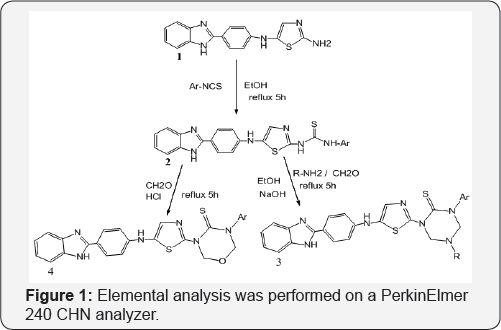
Melting points of the synthesized compounds are determined in open capillary tubes and are uncorrected. Reaction Progress was observed by TLC plates, Bruker 300MHz instrument was used to record 1HNMR spectra in DMSO/CDCl3 using TMS as internal standard. Chemical shifts (5) are expressed in ppm. Perkin Elmer BX series FT-IR was to record IR spectra, Elemental analysis were performed on a PerkinElmer 240CHN analyzer Figure 1.
Experimental section
Synthesis of 1-(5-(4-(1H-benzo[d]imidazol-2-yl)phenyl) amino)thiazol-2-yl)-3-phenyl thiourea (2): A mixture of compound [10] (1) (0.01mmol) and sodium hydride (0.5g, 20mmol) in Ethanol (80ml) was heated under reflux for 30min and cooled. Phenyl isothiocynate (0.01mmol) was added and refluxing continued for a further 4h. The solvent was evaporated off and the residue dissolved in DCM (50ml) and washed with dilute HCl. The organic phase was dried (MgSO4) and the solvent evaporated off to give the desired compound (2). The progress of the reaction was monitored by TLC and recrystallized from ethanol.
Synthesis of 1-(5-(4-(1H-benzo[d]imidazol-2-yl) Phenylamino)thiazol-2-yl)-5-methyl-3-phenyl-1,3,5 triazinane-2-thione (3): A mixture of compound (2) in (1mmol), 30% formaldehyde (2mmol) and methyl amine (1mmol) and (0.01mole) NaOH were taken in ethanol (30ml) and refluxed for about 5-6h. The progress of the reaction was monitored by TLC. After completion of the reaction it was cooled and the product was filtered. The crude product was passed through silica gel by column and the product was eluted from 60% ethyl acetate and hexane.
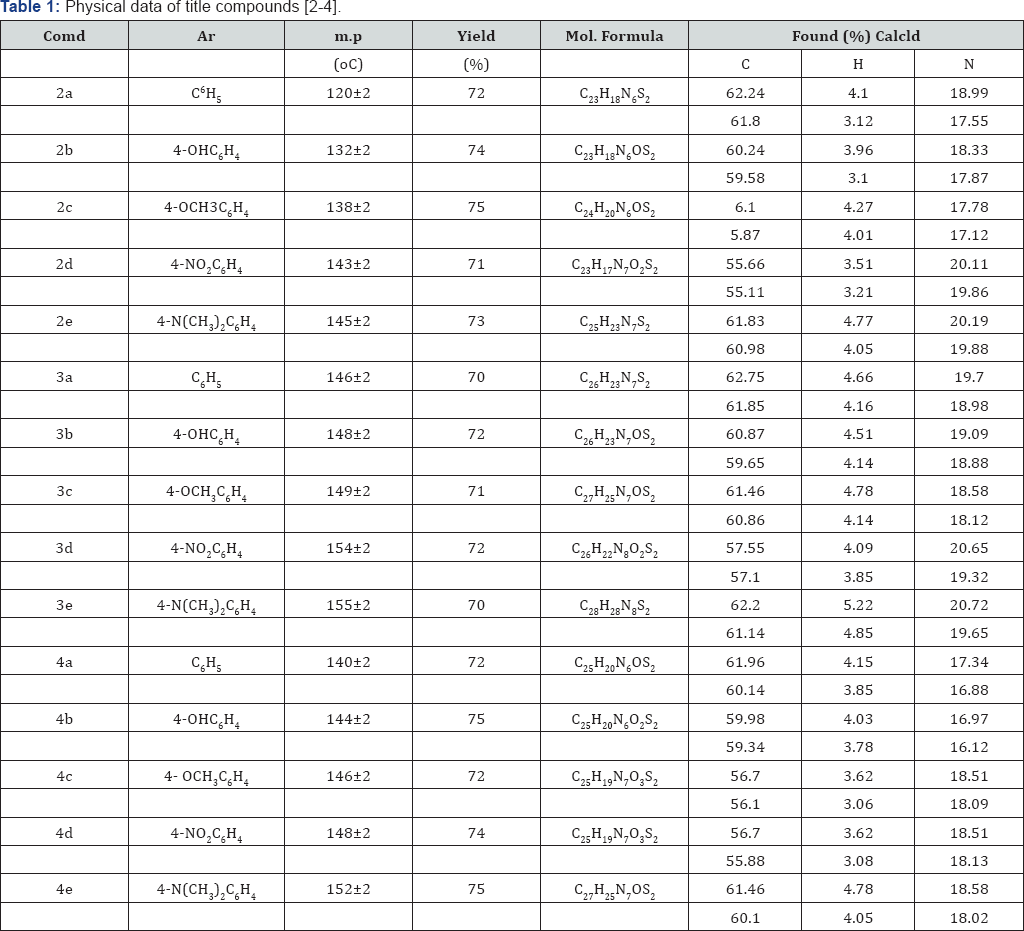
Synthesis of 3-(5-((4-(1H-benzo[d]imidazol-2-yl) phenyl)amino)thiazol-2-yl)-5-phenyl-1,3,5-oxadiazmane- 4-thione(4): A mixture of compound (2) in (1mmol), 30% formaldehyde (2mmol) were taken in ethanol (30ml) and add 1ml concentrated HCl (5ml) and refluxed for about 5-6hr at 100110 °C. The progress of the reaction was monitored by TLC. After completion of the reaction it was cooled and neutralized with 10% NaOH. The product was filtered. The crude product was passed through silica gel by column and the product was eluted from 60% ethyl acetate and hexane. These compounds were purified by recrystallisation from suitable solvents. Structures of the synthesized compounds [2-4(a-e] have been confirmed on the basis of analytical and spectral (IR, 1HNMR and Mass) data (Table 1).
Results and Discussion
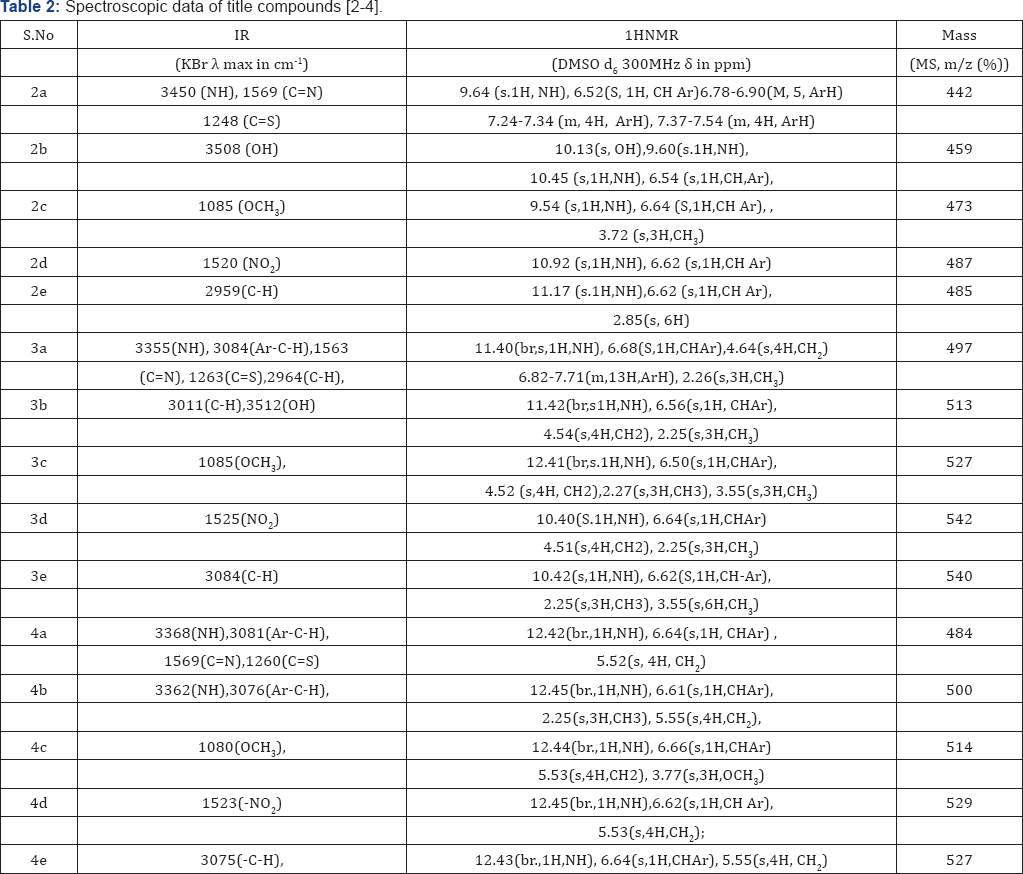
In the present study, it has been discussed that the synthesis of various benzimidazole fused triazinane 2-thione heterocyclic moieties by cyclic condensation based on classical Mannich amino methylation of N,N' unsymmetrical this ureas (2) with 30% HCHO and methyl amine in ethanol. An aliphatic amine has been yielded in the condensation between methyl amine and formaldehyde, it cyclizes immediately to produce corresponding 1,3,5-triazinane since imine is unstable. With respect to biological activity benzimidazole fused heterocyclic compounds such as triazinane derivatives are of very important than the cyclic compounds [11,12] (Table 2).
Conclusion
The research study reported efficiently synthesized new analogs of triazinane and oxadiazinane by classical amino methylation with different aerial-N,N' unsymmetrical thioureas by taking benzimidazole as starting material. All the compounds were characterized by standard spectroscopic techniques.
Acknowledgement
The authors are grateful acknowledge to the Department of chemistry Kakatiya university, Warangal for their constant support during this research work.
References
- Nagib DA, Scott ME, MacMillan DWC (2009) Enantioselective a-Trifluoromethylation of Aldehydes via Photoredox Organocatalysis. J Am Chem Soc 131: 10875-10877.
- Shushank D, Pramod KS, Niranjan K (2013) Synthesis of novel benzimidazole derivatives: as potent analgesic agent. Med Chem Res 22: 900-904.
- Emre M, Hakan B, Bahar BS, Mustafa E, Demet 5, et al. (2017) Synthesis and molecular docking study of some 5,6-dichloro-2-cyclopropyl- 1H-benzimidazole derivatives bearing triazole, oxadiazole, and imine functionalities as potent inhibitors of urease. Bioorg Med Chem Lett 27(13): 3014-3018.?
- Kumaraswamy G, Murali krishna T, Brahmeshwari G (2014) Synthesis and antimicrobial activity of 2-(4-amino phenyl) Benzimidazole based pyrimidine derivatives. Int J Pharm Bio Sci 5(1): 682-690.
- Anjani S, Kishor K, Ana C, Marina S, Irini D, et al. (2010) Synthesis of some new S-triazine based chalcones and their derivatives as potent antimicrobial agents. Eur J Med Chem 45(2): 510-518.
- Kamelia M, Mahdy El, Reda M, Abdel R (2011) A Convenient Methods for Synthetic Isomeric Structures of Pyrimido1,2,4-triazine Derivatives as Biocidal Agents. Acta Chim Slov 58: 755-764.
- Sugimoto H, Yamane Y, Inoue S (2000) Enantiomeric discrimination by novel optically active isocyanurates having peripheral amino acid units. Tetrahedron 11: 2067-2075.
- Han T, Cho JH, Oh CH (2006) Synthesis and biological evaluation of 10-methylcarbapenems having cyclic thiourea moieties and their related compounds. Eur J Med Chem 41(7): 825-832.
- Maruthamuthu, Shameela R, Christina R, Stella P, Bharathi Dileepan AG, et al. (2016) The chemistry and biological significance of imidazole, benzimidazole, benzoxazole, tetrazole and quinazolinone nucleus; Journal of Chemical and Pharmaceutical Research 8(5): 505-526.
- Brahmeshwari G, Kumaraswamy G (2014) Synthesis and antifungal activity of 3-(4-(4-(1H-Benzo[d] imidazole2-yl) Phenyl amino) thiazol 2-yl)-2-Phenyl thiazolidin-4-one derivatives. Int J Chem Sci 12(3): 1-9.
- Khairullina RR, Geniyatova AR, Ibragimov AG, Dzhemilev UM (2013) Synthesis of 5-alkyl-1,3,5-triazinan-2-ones and 5-alkyl-1,3,5- triazinane-2-thiones using Cu- and Sm-containing catalysts Russian Journal of Organic Chemistry 49(6): 904-908.
- Isis M Figueiredo, Luciane V, dos Santos, Willian F da Costa, Joao E de Carvalho, et al. (2006) Synthesis and antiproliferative activity of novel limonene derivatives with a substituted thiourea moiety. J Braz Chem Soc 17(5): 954-960.






























