Synthetic N-(Alkyl/Aralkyl)-N-(2,3-Dihydro-1,4-Benzodioxin-6-Yl)-4-Methylbenzenesulfonamides as Acetyl cholinesterase Inhibitors
Abbasi MA1*, AurangZeb1, Aziz-ur-Rehman1, Siddiqui SZ1, Shah SAA2,3, Ashraf M4 and Qurat-ul-Ain4
1Department of Chemistry, Government College University, Pakistan
2Department of Pharmacy, Universiti Teknologi MARA, Malaysia
3Atta-ur-Rahman Institute for Natural Products Discovery (AuRIns), Universiti Teknologi MARA, Malaysia
4Department of Chemistry, The Islamia University of Bahawalpur, Pakistan
Submission: March 31, 2017; Published: June 22, 2017
*Corresponding author: Abbasi MA, Department of Chemistry, Government College University, Pakistan, Tel: +9242111000010; Email: atrabbasi@yahoo.com/abbasi@gcu.edu.pk
How to cite this article: Abbasi M, AurangZeb, Aziz-ur-Rehman, Siddiqui S et al. Synthetic N-(Alkyl/Aralkyl)-N-(2,3-Dihydro-1,4-Benzodioxin-6-Yl)-4-Methylbenzenesulfonamides as Acetyl 002 cholinesterase Inhibitors. Nov Appro Drug Des Dev. 2017; 1(5): 555573. DOI: 10.19080/NAPDD.2017.01.555573
Abstract
The current research effort involved the reaction of 2,3-dihydro-1,4-benzodioxin-6-amine (1) with 4-methylbenzenesulfonyl chloride (2) in the presence of 10% Na2CO3 under dynamic pH control to obtain N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-methylbenzenesulfonamide (3) which was further coupled with a series of alkyl/aralkyl halides (4a-n) to attain N-alkyl/aralkyl-N-(2,3-dihydro-1,4-benzodioxin-6-yl)- 4-methylbenzenesulfonamides (5a-n), using polar aprotic solvent; DMF and catalytic amount of lithium hydride as base. The structural characterization of these newly synthesized compounds was done by IR, 1H-NMR and EIMS spectral data. All these compounds were assessed for their acetyl cholinesterase inhibitory potentials and were found to be moderate inhibitors of this enzyme. Two molecules, 5f and 5n displayed excellent to effective inhibitory potential respectively while the other showed moderate inhibitory potential against acetyl cholinesterase enzyme.
Keywords: 2,3-Dihydro-1; 4-Benzodioxin-6-amine; Sulfonamides; Spectral analysis; Acetyl cholinesterase
Introduction
The basic sulfonamide group SO2NH is found in numerous biological active compounds including antiviral, anticancer, anti-thyroid, antimicrobial, anti-inflammatory and antibiotics drugs along with inhibitors of carbonic anhydrase [1]. Because of their less cost, less toxicity and astonishing activity, these are extensively used as antibacterial agents [2-6]. Furthermore, sulfonamides are also employed as, anti-leprotic, diuretics, antitumor agents, tuberculostatics and oral hypoglycemic drugs [7-8]. Aliphatic sulfonamide derivatives act as antifungal agents [9]. Sulfonamide based antibiotics are utilized as veterinary medicines to treat infections in livestock herds [10]. Compounds bearing benzodioxane ring system exhibits different biological activities such as anti-oxidant [11], anti-hepatotoxic and anti-inflammatory [12,13]. Aryl sulfonamides having benzodioxane moiety have been recognized as inhibitors of ExoU [14]. Because of their non-interaction to defence mechanism of host and broad spectrum activity some effective derivatives of sulfonamides are widely used to treat gastrointestinal and urinary tract infections [15]. Some sulfonamides were also found to be potent carbonic anhydrase, COX-2 and caspase inhibitors [16-18].
Acetyl cholinesterase (AChE, EC 3.1.1.7) belongs to serine hydrolases class of enzymes. This enzyme system is accountable for the termination of acetylcholine at cholinergic synapses. The main function of AChE is to catalyze the hydrolysis of the neurotransmitter acetylcholine and termination of the nerve impulse in cholinergic synapses [19]. The inhibitors of this enzyme are the targets for the treatment of Alzheimer's disease [20]. Biological literature review on sulfonamides displayed that little modification in the structure of sulfonamides can cause remarkable changes in biological activity. These outcomes stimulated us to focus on synthesis of variety of N-alkyl/aralkyl-N-(2,3-dihydrobenzo[1,4]dioxin-6-yl)-4- methylbenzenesulfonamides. Recent research work was a successful attempt to synthesize new therapeutic agents for the inhibition of cholinesterase enzyme.
Experimental
Measurements and materials
All of the essential chemicals/solvents were of analytical grade and procured from authorized suppliers, Merck and Alfa Aeser branded. The pre-coated silica gel G-25-UV254 plates were applied for TLC to monitor the completion of reactions using various percentages of n-hexane and ethyl acetate as mobile phase. Open capillary tubes were used in Gallon kamp melting point apparatus to record the melting points. Developed TLC visualized under 254nm UV lamp and UV inactive substances were identified with the spray of ceric sulfate solution. Infrared spectra were noted in KBr pellet on a Jasco-320-A spectrophotometer. 1H-NMR spectra were recorded by Bruker spectrometer in CDCl3 operating at 400MHz at 25 °C. The chemicals shifts (5) were observed in ppm and coupling constants (δ) were noted in Hertz (Hz). The abbreviations used in 1HNMR spectral analysis were; s=singlet, d=doublet, dd=doublet of doublet, t=triplet, br.t=broad triplet, q=quartet, quint=quintet, sex=sextet, sep=septet, m=multiplet. Mass spectra (EIMS) were taken on Finngen Mass Spectrometer.
Synthesis of N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4- methylbenzenesulfonamide (3)
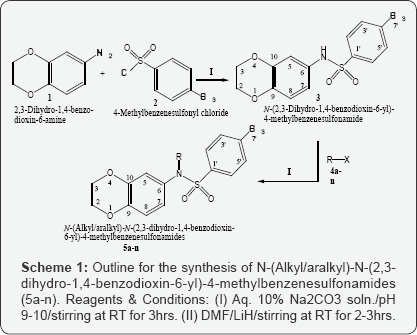
2,3-Dihydro-1,4-benzodioxin-6-amine (1.22mL;0.01mol;1) and 4-methylbenzenesulfonyl chloride (0.90g; 0.01mol; 2) were poured into a 250ml round bottom flask having 30ml of distilled water. The pH of the suspension was maintained at 9.0 by introducing 10% Na2CO3 solution at room temperature. The content of reaction was stirred for 2-3 hours and progress of the reaction was examined by TLC time to time till single spot confirm the completion of reaction. The product was obtained by the slow addition of concentrated HCl at pH 2-3 as brown coloured precipitates. These were filtered, washed with distilled water and air-dried to afford pure N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4- methylbenzenesulfonamide (3); Yield: 82%, IR (KBr, cm-1): vmax: 3284 (N-H stretching), 2970 (C-H stretching of aromatic ring], 1650 C=C stretching of aromatic ring), 1410 (SO2 stretching), 1175 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 9.87 (s, -NH, 1H), 7.58 (d, J=8.4Hz, 2H, H-2' & H-6'), 7.32 (d, J=8.0Hz, 2H, H-3' & H-5'), 6.68 (d, J=8.4Hz, 1H, H-8), 6.55 (d, J=2.0Hz, 1H, H-5), 6.50 (dd, J=2.4, 6.5Hz 1H, H-7), 4.14 (s, 4H, H-2 & H-3), 2.33 (s, 3H, H-1''); EI-MS (m/z): 305 (M+; C15H15NO4S), 304 (C15H14NO4S)+, 277 (C13H11NO4S)+, 241 (C15H15NO2)+, 213 (C8H7NO4S)+, 155 (C7H7SO2)+, 150 (C8H8NO2)+,, 135(C8H7O2)+,, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+, 65 (C6H5)+, 51 (C4H3)+.
General procedure for the synthesis of N-substituted derivatives 5a-n
Compound 3 (0.1g; 0.3mmol) solubilised in 10ml of N, N-dimethyl formamide (DMF) aprotic solvent in 100ml round bottom flask. Lithium hydride (0.004g) was mixed in the reaction mixture to activate the reaction followed by stirring for 2-3 hours at room temperature. Then various alkyl/aralkyl halides (4a-n) were introduced and stirring was continued for further 3-4 hours. Completion of reaction was assured by TLC displaying single spot. Then reaction content was quenched with ice cold distilled water along with vigorous shaking to get the precipitates of N-alkyl/aralkyl-N-(2,3-dihydro-1,4-benzodioxin- 6-yl)-4-methylbenzenesulfon-amides (5a-n) which were left for some time undisturbed and collected by the filtration or solvent extraction (using CHCl3) depending upon the nature of the derived compound.
Spectral analysis
The spectral analysis of 5b, 5g, 5h, 5j and 5l has already been reported by our group [21] while that of other compounds is given hereby.
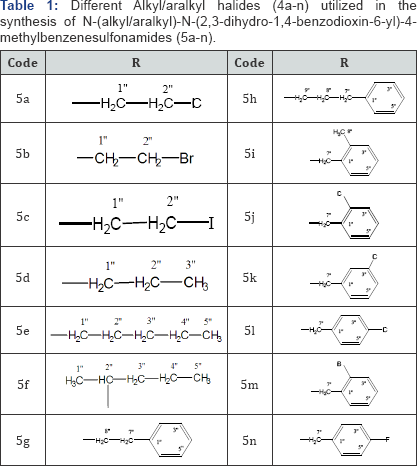
N-(2-Chloroethyl)-N-(2,3-dihydro-1,4-benzodioxin-6- yl)-4-methylbenzenesulfonamide (5a): Off white powder; Yield: 95 %; m.p: 142 °C; Molecular formula: C17H18ClNO4S; Molecular weight: 371g mol-1; IR (KBr, cm-1): vmax: 2976 (C-H stretching of aromatic ring), 1657 (C=C stretching of aromatic ring), 1395 (-SO2 stretching), 1140 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 5 7.50 (d, J=8.5Hz, 2H, H-2' & H-6'), 7.40 (d, J=8.4Hz, 2H, H-3' & H-5'), 6.66 (d, J=7.6Hz, 1H, H-8), 6.50 (dd, J=2.5, 8.2Hz, 1H, H-7), 6.42 (d, J=2.6Hz, 1H, H-5),4.28 (br.s, 4H, CH2-2 & CH2-3), 3.60 (t, J=6.5Hz, 2H, CH2-2"), 3.46 (t, J=7.4Hz, 2H, CH2-1"), 2.34 (s, 3H, CH3-7'); EI-MS (m/z): 371 (M+ C17H18CINO4S), 340 (C15H14ClNO4S)+, 304 (C15H14NO4S)+, 300 (C12H11ClNO4S)+,, 237 (C12H11ClNO2)+, 213 (C10H11ClNO2)+, 155 (C7H7SO2+, 135(C8H7O2)+, 107(C6H3O2)+, 91(C7H7)+, 81(C4HO2)+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-N-(2-Iodoethyl)- 4-methylbenzenesulfonamide (5c): Greyish white solid; Yield: 95%; m.p: 132 °C; Molecular formula: C17H18INO4S; Molecular weight: 459g mol-1; IR (KBr, cm-1): vmax: 2989 (C-H stretching of aromatic ring), 1660 (C=C stretching of aromatic ring), 1379 (-SO2 stretching), 1168 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 5 7.50 (d, J=7.4Hz, 2H, H-2' & H-6'), 7.42 (d, J=8.5Hz, 2H, H-3' & H-5'), 6.68 (d, J=6.68Hz, 1H, H-8), 6.54 (d, J=2.5Hz, 1H, H-5), 6.40 (dd, J=2.5, 8.5Hz, 1H, H-7), 4.26 (br.s, 4H, CH2-2 & CH2-3), 3.72 (t, J=7.5Hz 2H, CH2-1''), 3.36 (t, J=6.8Hz, 2H, CH2-2"), 2.36 (s, 3H, CH3-7'); EI-MS (m/z): 459 [M+ C17H18IO4S], 431 (C15H14INO4S)+, 395 (C17H18INO2)+, 368(C10HuINO4S)+,, 326 (C10H10INO2)+, 304 (C15H14NO4S)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4-methyl-N-(1-propyl)benzenesulfonamide (5d): Dark brown powder; Yield: 88%; mp: 136 °C; Molecular formula: C18H21NO4S; Molecular weight: 347g mol-1; IR (KBr, cm-1): vmax: 2988 (C-H stretching of aromatic ring), 1662 (C=C stretching of aromatic ring), 1374 (SO2 stretching), 1148 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 5 7.46 (d, J=8.2Hz, 2H, H-2' & H-6'), 7.34 (d, J=8.5Hz, 2H, H-3' & H-5'), 6.68 (d, J=7.5Hz, 1H, H-8),6.40 (d, J=2.5Hz, 1H, H-5), 6.22 (dd, J=2.6, 8.4Hz, 1H, H-7), 4.28 (br.s, 4H, CH2-2 & CH2-3), 3.10 (t, J=7.5Hz, 2H, CH2-1''), 2.34 (s, 3H, CH3-7'), 1.60-1.56 (m, 2H, CH2-2''), 1.05 (t, J=7.4Hz, 3H, CH3-3''); EI-MS (m/z): 347 (M+ C18H21NO4S), 319 (C16H17NO4S)+, 304 (C15H14NO4S)+, 283 (C18H21No2)+, 256 (C11H14NO4S)+, 178 (C11H14NO2)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4-methyl-N-(1-pentyl)benzenesulfonamide (5e): Brown powder; Yield: 85%; m.p: 114 °C; Molecular formula: C20H25NO4S; Molecular weight: 375g mol-1; IR (KBr, cm-1): vmax: 2984 (C-H stretching of aromatic ring), 1678 (C=C stretching of aromatic ring), 1376 (SO2 stretching), 1142 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400 MHz, δ in ppm): 5 7.40 (d, J=8.4Hz, 2H, H-2' & H-6'), 7.34 (d, J=8.5Hz, 2H, H-3' & H-5'), 6.66, (d, J=7.8Hz, 1H, H-8), 6.44 (dd, J=2.7, 8.5Hz, 1H, H-7), 6.28 (d, J=2.6Hz, 1H, H-5), 4.28 (br.s, 4H, CH2-2 & CH2-3), 3.20 (t, J=7.5Hz, 2H, CH2-1''), 2.34 (s, 3H, CH3-7'), 1.40-134 (m, 6H, CH2-2" to CH2-4"), 0.90 (t, J=7.6Hz, 3H, CH3-5"); EI-MS (m/z): 375 (M + C20H25NO4S), 347 (C18 H21NO4S)+, 311 (C20H25NO2)+, 304 (C15H14NO4S)+, 284 (C13H18NO4S)+, 220 (C13H18NO2)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+, 71 (C5H2+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4-methyl-N-(2-pentyl)benzenesulfonamide (5f): Brown solid; Yield: 92%; m.p: 124 °C; Molecular formula: C20H25NO4S; Molecular weight: 375g mol-1; IR (KBr, cm-1): vmax: 2994 (C-H stretching of aromatic ring), 1660 (C=C stretching of aromatic ring), 1372 (-SO2 stretching), 1144 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 57.44 (d, J=8.5Hz, 2H, H-2' & H-6'), 7.30 (d, J=8.6Hz, 2H, H-3' & H-5'), 6.64, (d, J=8.6Hz, 1H, H-8), 6.54 (dd, J=2.6, 8.8Hz, 1H, H-7), 6.30 (d, J=2.5Hz, 1H, H-5), 4.28 (br.s, 4H, CH2-2 & CH2-3), 2.84-2.80 (m, 1H, H-2"), 2.34 (s, 3H, CH3-7'), 1.51-1.50 (m, 2H, CH2-3"), 1.34 (Sext., J=7.4Hz, 2H, CH2-4"), 0.90 (t, J=7.5Hz, 3H, CHr5"); EI-MS (m/z): 375 (M+; C20H25NO4S), 347 (C18 H21NO4S)+, 311 (C20H25NO2)+, 304 (C15H14N04S)+, 284(C13H18NO4S)+, 220 (C13H18NO2)+, 155 (C7H7SO2)+, 135 (C8H7O2)+,107 (C6h3O2+, 91 (C7H7)+, 81 (C4HO2)+, 71 (CSHU)+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4-methyl-N-(2-methybenzyl)benzenesulfonamide (5i): Brown powder; Yield: 88%; m.p: 160 °C; Molecular formula: C23H23NO4S; Molecular weight: 409g mol-1; IR (KBr, cm-1): vmax: 2998 (C-H stretching of aromatic ring), 1680 (C=C stretching of aromatic ring), 1374 (-SO2 stretching), 1162 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400 MHz, δ in ppm): 5 7.44 (d, J=8.4Hz, 2H, H-2' & H-6'), 7.38 (d, J=7.7Hz, 2H, H-3' & H-5'), 7.34-7.28 (m, 4H, H-3'' to H-6''), 6.60, (d, J=7.5Hz, 1H, H-8), 6.50 (dd, J=2.5, 8.5Hz, 1H, H-7), 6.25 (d, J=2.6Hz, 1H, H-5), 4.38 (s, 2H, CH2-7''),4.28 (br.s, 4H, CH2-2 & CH2-3), 2.34 (s, 3H, CH3-7'), 2.26 (s, 3H, CHr8"); EI-MS (m/z): 409 (M+; C23H23N04S), 381 (C21H19NO4S)+, 345 (C23H23NO2)+, 318 (C16H16NO4S)+, 304 (C15H14NO4S)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 105 (C8H9)+, 107 (C6H3O2)+, 91(C7H7)+, 81 (C4HO2)+.
N-(3-Chlorobenzyl)-N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-methylbenzenesulfonamide (5k): Grey powder; Yield: 96%, m.p: 152 °C; Molecular formula: C22H20ClNO4S; Molecular weight: 429gmol 1; IR (KBr, cm-1): vmax: 3010 (C-H stretching of aromatic ring), 1672 (C=C stretching of aromatic ring), 1382 (-SO2 stretching), 1090 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400 MHz, δ in ppm): 5 7.50 (br.s, 1H, H-2"), 7.42 (d, J=8.8Hz, 2H, H-2' &H-6'), 7.36 (d, J=8.5Hz, 2H, H-3' & H-5'), 7.30-7.25 (m, 3H, H-4" to H-6'"), 6.66, (d, J=6.5Hz, 1H, H-8), 6.52 (dd, J=2.6, 8.5Hz, 1H, H-7), 6.18 (d, J=2.6Hz, 1H, H-5), 4.42 (s, 2H, CH2-7''), 4.28 (br.s, 4H, CH2-2 & CH2-3), 2.36 (s, 3H, CH3- 7'); EI-MS (m/z): 429 (M+; C22H20ClNO4S), 402 (C20H16ClNO4S)+, 365 (C22H20ClNO2)+, 338 (C15H13ClNO2)+, 304 (C15H14N04S)+, 274 (C15H13ClNO2)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 171 (C7H6Cl)+, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+.
N-(2-Bromobenzyl)-N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-methylbenzenesulfonamide (5m): Brown powder; Yield: 96%; m.p: 168 °C; Molecular formula: C22H20BrNO4S; Molecular weight: 475gmol-1; IR (KBr, cm-1): vmax: 3010 (C-H stretching of aromatic ring), 1670 (C=C stretching of aromatic ring), 1366 (-SO2 stretching), 1156 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 57.50 (br.d, J=6.8Hz, 1H, H-3''), 7.42 (d, J=8.5Hz, 2H, H-2' &H-6'), 7.36 (d, J=8.5Hz, 2H, H-3' & H-5'), 7.28-7.24 (m, 4H, H-4'' to H-6"), 6.62, (d, J=6.8 Hz, 1H, H-8), 6.48 (dd, J=2.5, 8.6Hz, 1H, H-7), 6.20 (d, J=2.8Hz, 1H, H-5),4.41 (s, 2H, H-7"), 4.28 (br.s, 4H, CH2-2 & CH2-3), 2.34 (s, 3H, CH3- 7'); EI-MS (m/z): 475 [M+; C22H20BrNO2], 447 (C20H16BrNO4S)+, 411 (C22H20BrNO2)+, 320 (C15Hl3BrN0l)+, 304 (C15H14NO4S)+, 384 (C15H13BrNO4S)+, 171 (C7H6Br)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 107 (C6H0l)+, 91 (C7H7)+.
N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-4-methyl-N-(4-fluorobenzyl)benzenesulfonamide (5n): Light grey powder; Yield: 90%, m.p: 122 °C; Molecular formula: C22H20FN04S; Molecular weight: 413g mol 1; IR (KBr, cm-1): vmax: 2994 (C-H stretching of aromatic ring), 1686 (C=C stretching of aromatic ring), 1380 (-SO-, stretching), 1195 (C-F stretching), 1086 (C-O-C stretching of ether); 1H-NMR (CDCl3, 400MHz, δ in ppm): 57.44 (d, J = 8.6Hz, 2H, H-2' & H-6'), 7.34 (d, J = 7.5Hz, 2H, H-3' & H-5'), 7.38 (dd, J=5.3, 8.5Hz, 2H, H-2” & H-6”, multiplicity due to coupling of F19), 7.14 (br.t, J=8.6, 2H, H-3” & H-5”, due to coupling of F19), 6.62, (d, J=7.5Hz, 1H, H-8), 6.50 (dd, J=2.5, 7.8Hz, 1H, H-7), 6.20 (d, J=2.5Hz, 1H, H-5), 4.36 (s, 2H, CHr7"),4.28 (br.s, 4H, CH2-2 & CH2-3), 2.32 (s, 3H, CH3-7'); EI-MS (m/z): 413 (M+; C22H20FNO4S), 385 (C20H16FNO4S)+, 349 (C22H20FNO2)+, 322 (C15H13FNO4S)+, 304 (C15H14NO4S)+, 155 (C7H7SO2)+, 135 (C8H7O2)+, 109 (C7H6F)+, 107 (C6H3O2)+, 91 (C7H7)+, 81 (C4HO2)+, 65 (C6H5)+, 51(C4H3)+.
Acetylcholinesterase inhibition assay
The inhibition activity of acetylcholinesterase was performed by according to a reported method [22] with little modifications. Total volume of the reaction mixture was 100μL and it also contained 60μL Na2HPO4, which acts as buffer having 50mm concentration (pH 7.7). Ten μL test compound 0. 5mm well-1, followed by the addition of 10μl (0.5 unit well-1) enzyme. The reaction contents was agitated well and read prior at the wavelength 405nm. Then mixture was pre-incubated at 37 °C for 10 minute. The reaction was started by adding 10μl of 0.5mm well-1 substrate (acetylthiocholine iodide) followed by the addition of 10μl DTNB (0.5mm well-1). After 15 min of incubation at 37 °C, absorbance was recorded at 405nm using 96-well plate reader Synergy HT (BioTek, USA). Each and every experiment was carried out with their particular controls in triplicate. Eserine (0.5mm well-1) was act as positive control. The equation that was used to calculate % inhibition was:
Inhibition (%)=(Control-Test)/Controlx100
IC50 values (concentration at which there is 50% enzyme inhibition) of compounds were calculated using EZ-Fit Enzyme kinetics software (Perella Scientific Inc. Amherst, USA).
Statistical analysis
All the measurements were done in triplicate and statistical analysis was performed by Microsoft Excel 2010. Results are presented as mean ± SEM.
Results and Discussion
Chemistry
The synthesis of various derivatives, 5a-n, derived by the N-substitution of N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4- methylbenzenesulfonamide (3) has been outlined in Scheme 1 and Table 1. All methods and conditions for this research work are mentioned in experimental section. The synthesis was initiated by the reaction of N-2,3-dihydro-1,4-benzodioxine-6-amine (1) with 4-methylbenzenesulfonyl chloride (2) in the presence of 10% Na2CO3 at adjusted pH 9 under constant stirring for 2-3 hours at room temperature to yield the parent sulfonamide, N-(2,3- dihydro-1,4-benzodioxin-6-yl)-4-methylbenzenesulfonamide (3). Then, N-alkylation/aralkylation of this parent 3 was carried out with different alkyl/aralkyl halides (4a-n) in DMF as a polar aprotic solvent and LiH as the base to yield the target compounds, 5a-n. We have already reported 5b, 5g, 5h, 5j and 5l along with their structural characterizations [21], however, we are reporting other compounds as new molecules in this investigation. The structures of the studied molecules were deduced through IR, 1H-NMR and EI-MS spectral techniques. For the expediency of the readers, one of the compounds is discussed hereby in detail. The molecule 5f was obtained as brown solid having melting point 124 °C. The molecular formula C20H25NO4S of this molecule was established by its EI-MS showing the molecular ion peak at m/z 375 and by counting the number of protons in its 1H-NMR spectrum. The subsequent fragmentation peaks in its EI-MS spectrum also supported this assignment. The IR spectrum showed absorption bands at 2994, 1660, 1372 and 1144 for the C-H stretching of aromatic ring, C=C stretching of aromatic ring, SO2 and C-O-C stretching respectively. In its 1H-NMR spectrum, the presence of a 4-methylbenzenesulfonyl moiety was assured by two ortho-coupled doublets at 5 7.44 (d, J = 8.5Hz, 2H, H-2', H-6') and 7.30 (d, J = 8.6Hz, 2H, H-3', H-5') along with a singlet at 5 2.34 (s, 3H, CH3-7') for methyl group. The 2,3-dihydro-1,4- benzodioxin-6-yl moiety in this molecule was corroborated by an otho-coupled doublet at 5 6.64 (d, J=8.6Hz, 1H, H-8), a meta-coupled doublet at 5 6.30 (d, J=2.5Hz, 1H, H-5) and corresponding doublet of doublet at 5 6.54 (dd, J=2.6, 8.8Hz, 1H, H-7) along with a broad singlet at 5 4.28 (br.s, 4H, CH2-2 & CH2- 3) for two oxygenated methylenes. The N-substituted 2-pentyl group in this molecule was characterized by its typical signals in aliphatic region at 5 2.84-2.80 (m, 1H, H-2"), 1.51-1.50 (m, 2H, CH2-3"), 1.34 (Sext., J=7.4Hz, 2H, CH2-4"), and 0.90 (t, J=7.5Hz, 3H, CH3-5"). So, on the basis of above collected evidences, the structure of 5f was named as N-(2,3-dihydro-1,4-benzodioxin-6- yl)-4-methyl-N-(2-pentyl) benzenesulfonamide. In an analogous manner, the structures of other derivatives of the series were characterized [22].
Acetylcholinesterase inhibition
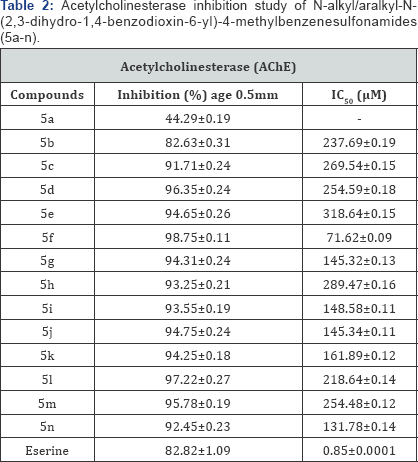
The screening of all the derivatives 5a-n, against acetylcholinesterase (AChE) enzyme demonstrated that all the molecules of the series were active, except 5a. These molecules exhibited moderate to weak inhibitory potential and the results are tabulated in Table 2 in the form of % age inhibition and IC50 values. Among these molecules, 5f, was found to be better inhibitor against this enzyme having IC50 value of 71.62±0.09μM, probably due to the substitution of a branched aliphatic group i. e. 2-pentyl group. The molecule, 5n, having substitution of 4-fluorobenzyl group, also showed notable inhibitory potential with IC50 value of 131.78±0.14μM. An extremely potent, eserine molecule was used as reference standard in this assay which has an IC50 value of 0.85±0.0001μM.
Conclusion
The targeted derivatives, 5a-n, were synthesized in good yields with a facile method and some of them exhibited a notable inhibitory potential against acetyl cholinesterase enzyme, therefore, these molecules might find their utility as possible therapeutic agents for the treatment of Alzheimer's disease.
Acknowledgement
Special thanks are paid to the Higher Education Commission (HEC) of Pakistan for financial grant to execute this study.
References
- Abbasi MA, Tariq S, Rehman A, Zahra SS, Irshad A, et al. (2016) Synthesis of some new N-substituted-N-(2,3-Dihydro-1,4 benzodioxin-6-yl)-4- acetamidobenzenesulfonamides as valuable antibacterial agents. Russ J Bioorg Chem 42(2): 198-209.
- Abdulhakeem A, Abdel-Zaher AE, Seham M, Fakhreia AlS (2011) Synthesis, structure analysis and antibacterial activity of new potent sulfonamide derivatives. Bioorg Med Chem Lett 2: 144-149.
- Santosh K, Niranjan MS, Chaluvaraju KC, Jamakhandi CM, Dayanand K (2010) Synthesis and antimicrobial study of some schiff bases of sulfonamides. J Curr Pharm Res 1: 39-42.
- Jeremy MB, Wang Z (2002) A mild, convenient synthesis of sulfinic acid salts and sulfonamides from alkyl and aryl halides. Tetrahedron Lett 43(47): 8479-8483.
- Thakur A, Thakur M, Khadikar PV (2006) QSAR study on inhibition of E. Coli by sulfonamides. ARKIVOC 2006(14): 87-102.
- Shi F, Tse MK, Zhou S, Phol MM, Radnik J, et al. (2009) Green and Efficient Synthesis of Sulfonamides Catalyzed by Nano-Ru/Fe3O4. J Am Chem Soc 131(5): 1775-1779.
- Mirian M, Zarghi A, Sadeghi S, Tabaraki P, Tavallaee M, et al. (2011) Synthesis and cytotoxic evaluation of some novels sulfonamide derivatives against a few human cancer cells. Iran J Pharm Res 10(4): 741-748.
- Harry J Strek (1998) Fate of chlorsulfuron in the environment. 1. Laboratory evaluations. Pestic Sci 53(1): 29-51.
- Ozbek N, Katircioglu H, Karacan N, Baykal T (2007) Synthesis, characterization and antimicrobial activity of new aliphatic sulfonamide. Bioorg Med Chem 15(15): 5105-5109.
- Andrews KT, Fisher GM, Subathdrage DMS, Skinner-Adams T, Skinner- Adams T, et al. (2013) Antimalarial activity of compounds comprising a primary benzene sulfonamide fragment. Bioorg Med Chem Lett 23(22): 6114-6117.
- Mallesha L, Kikkeri NM (2011) Synthesis, antimicrobial and antioxidant activities of 1-(1,4-benzodioxane-2-carbonyl)piperazine derivatives. Eur J Chem 2(2): 193.
- Ahmed B, Khan SA, Alam T (2003) Synthesis and anti-hepatotoxic activity of some heterocyclic compounds containing the 1,4-dioxane ring system. Pharmazie 58(3): 173-176.
- Khan SA, Ahmed B, Alam T (2006) Synthesis and antihepatotoxic activity of some new chalcones containing 1, 4-dioxane ring system. Pak J Pharm Sci 19(4): 290-294.
- Irshad M, Athar MA, Rehman A, Zahra SS, Ashraf M, et al. (2014) Synthesis, characterization and biological screening of some new sulfonamides derivatives of 1,4-benzodioxane-6-amine. J Chem Soc Pak 36(4): 660-673.
- Gadad AK, Mahajanshetti CS, Nimbalkar S, Raichurkar A (2000) Synthesis and antibacterial activity of some 5-guanylhydrazone/ thiocyanato-6-arylimidazo[2,1-b]-1,3, 4-thiadiazole-2-sulfonamide derivatives. Eur J Med Chem 35(9): 853-857.
- Brendan LW, Laurent FB, Todd AH, Alessio I, Daniela V, et al. (2007) Carbonic anhydrase inhibitors: Inhibition of isozymes I, II, and IX with triazole-linked O-Glycosides of benzene sulfonamides. J Med Chem 50(7): 1651-1657.
- Almansa C, Bartroli J, Belloc J, Cavalcanti FL, Ferrando R, et al. (2004) New water-soluble sulfonylphosphoramidic acid derivatives of the COX-2 selective inhibitor cimicoxib. A novel approach to sulfonamide prodrugs. J Med Chem 47(22): 5579-5582.
- Chu W, Rothfuss J, Zeng C, Zhou D, Hotchkiss RS, et al. (2007) Isatin sulfonamide analogs containing a Michael addition acceptor: a new class of caspase 3/7 inhibitors. J Med Chem 50(15): 3751-3755.
- Cygler M, Schrag JD, Sussman JL, Harel M, Silman I (1993) Relationship between sequence conservation and three-dimensional structure in a large family of esterases, lipases, and related proteins. Protein Sci 2(3): 366-382.
- Athar MA, Amna S, Aziz-ur-R, Mohmmed KK, Muhammad A, et al. (2014) Synthesis of Brominated 2-Phenitidine Derivatives as Valuable Inhibitors of Cholinesterases for the Treatment of Alzheimer's Disease. Iran J Pharm Res 13(1): 87-94
- Athar MA, Aziz-ur R, Zahra SS, Sheeza ASN, Ahmad I, et al. (2017) Synthesis, antibacterial and lipoxygenase inhibition studies of N-(Alkyl/aralkyl)-N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-methylbenzenesulfonamides. Turk J Pharm Sci 14(1): 49-55.
- Tougu V (2001) Acetylcholinesterase: Mechanism of Catalysis and Inhibition. Curr Med Chem 1(2): 155-170.






























