Nanoparticles in Protein Formulation: Challenges, Current Trends and Future Perspectives
Ashutosh Naik and Dinesh V Palanivelu*
Biocon Research Centre, India
Submission: March 28, 2017; Published: Jue 28, 2017
*Corresponding author: Dinesh V Palanivelu, Biocon Research Centre, India, Email: Dinesh.Palanivelu@biocon.com
How to cite this article: Ashutosh N, Dinesh V P. Nanoparticles in Protein Formulation: Challenges, Current Trends and Future Perspectives. Nov Appro Drug Des Dev. 2017; 2(1): 555580. DOI: 10.19080/NAPDD.2017.02.555580
Abstract
Nanotechnology involves imaging, measuring, modelling and manipulating matter at dimensions ranging from 1 to 100 nanometers. They are used as a carrier for target delivery system for altering the pharmaco dynamic and pharmacokinetic properties. In Biotherapeutics applications, the proteins bio molecules whose size are at the nano scale can be used effectively in the nanoparticle system which acts as a protein delivery vehicle to increase the efficacy of proteins, reduce dose levels and improve bioavailability when compared with conventional alternatives. This paper attempts to review the different nanoparticles and state of the art technology that could be used for protein delivery while also addressing the challenges and opportunities in using these alternatives.
Keywords: Nanotechnology; Nano-emulsions; Liposomes; Polymerosomes; Hydrogel; Single protein nano capsules; Protein cages; Tunable pistons
Introduction
There’s plenty of room at the bottom was the title of the lecture given by Richard Feynman to introduce the concept of Nanotechnology to the world [1]. Over the last five decades, significant progress has been made in the understanding of materials at the nano scale level. There have been many developments and improvements in formulation of drugs, however; the delivery of drugs in the required quantity at the desired target site still remains a challenge [2]. Nanoparticles are designed using methodologies that permit complete control over physicochemical attributes at the molecular scale while overcoming pharmacokinetic limitations associated with using conventional methods [2,3].
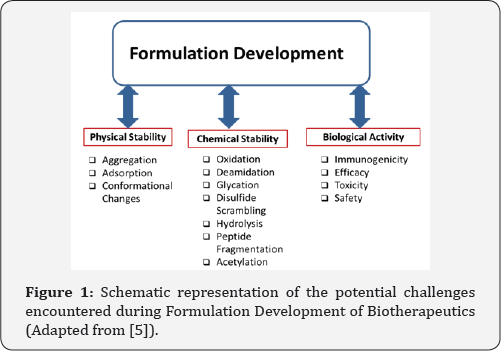
Over the last decade, number of proteins and other bio molecules that are targeted towards various cellular processes have emerged, necessitating the focus on intelligent "drug delivery carriers" that respond to various physiological stimuli [3] . These intelligent carriers at the micro and nanoscale greatly ameliorate the therapeutic efficacy of drugs because of the ability to differentiate between healthy and diseased cells thereby improving the quality of life of the patient [3]. This new class of “intelligent therapeutics” refers to intelligent and responsive delivery systems that are designed to perform various functions like detection, isolation and/or release of therapeutic agents for the treatment of diseased conditions. In order for these carriers to be effective, the materials of choice for nanoparticle formation either synthetic or hybrid must be designed to elicit a positive response within the biological systems at that nano scale. Materials that are responsive to physiological stimuli are also being considered for developing carriers at the nanoscale [4] . There are many challenges associated with formulation development and Figure 1 shows a schematic view of some of the barriers associated with physical, chemical and biological stability as well as activity. Nanoparticles and their use in formulation development as carriers for proteins and drugs are expected to overcome some of the challenges.
Nanoparticles employed in protein drug delivery system
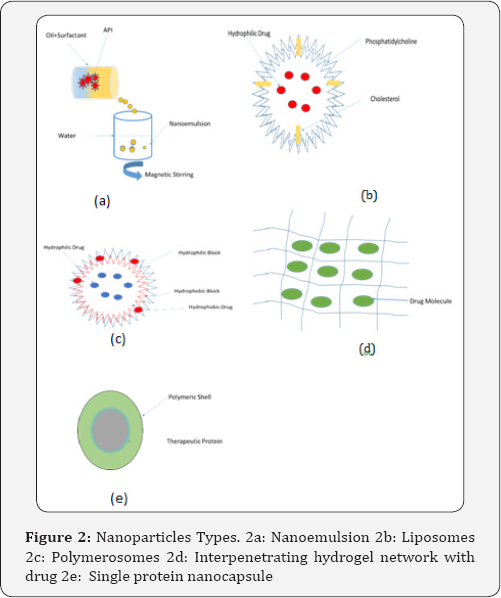
There are a number of different nanoparticle that can be used in the delivery of proteins and other bio molecules. Figure 2a shows the different nanoparticle that has been well characterized. These include but are not limited to nanoemulsions [5-9], liposomes [10-13], polymerosomes [14-17], hydrogel nanoparticle [18-21] and single protein nanocapsules [22,23]. The methods of preparation, applications, advantages and disadvantages of these nanoparticles as carriers will be discussed in the following sections.
Nano-emulsions
Nanoemulsions (Figure 2b) are a class of emulsions whose droplet size is between 20-500nm but typically around ~100nm. It is usually comprised of oil, water and an emulsifier. The addition of an emulsifier is crucial in reducing the size of the droplets, as it directly affects the interfacial tension [24]. The emulsifier used is generally a surfactant, but certain proteins and lipid molecules have also been effectively used in the preparation of nano emulsions [25-28].
Method of preparation: The method of preparation of nano emulsions can be classified into two broad categories; High energy and low energy methods [29-31]. High energy methods include high pressure homogenization and ultrasonication [31]. Phase Inversion temperature [32] and emulsion inversion point [33-35] are examples of low energy methods.
Applications: Nanoemulsions have applications in the food, cosmetic, pharmaceutical industries and in drug delivery. Nanoemulsions have been used in most forms of drug delivery, including topical, ocular, intravenous, internasal and oral delivery. These applications take advantage of the lipophilic nature of nano emulsions to improve the solubility of water- insoluble drugs; in addition the charge and rheology of nano emulsions to formulate aqueous solutions that allows simplified delivery of drugs [24].
Advantages and disadvantages: Advantages of nano emulsions include increased drug loading, enhanced solubility, improved bioavailability and controlled release of drug [25]. Despite these obvious advantages there are challenges associated with nano emulsions which are cost, process associated with nano emulsions preparation, and the nature of the associated interfacial chemistry is not very well understood yet [36].
Liposomes
Liposomes (Figure 2c) are spherical bilayer lipid structures whose main constituents are phospholipids [37-41]. The phospholipids are surrounded by cholesterol and other hydrophilic polymer conjugated lipids around the vesicle [37,39]. Cholesterol is an important component of the liposome because once incorporated the hydroxyl group within cholesterol is towards the aqueous phase and its tetracyclic ring is towards the membrane core [37,42]. This arrangement results in decreasing the permeability of the membrane to water-soluble molecules and improving the stability of liposomes [37,42].
Types of liposomes and their methods of preparation: Liposomes can be classified based on either their method of preparation, number of bilayer or size. However the most common classification is as multilamellar vesicles and unilamellar vesicles [37]. The method of preparation for multilamellar and unilamellar vesicles is given below.
Multilamellar vesicles (MLV): MLVs are lipid vesicles that are composed of more than two bilayer and the size of these vesicles is between 0.05 and 10|im [37]. The simplest and the most popular method for the preparation of MLVs is thin film hydration [37,43-46]. In this method, MLVs can be formed spontaneously by adding an excess volume of aqueous buffer to a thin film of dry lipids at a temperature above the phase transition temperature (PTT) of lipids [37].
Unilamellar vesicles (ULV)
ULVs can be divided into two types; large unilamellar vesicles (LUV) and small unilamellar vesicles (SUV). LUVs are considered to be vesicles with a size >100nm [37]. The two most common methods to produce LUVs are the reverse phase evaporation technique [37,47] and the detergent removal technique [37,48]. In the reverse phase evaporation method, water in-oil emulsion composed of water and phospholipids are produced in the presence of excessive organic solvent mechanical means or by sonication. On removal of the organic solvent under vacuum, phospholipid droplets containing water are formed. These droplets coalesce to form a gel-like matrix, which is converted to a smooth paste of LUV on complete removal of organic solvent. The reported drug encapsulation efficiency for this method is 60-65%. In the detergent removal technique, micelles are formed using a combination of phospholipids and a detergent. The detergent is then removed step-wise which results in micelles composed mainly of phospholipids which coalesce to form single bilayer vesicles [37,48,49]. SUVs are in the size range of 25-50nm. These are produced from LUVs or multilamellar vesicles (MLV) by sonication or extrusion under high pressure [37]. Sonication can be carried out using either a probe or bath sonicator. A bath sonicator is preferred as it is easier to maintain aseptic conditions [37]. SUVs produced using sonication are usually in the size range of 25-50nm. The other method is extrusion under high pressure (20,000psi) at 4 °C. The vesicles obtained are in the size range of 15-30nm and extrusion offers better control on size compared to sonication [37].
Applications: Liposomes are primarily used in medicine and pharmacology [50]. These applications can be divided into therapeutic and diagnostic, involving the delivery of various biomarkers or drugs (therapeutic) or as a tool, model or reagent in further understanding cell-based interactions and processes [50]. The most common application of liposomes is in the delivery of anticancer drugs [37,50].
Advantages and disadvantages: Liposomes are non-toxic, biocompatible and completely biodegradable [50,51]. In addition they improve the efficacy, stability and therapeutic index of the encapsulated drug. Despite the obvious advantages there are some drawbacks associated with liposomes these include high cost, low solubility and a shorter half-life [51,52].
Polymersomes
Polymersomes Figure 2c are synthetic vesicles made of amphiphilic block copolymers [52]. In these vesicles an aqueous core is surrounded by a hydrophobic membrane [52,53]. The size of polymer somes can vary from 10nm-5|im. The thickness of the membrane can be controlled by changing the hydrophobic ratio of copolymers [52,53].
Method of preparation: The two most common methods of preparation for polymer somes are the solvent switching technique [54] and the bilayer phospholipid theory [55]. In the solvent switching technique, the organic polymer is dissolved in an organic solvent followed by the drop-wise addition of aqueous phase. The organic solvent is then removed resulting in polymer some suspensions [54]. The phospholipid bilayer theory involves the hydration of block copolymers to induce self-assembly of thin films, produced by evaporation of organic solvents which can be rehydrated using water.
Applications: Similar to liposomes, polymer somes are primarily used for therapeutic and diagnostic purposes [56]. These involve the delivery of anticancer drugs [57,58] and gene therapy from a therapeutic standpoint and intracellular tracking [56] that aids in medical diagnosis.
Advantages and disadvantages: The polymersomes are suitable for delivery of hydrophobic, hydrophilic, amphipathic drugs and multiple drug loading. They have an improved half-life, enhanced stability for encapsulated drugs and exhibit stimuli responsive release [52]. The disadvantages associated with polymer somes are high production cost and low encapsulation efficiency [59].
Hydrogel nanoparticles
Hydrogels are three dimensional polymeric networks capable of absorbing large amounts of water [60,61]. These are formed using natural polymers such as chitosan or pectin, synthetic polymers like PEG-PLGA or a combination of the two PEG-Alginate [60]. Hydrogels can be classified based on nature of side groups (neutral or ionic), mechanical and structural features method of preparation (homo- or co-polymer), physical structure and responsiveness to physiologic environment stimuli Figure 2d [61].
Methods of preparation of hydrogel and network formation: Gelation that leads to network formation can either be based on physical or chemical linkages [62]. Physical gels can further be divided into strong and weak gels. The strong gels in certain conditions may be considered as chemical gels because of the strength of the bond. Examples of strong physical gels include glassy nodules and triple helices. Weak physical gels have weak hydrogen bonds between them and are reversible Chemical gelation is a method that results in strong bonds produced using condensation, vulcanisation and polymerization [62].
Applications: Hydrogels have a variety of therapeutic applications [63]. Hydrogels have also been used in the delivery of DNA and vaccines [63].
Advantages and disadvantages: The advantages of hydrogels include safe delivery of drugs, improved transport of nutrient to cells and products from cells. It also has the advantage of being injected as a liquid that gels at body temperature. The disadvantages include that they are mechanically weak and drug-loading is difficult [63].
Single protein nano capsules
These carriers consist of a protein core with a polymeric shell attached to the core. This is a relatively new nanoparticle delivery system [64]. The choice of monomer whether neutral or cationic allows for control over surface charge of the protein. The protein core that forms the basis of the nanocapsule can be chosen from a wide variety of proteins including but not limited to enhanced green fluorescent protein (EGFP), horseradish peroxidase (HRP), bovine serum albumin (BSA), superoxide dismutase (SOD) and caspase-3 [64]. As can be seen from Figure 2e the polymeric shell surrounding the protein may be biodegradable or non-biodegradable [64].
Method of preparation: Polymerisable vinyl groups are attached to the protein through covalent linkages, further polymerization takes place when the mixture is placed in an aqueous solution containing monomers [65] and cross linkers [66]. This results in each protein core being wrapped in a polymeric shell which may be either biodegradable or non- biodegradable [64].
Advantages and disadvantages: The single protein nanocapsule is suitable for cell internalization and provides enhanced stability for protein molecules [64]. However, it might result in the reduced protein activity.
Future trends
Apart from the nanoparticles discussed earlier there have been some new developments with regard to protein based drug delivery. Protein cages have emerged as one such example; they are hollow protein nanoparticles, such as viral capsids, virus-like particles, ferritin, heat- shock proteins and chaperonins. These nanoparticles have a defined structure and uniform size. Their protein subunits can be modified using genetic engineering [67]. Another such example is the use of si-RNA which involves the use of a hollow nano capsule, the group II chaperonin thermo some (THS) is a hollow protein nanoparticle that can encapsulate macromolecular structures.
Two large pores grant access to the protein cage. THS- Polyamidoamine (PAMAM) protects siRNA from degradation by RNase A and traffics siRNA into U87 cancer cells [68]. The use of pH as stimuli to puncture cell membranes to enable entry into the cell has also been reported [69]. The protein 'piston' consists of modified R bodies found in a bacterial endosymbiont of paramecium. These pistons go through multiple cycles of rapid extension and retraction [69].
Conclusion
The field of nanotechnology, in particular the use of nanoparticles for formulation and delivery of proteins and other bimolecular is indeed an exciting arena for biopharmaceutical companies. The recent developments in the field of drug delivery, has given the scope that one of these nanoparticles and their use as a delivery system could soon be viable option for commercialization. Two recent examples of which area PEGylated liposome for patients suffering from Kaposi's Sarcoma and a nanoemulsion of cyclosporine A used to treat dry eye syndrome have been given clinical approval in 2013 and 2015 respectively [70].
References
- Feynman RP (1960) There's plenty of room at the bottom. Engineering and science 23(5): 22-36.
- Blanco E, Shen H, Ferrari M (2015) Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat Biotechnol 33(9): 941-951.
- Safari J, Zarnegar Z (2014) Advanced drug delivery systems: Nanotechnology of health design A review. Journal of Saudi Chemical Society 18(2): 85-99.
- Caldorera-Moore M, Peppas NA (2009) Micro and nanotechnologies for intelligent and responsive biomaterial-based medical systems. Adv Drug Deliv Rev 61(15): 1391-1401.
- Chaurasia G (2016) A Review On Pharmaceutical Preformulation Studies In Formulation and Development of New Drug Molecules. International Journal of Pharmaceutical Sciences and Research 7(6): 2313.
- Das D, Sahu P, Kashaw V, Kashaw KS (2017) Formulation and Assessment of In-Vivo Anti-Inflammatory Potential of Omega-3-Fatty Acid Loaded Self Emulsifying Nanoemulsion. Current Nanomedicine (Formerly: Recent Patents on Nanomedicine) 7(3): 47-58.
- Artigas MA, Fani AA, Belloso OM (2017) Improving the shelf life of low- fat cut cheese using nanoemulsion-based edible coatings containing oregano essential oil and mandarin fiber. Food Control 76: 1-12.
- Chan MY, Fedor DM, Phan T, Kramer RM (2017) Interactions Between Antigens and Nanoemulsion Adjuvants: Separation and Characterization Techniques. Methods Mol Biol 1494: 285-294.
- Tanaka H, Oasa S, Kinjo M, Tange K, Nakai Y, et al. (2017) Temperature and pH sensitivity of a stabilized self-nanoemulsion formed using an ionizable lipid-like material via an oil-to-surfactant transition. Colloids Surf B Biointerfaces 151: 95-101.
- Milorey B, Schweitzer-Stenner R (2017) Oxidation State and PH Dependence of Cytochrome C Binding to Cardiolipin-Containing Liposomes. Biophysical Journal 112(3 Suppl 1): 387a.
- Bertrand N, Simard P, Leroux JC (2017) Serum-Stable, Long-Circulating, pH-Sensitive PEGylated Liposomes. Methods Mol Bio 1522: 193-207.
- Sala M, Miladi K, Agusti G, Elaissari A, Fessi H (2017) Preparation of liposomes: A comparative study between the double solvent displacement and the conventional ethanol injection-From laboratory scale to large scale. Colloids and Surfaces A: Physicochemical and Engineering Aspects.
- Benson HA (2017) Elastic Liposomes for Topical and Transdermal Drug Delivery. Liposomes: Methods Mol Biol 1522: 107-117.
- Hu X, Zhang Y, Xie Z, Jing X, Bellotti A, et al. (2017) Stimuli-Responsive Polymersomes for Biomedical Applications. Biomacromolecules 18(3): 649-673.
- Discher DE, Ahmed F (2006) Polymersomes. Annu Rev Biomed Eng 8: 323-341.
- Discher BM, Won YY, Ege DS, Lee JC (1999) Polymersomes: tough vesicles made from diblock copolymers. Science 284(5417): 11431146.
- Meng F, Zhong Z, Feijen J (2009) Stimuli-responsive polymersomes for programmed drug delivery. Biomacromolecules, 10(2): 197-209.
- Hamidi M, Azadi A, Rafiei P (2008) Hydrogel nanoparticles in drug delivery. Adv Drug Deliv Rev 60(15): 1638-1649.
- Na K, Lee KH, Bae YH (2004) pH-sensitivity and pH-dependent interior structural change of self-assembled hydrogel nanoparticles of pullulan acetate/oligo-sulfonamide conjugate. J Control Release 97(3): 513525.
- Gan D, Lyon LA (2001) Tunable swelling kinetics in core-shell hydrogel nanoparticles. J Am Chem Soc 123(31): 7511-7517.
- Missirlis D, Kawamura R, Tirelli N, Hubbell JA (2006) Doxorubicin encapsulation and diffusional release from stable, polymeric, hydrogel nanoparticles. Eur J Pharm Sci 29(2): 120-129.
- Gu Z, Biswas A, Joo KI, Hu B, Wang P, et al. (2010) Probing protease activity by single-fluorescent-protein nanocapsules. Chem Commun (Camb) 46(35): 6467-6469.
- Zhao M, Biswas A, Hu B, Joo KI, Wang P, et al. (2011) Redox-responsive nanocapsules for intracellular protein delivery. Biomaterials 32(22): 5223-5230.
- Gupta A, Eral HB, Hatton TA, Doyle PS (2016) Nanoemulsions: formation, properties and applications. Soft matter 12(11): 28262841.
- McClements DJ (2011) Edible nanoemulsions: fabrication, properties, and functional performance. Soft Matter 7(6): 2297-2316.
- McClements DJ, Rao J (2011) Food-grade nanoemulsions: formulation, fabrication, properties, performance, biological fate, and potential toxicity. Crit Rev Food Sci Nutr 51(4): 285-330.
- Troncoso E, Aguilera JM, McClements DJ (2012) Fabrication, characterization and lipase digestibility of food-grade nanoemulsions. Food Hydrocolloids 27(2): 355-363.
- Qian C, McClements DJ (2011) Formation of nanoemulsions stabilized by model food-grade emulsifiers using high-pressure homogenization: factors affecting particle size. Food Hydrocolloids 25(5): 1000-1008.
- Tadros T, Izquierdo P, Esquena J, Solans C (2004) Formation and stability of nano-emulsions. Adv Colloid Interface Sci 108: 303-318.
- Solans C, Izquierdo P, Nolla J, Azemar N, Garcia-Celma MJ (2005) Nano-emulsions. Current opinion in colloid & interface science 10(3): 102110.
- Fryd MM, Mason TG (2012) Advanced nanoemulsions. Annu Rev Phys Chem 63: 493-518.
- Izquierdo P, Esquena J, Tadros TF, Dederen C, Garcia MJ, et al. (2002) Formation and stability of nano-emulsions prepared using the phase inversion temperature method. Langmuir 18(1): 26-30.
- Forgiarini A, Esquena J, Gonz lez C, Solans C (2000) Studies of the relation between phase behavior and emulsification methods with nanoemulsion formation. In Trends in colloid and interface science XIV, pp. 36-39.
- Forgiarini A, Esquena J, González C, Solans C (2001) Formation and stability of nano-emulsions in mixed nonionic surfactant systems. In Trends in colloid and interface science XV, pp. 184-189.
- Forgiarini A, Esquena J, Gonzalez C, Solans C (2001) Formation of nano-emulsions by low-energy emulsification methods at constant temperature. Langmuir 17(7): 2076-2083.
- Thakur A, Walia MK, Kumar S (2013) Nanoemulsion in enhancement of bioavailability of poorly soluble drugs: a review. Pharmacophore 4(1): 15-25.
- Yingchoncharoen P, Kalinowski DS, Richardson DR (2016) Lipid-based drug delivery systems in cancer therapy: what is available and what is yet to come. Pharmacol Rev 68(3): 701-787.
- Gregoriadis G (1976) The carrier potential of liposomes in biology and medicine. N Engl J Med 295(14): 765-770.
- Sharma A, Sharma US (1997) Liposomes in drug delivery: progress and limitations. International Journal of Pharmaceutics 154(2): 123-140.
- Torchilin VP (2005) Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov 4(2): 145-160.
- Wacker M (2013) Nanocarriers for intravenous injection-the long hard road to the market. Int J Pharm 457(1): 50-62.
- Vemuri S, Rhodes CT (1995) Preparation and characterization of liposomes as therapeutic delivery systems: a review. Pharm Acta Helv 70(2): 95-111.
- Bangham AD (1982) Preparation of liposomes and methods for measuring their permeabilities, in Technique in Life Science - Technique in Lipid and Membrane Biochemistry Part II. In: Hesketh TR, Kornberg HL, et al. (Eds.), Elsevier/North-Holland, Amsterdam, Netherlands, pp. 1-25.
- Olson F, Hunt CA, Szoka FC, Vail WJ, Papahadjopoulos D (1979) Preparation of liposomes of defined size distribution by extrusion through polycarbonate membranes. Biochim Biophys Acta 557(1): 9-23.
- Papahadjopoulos D, Watkins JC (1967) Phospholipid model membranes. II. Permeability properties of hydrated liquid crystals. Biochim Biophys Acta 135(4): 639-652.
- Gruner SM, Lenk RP, Janoff AS, Ostro MJ (1985) Novel multilayered lipid vesicles: comparison of physical characteristics of multilamellar liposomes and stable plurilamellar vesicles. Biochemistry 24(12): 2833-2842.
- Szoka F, Papahadjopoulos D (1978) Procedure for preparation of liposomes with large internal aqueous space and high capture by reverse-phase evaporation. Proc Natl Acad Sci USA 75(9): 4194-4198.
- Kagawa Y, Racker E (1971) Partial Resolution of the Enzymes Catalyzing Oxidative Phosphorylation Xxv. Reconstitution of Vesicles Catalyzing 32pi-Adenosine Triphosphate Exchange. Journal of Biological Chemistry 246(17): 5477-5487.
- Milsmann MH, Schwendener RA, Weder HG (1978) The preparation of large single bilayer liposomes by a fast and controlled dialysis. Biochim Biophys Acta 512(1): 147-155.
- Akbarzadeh A, Rezaei SR, Davaran S, Joo SW, Zarghami N, et al. (2013). Liposome: classification, preparation and applications. Nanoscale Res Lett 8(1): 102.
- Himanshu A, Sitasharan P, Singhai AK (2011) Liposomes as drug carriers. IJPLS 2(7): 945-951.
- Thambi T, Park JH, Lee DS (2016) Stimuli-responsive polymersomes for cancer therapy. Biomaterials science 4(1): 55-69.
- Yin H, Kang SW, Bae YH (2009) Polymersome formation from AB2 type 3 miktoarm star copolymers. Macromolecules 42(19): 7456-7464.
- Marsden HR, Gabrielli L, Kros A (2010) Rapid preparation of polymersomes by a water addition/solvent evaporation method. Polymer Chemistry 1(9): 1512-1518.
- Lombardo D, Kiselev MA, Magazu S, Calandra P (2015) Amphiphiles self-assembly: basic concepts and future perspectives of supramolecular approaches. Advances in Condensed Matter Physics 2015(2015): 22.
- Guan L, Rizzello L, Battaglia G (2015) Polymersomes and their applications in cancer delivery and therapy. Nanomedicine (Lond) 10(17): 2757-2780.?
- Gustafson DL, Merz AL, Long ME (2005) Pharmacokinetics of combined doxorubicin and paclitaxel in mice. Cancer Lett 220(2): 161-169.
- Briasoulis E, Karavasilis V, Tzamakou E, Rammou D, Soulti K, et al. (2004) Interaction pharmacokinetics of pegylated liposomal doxorubicin (Caelyx) on coadministration with paclitaxel or docetaxel. Cancer Chemother Pharmacol 53(5): 452-457.
- Egli S (2011) Polymersomes for biomedical applications: surface functionalization of silicone-based polymer vesicles Doctoral dissertation. University of Basel, Switzerland.
- Hamidi M, Azadi A, Rafiei P (2008) Hydrogel nanoparticles in drug delivery. Adv Drug Deliv Rev 60(15): 1638-1649.
- Peppas NA, Mikos AG (1986) Preparation methods and structure of hydrogels. Hydrogels in medicine and pharmacy 1: 1-27.
- Gulrez SK, Phillips GO, Al-Assaf S (2011) Hydrogels: methods of preparation, characterisation and applications.
- Yahia L, Chirani N, Gritsch L, Motta FL (2015) History and Applications of Hydrogels. Biomedical Sciences 4: 2.
- Yan M, Du J, Gu Z, Liang M, Hu Y, et al. (2010) A novel intracellular protein delivery platform based on single-protein nano capsules. Nature nanotechnology 5: 48-53.
- Birch JR, Onakunle Y, Smales CM, James DC (2005) Therapeutic proteins: methods and protocols.
- Sato H, Sugiyama Y, Tsuji A, Horikoshi I (1996) Importance of receptor- mediated endocytosis in peptide delivery and targeting: kinetic aspects. Advanced drug delivery reviews 19(3): 445-467.
- Rother M, Nussbaumer MG, Renggli K, Bruns N (2016) Protein cages and synthetic polymers: a fruitful symbiosis for drug delivery applications, bionanotechnology and materials science. Chemical Society Reviews 45(22): 6213-6249.
- Nussbaumer MG, Duskey JT, Rother M, Renggli K, Chami M, et al. (2016) Chaperonin-Dendrimer Conjugates for siRNA Delivery. Adv Sci (weinh) 3(10): 1600046.
- Polka JK, Silver PA (2016) A tunable protein piston that breaks membranes to release encapsulated cargo. ACS Synth Biol 5(4): 303311.
- Ragelle H, Danhier F, Preat V, Langer R, Anderson DG (2016) Nanoparticle-based drug delivery systems: a commercial and regulatory outlook as the field matures. Expert Opin Drug Deliv 14: 1-14.






























