Mechanistic Aspects of Autism Involving Electron Transfer, Reactive Oxygen Species, Oxidative Stress, Pollutants, Antioxidants, Cell Signaling and Genes
Peter Kovacic1* and Ratnasamy Somanathan2
1Department of Chemistry and Biochemistry, San Diego State University, San Diego,CA92182-1030,USA
2Centro de Graduados e Investigación del instituto Tecnológico de Tijuana, apdo. postal 1166,Tijuana,B.C.Mexico
Submission: May 05,2017; Published: June 06, 2017
*Corresponding author: Peter Kovacic, Department of Chemistry and Biochemistry, San Diego State University, USA, Tel: +619594-5595;Fax: +619594-4634; Email: pkovacic@mail.sdsu.edu; rsomanathan@mail.sdsu.edu
How to cite this article: Peter K, Ratnasamy S. Mechanistic Aspects of Autism Involving Electron Transfer, Reactive Oxygen Species, Oxidative Stress, Pollutants, Antioxidants, Cell Signaling and Genes. Nov Appro Drug Des Dev. 2017; 1(3): 555562. DOI: 10.19080/NAPDD.2017.01.555562
Abstract
Autism is a center of attention both in research and the media. This review is mainly concerned with mechanistic involvement of electron transfer, reactive oxygen species, oxidative stress and antioxidants, in addition to cell signaling and genetic aspects. Pollution from the environment is a factor. The most sensitive stage is the fetus. The neuronal network in the brain plays an important role. Antioxidants can act in prevention or amelioration. Interaction between the various aspects is addressed. The effect of vaccine is discussed.
Keywords: Autism; Electron transfer; Radicals; Oxidative stress; Pollutants; Antioxidants; Genes
Abbrevuations: ET: Electron Transfer; ROS: Reactive Oxygen Species; OS: Oxidative Stress; AO: Anti Oxidant
Introduction
Autism has attracted much attention in recent years, both in research and in the media. This review summarizes evidence for important involvement of electron transfer (ET), reactive oxygen species (ROS), oxidative stress (OS), antioxidants (AOs) and genes. Evidence points to a key role for pollution. The fetus is the stage most susceptible to damaging effects, mostly to the brain and central nervous system. The unifying mechanistic theme has been widely applied previously as set forth as follows:
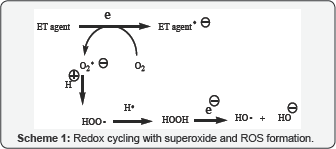
"The preponderance of bioactive substances, usually as the metabolites, incorporate ET functionalities. We believe these play an important role in physiological responses. The main group include quinones (or phenolic precursors), metal complexes (or complexors), aromatic nitro compounds (or reduced hydroxylamine and nitroso derivatives), and conjugated imines (or iminium species). Resultant redox cycling is illustrated in (Scheme 1). In vivo redox cycling with oxygen can occur, giving rise to oxidative stress (OS) through generation of reactive oxygen species (ROS), such as hydrogen peroxide, hydro peroxides, alkyl peroxides, and diverse radicals (hydroxyl, alkoxyl, hydro peroxyl, and superoxide) (Scheme 1). Cellular and mitochondrial enzymes can also perform catalytically in the reduction of O2.
In some cases ET results in involvement with normal electrical effects (e.g., in respiration or neurochemistry). Generally, active entities possessing ET groups display reduction potentials in the physiologically responsive range, (i.e., more positive than about -0.5 V). Hence, ET in vivo can occur resulting in production of ROS which can be beneficial in cell signaling at low concentrations, but produce toxic results at high levels. Electron donors consist of phenols, N-hetero cycles or disulfides in proteins which produce relatively stable radical cations. ET, ROS and OS have been increasingly implicated in the mode of action of drugs and toxins, (e.g., anti-infective agents [1], anticancer drugs [2], carcinogens [3], reproductive toxins [4], nephrotoxins [5], hepatotoxins [6], cardiovascular toxins [7], nerve toxins [8],mitochondrial toxins [8], abused drugs [9], pulmonary toxins [10] ototoxins [11] and various other categories [12].
There is a plethora of experimental evidence supporting the ET-ROS theoretical framework [1-12]. This evidence includes generation of the common ROS, lipid per oxidation, degradation products of oxidation, depletion of AOs, effect of exogenous AOs, and DNA oxidation and cleavage products, as well as electrochemical data. This comprehensive, unifying mechanism is consistent with the frequent observation that many ET substances display a variety of activities (e.g., multiple-drug properties), as well as toxic effects. It is important to recognize that mode of action in the bio domain is often involved with many physiological actions and is multifaceted. In addition to the ET- ROS-OS approach, other aspects may pertain, such as, enzyme inhibition, allosteric effects, receptor binding, metabolism and physical factors. A specific example involves protein binding by quinones in which protein and nucleophiles, such as amino or thiol, effect conjugate addition" [13].
Pollution and ET-ROS-OS
A recent study deals with effect of air pollution on rates of autism in different locations [14,15]. The rate in California was related to that in North Carolina involving similar environmental chemical pollution. A similar study showed autism spectrum disorders in relation to distribution of hazardous air pollutants in the San Francisco Bay area [16]. The temporal trends in autism for birth years 1970-2005 were studied. Autism prevalence has risen dramatically in the U.S over the last several decades and this effect is correlated to environmental factors [17]. Also, women in the third trimester of pregnancy were more susceptible to the damaging effects of air pollution on the fetus. Focus of the research was on exposure to coarse and fine particulate matter in the air. Results are consistent with the theory involving links between autism and altered brain neuro development, specifically synaptic connections.
A recent, relevant review involves the unifying mechanism of ET-ROS-OS. An investigation revealed perinatal exposure to the highest and lowest quintile of diesel, lead, manganese and cadmium, and an overall measure of metals were significantly associated with autism spectrum disorders [18]. Autism is also related to toxicity of poly nuclear aromatic nitro compounds present in pollutants from engine exhausts [19]. The report provides a mechanistic rationale at the molecular level for the adverse effects. As indicated in the Introduction, aromatic nitro compounds comprise one of the classes of ET agents. Generally, the nitroso metabolite serves as a better agent for ET leading to redox cycling with generation of ROS-OS.
The literature contains numerous reports dealing with involvement of ROS-OS in autism. The articles involve various aspects of the association. A review on physiological abnormalities in autism places focus on immune dysregulation, OS, inflammation, environmental toxicity and mitochondrial dysfunction [20]. Autistic children exhibit higher urinary levels of aspartame which is an indication of lipid per oxidation [21]. Levels of per oxidation correlated with vascular biomarker ratios. Hence, increased OS could play a role in autism development and manifestations. Results indicate that bisphenol A, an important industrial chemical, induces enhanced OS and mitochondrial dysfunction in autism [22]. A review presents a relation between phenol and toxicity based on ET-ROS-OS [23]. Genetic predisposition and environmental poisons have been associated with autism [24]. Enhanced concentrations of 3-nitrotyrosine, an indicator of OS, are present in the cerebella of autistics. Findings indicate brain changes in the OS marker. Camel milk, an AO, is a potential therapeutic agent in autism [25]. There is decrease in OS by AO enzymes and other AO molecules. ROS are known to be involved in many neuro psychiatric disorders [26]. NO and other agents related to OS may play a role. An article raises the question: could oxidative stress (OS) from psychosocial stress affect neurodevelopment in autism? [27]. There is increasing support for involvement of OS and signaling in autism [28]. An up regulation of signaling results in disturbance of OS homeostasis that leads to increased risk of autism. Autistic children are more susceptible to OS in the form of enhanced lipid per oxidation and deficiency of AOs [29]. There may be benefit from AO supplementation. AO enzymes, SOD and GSH peroxidase, were employed. Levels of malondialdehyde, an indication of lipid per oxidation, were measured. Early data on AO status may lead to less OS before brain injury can occur. Studies reveal that autism is associated with OS, mitochondrial dysfunction, inflammation and immune dysregulation [30]. ROS production and elevated OS are present in autism [31]. The condition is responsible for damage to mitochondrial DNA. A combination of genetic and epigenetic factors in utero leads to DNA alterations similar to that in older individuals. The importance of external factors, including environmental pollution, is related to increase in autism [32]. The condition is considered to be an epidemic. Toxic factors, such as OS, may be responsible for nerve injury to the brain. Multiple forces may interplay leading to greater vulnerability to OS, toxicity and neuronal insult. The neuro disorders of autism are believed to be related to OS arising from ROS, which may be a target for therapy [33]. GSH can serve as an AO for protection against ROS and neuro inflammation. Decreasing OS could be a treatment. Genetic factors may be involved in autism, including abnormal genes of OS pathways and increased OS [34]. In a related report, the relationship of gene polymorphism and OS were studied [35]. Change in iron metabolism in the CNS may be a factor in autism. As noted in the Introduction, metal compounds, such as Fe, are well known ET agents. An autism hypothesis is based on a connection between OS and altered sulfur metabolism [36]. Environmental bacterial contaminants might result in increased OS. 8-OH-dG, a well known product arising from oxidative damage to DNA by ROS, is present at increased concentrations in autism [37]. The investigation involved cerebella DNA. A report is based on the idea that there is interaction between genetic and environmental factors with involvement of OS [38]. A result may be a change in redox status. Lipid per oxidation is enhanced as part of the scenario. Immune cells in autism exhibit greater oxidation overall [39]. A deficiency exists in GSH redox/AO capacity. Findings indicate that loss of the redox homeostasis and chronic OS may lead to immune dysregulation. In the autistic condition, there are lower levels of AOs, such as GSH, cysteine and homo cysteine [40]. The condition results in enhanced danger of OS. A related study led to a similar conclusion [41]. Another article deals with a link between OS and erythrocyte membrane alterations[42]. Greater OS in autism is characterized by enhanced free radical production, impaired energetic and higher excitotoxicity [43]. The abnormal brain and gut are more prone to oxidative insult. Increased red cell lipid peroxides and urine isoprostanes, products of OS, point to enhanced oxidative attack. Powerful AOs, e.g., vitamin C, improved the behavior of patients. The benefits appear to be related to lesson ed OS, an understanding of which should be beneficial. A mechanism is proposed linking OS in autism with abnormal membrane lipids, inflammation, altered immune response, excitotoxicity and changed energy metabolism [44]. These factors play an important role in clinical symptoms and pathogenesis. A 2005 article deals with a general overview of OS in the disease [45].
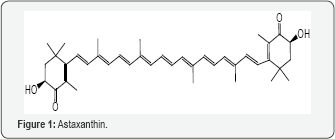
A recent review deals with environmental toxicants and autism [46]. Although a role for genes is widely acknowledged, there is evidence for equal involvement of environmental pollutants. The toxic materials include heavy metals, air toxicants, site waste, solvents, polychlorinated biphenyls, phthalates and pesticides, with the strongest being pesticides and air pollutants. Genes and environmental toxicants may act synergistically during neurodevelopment. In utero exposure to air pollutants was studied in Los Angeles in relation to autism [47]. The pollutants included 1,3-butadiene, formaldehyde, Perchloroethylene, lead and aromatic solvents, such as xylenes. The conclusion was that autism in children may increase after in utero exposure to toxic air materials from industrial emissions and traffic. Oxidative stress was examined as a factor in autism and possible target in therapy [48]. In recent years, OS has been a focus in the pathogenesis of various neuropsychiatric disorders including autism. Evidence points to higher levels of OS and lower levels of AO defenses in the brain. The review addresses the role of OS and oxidative balance, together with therapeutic strategies. In a study, a number of autistic children exhibited a lower level of oxidized GSH, a biomarker of OS, in a clean room environment [49]. Improvement in 4 of 5 markers of OS was observed. Reduced AO levels were noted in autistic Chinese children which is a sign of OS [50]. Lowered AO concentrations were recorded for the thiols homocystiene and GSH. The pathogenesis of autism is often associated with OS in the brain [51]. Administration of astaxanthin (Figure 1) proved beneficial and diminished OS in several organs, including the brain. The extensively conjugated dicarbonyl can act as an AO or a pro-oxidant. As an AO, the absorbed electron in ET can be donated to an oxidant or an electron can be provided from the long conjugated system. As an oxidant, the vinylogous conjugated system could participate in ET by generating ROS (52).
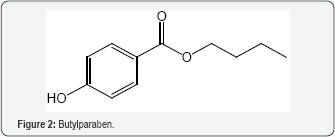
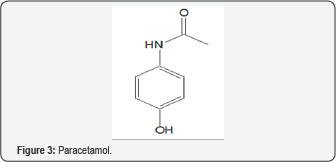
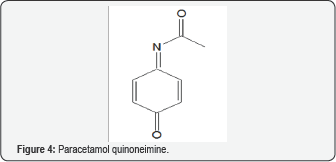
A cell's resistance to OS depends on genes for RNA in the cell's genome. OS markers are found in most studies of autism. Evidence indicates a connection between autism and mitochondrial dysfunction [53]. The dysfunction may make the ill children more sensitive to sources of ROS, such as immune activation and pro-oxidant external toxicants. Various substances are reported to influence the condition. Often there is a connection to the unifying theme of ET-RSO-OS. A report describes autism as a form of heavy metal toxicity [54]. Lead and mercury are believed to be the main causes of autism. Detoxification by chelating agents leads to improvement in the condition [55]. A 2008 review article deals with the mercury provoked autism in children [56]. Related articles showed severity of autism is associated with toxic metal body burden and red blood cell glutathione levels [57,58]. A study showed the connectivity between toxic metals in the hair and autism spectrum disorder in young children [59,60]. The Introduction points to the role of heavy metals in the thesis of ET-ROS-OS, Butyl paraben (Figure 2), a potent estrogen, is a preservative which is suspected as contributor to the autistic condition [61]. The compound increased OS, decreased GSH levels, elevated GSSG levels, elevated mitochondrial dysfunction and increased protein oxidation, along with increase in 3-nitrotyrosine. Phenols, of which Fig. 2 is a part, can exhibit both oxidant and AO properties [24]. Paracetamol (Figure 3) has been implicated with autism in various reports [62]. OS may be involved in the mechanism, possibly by means of the quinoneimine metabolite (Figure 4) [9]. The AO vitamin D appears to play a role. Supplementation increased total plasma AO capacity. Activities of SOD, GSH reductase and GSH peroxidase are associated with the serum vitamin. Levels of the vitamin are lower with the affected children. Precoporphyrin, a specific indicator of metal toxicity, was also elevated in autistic disorder. Treatment with dimercaptosuccinic acid, an AO, led to a significant drop in urinary porphyrin excretion [63]. Other insults that enhance OS are involved, such as toxins, fever, infection and inflammation.
An ecological study provides evidence for involvement of environmental factors [64]. CNS pathology, including OS, neuro inflammation and mitochondrial dysfunction, suggests involvement of environmental factors, e.g., pollution [65]. Biochemical abnormalities, such as GSH imbalance, may play a role. A report indicates that autism and Parkinson's disease display commonality in OS from toxins [66]. There may be difficulty in handling ROS. AO therapy may be beneficial. Developmental neuro toxicants are shown to play a role in autism [67]. Examples are manganese (Mn) fluoride, chlorpyrifos and brominated diphenyl ethers. Genetic factors appear to account for less than 30-40% of neuro developmental disorders.
Therapy with dimercapto succinic acid is fairly effective and safe in counteracting the toxicity of several toxic metals, particularly lead [68]. There is evidence that hyperbaric oxygen can be beneficial in autism [69]. Data reveal that OS is reduced by this treatment through the up regulation of AO enzymes. Also, there is increase in the formation of mitochondrial enzymes that assist in detoxification. A report describes autism as an epidemic and advances the hypothesis that toxicity and OS may contribute to neuronal insult [70]. There is possible involvement of GSH, a common in vivo AO which combats unwanted OS.
Reviews puts focus on the many ways that OS may play a role in autism [44,71]. Lipid per oxidation is involved with increase in OS. Increased inflammation, excitotoxicity, as well as mitochondrial and immune dysfunction, appear to play a role. There are various reports dealing with the favorable effects of AOs in reversing OS. Concentrations of major AO serum proteins are decreased along with alterations in activities of AO enzymes, such as GSH and homocysteine. A study revealed changes in nitric oxide levels and antioxidant enzyme activities may have a role in the patho physiological mechanisms involved in autism [26,72]. A mechanism links OS with membrane lipid alteration, inflammation, altered immune response, excitotoxicity and impaired energy metabolism. Isoprostanes, which is a product of lipid per oxidation, is enhanced in autism [73]. In addition to OS, endothelial activation could play a role in autism.
Genetics and associated factors
Evidence indicates that about 25% of autistic children suffer from genetic influences [74]. The review identified the genes involved in autism and addressed causal theories. Autism is believed to be one of the most heritable of mental disorders [75]. Studies, which have been faulted, estimate the figure to be more than 90%. Various neuro psychotic disorders appear to posses common genetics. A review describes autism as a neurological disorder with pronounced genetic input, but with appreciable environmental involvement [76]. Evidence indicated that rates of the illness were markedly associated with congenital malformations of the male reproductive system [77]. A study addresses the role of co morbidity in autism [78]. The underlying illness mechanism is probably polygenic and perhaps epistatic with interaction of genetics and environmental factors [79]. A report suggests that autism is not a single disorder, but has multiple characters [80]. Brain developmental syndrome may play a role.
Autism is believed to be the most heritable of neuro developmental disorders [81]. A study with twin pairs showed moderate genetic heritability together with a substantial shared environmental component. A conclusion is that genetic factors involved with autism susceptibility have been overestimated. An article is titled "Searching for ways out of the autism maze" [82]. The ways discussed include genetic, epigenetic and environmental involving complex patho-genetic pathways. A related report addresses both genetic and environmental factors [83]. Animal models are described that occur following insertion of different autism related genes. Both genes and the environment can alter the structure of the developing brain in different ways. A research study deals with common genetic variants, acting additively, as a major factor for autism risk [84]. A myriad of genetic variants of small effects input autism liability. There is an overview of genetics, including causes, therapy, and treatment [85]. Heritability is believed to comprise at least 80% for autism, bipolar disorders and schizophrenia, similar to diabetes, but more than for breast cancer or Parkinson's disease [86]. A review deals with phenotypes of autistic disorders within the group of autism spectrum disorders [87]. Emphasis is on whole genome screens. Another review discussed the contribution of epigenetic to the understanding of genetic factors in autism [88]. Epigenetic refers to changes that alter expression of genes without changing DNA sequence, and considers the role of environmental contributions. Evidence is presented for epigenetic dysregulation in autism. An investigation deals with familial risks of autism [89]. In Sweden, the risk of autism increases with increasing genetic relatedness. Heritability of the disorder appears to be about 50%. A 2014 article suggests from a large study in Sweden that about half of the risk for autism comes from genetics and the other half from environmental factors [90]. Spontaneous mutations appear to play an important role in autism risk. A mathematical model in California attributed 38% of risk for autism to genetic and 58% to the environment. Another analysis suggested that up to 90% of autism is genetic rather than environmental. Underlying mechanisms are addressed concerning genetics in association with environmental factors [91]. The role of epigenetic mechanisms is discussed. A redox/ methylation hypothesis was advanced to rationalize the cause of autism based on the combination of genetic and environmental factors [92]. Autistic children exhibit evidence of OS, including a relation to methylation. A unique membrane signaling process is impaired in the condition. Genetic polymorphism occurs more frequently in autistic children. OS, initiated by environmental factors, leads to neurological deficits. A 2015 investigation finds that genetic aspects provide substantial impact in etiology of autism [93].
Vaccine
There has been recent attention concerning autism and vaccine [94,95]. In 1998, a report suggested a link between autism and vaccine. Vaccination rates dropped because parents were concerned about a possible connection between autism and vaccine. Soon thereafter studies were reported refuting the link [96].
Cell signaling
Various excitations can complicate pathological problems by excitotoxicity and microglial priming [97]. Also, there are effects on cell signaling that can influence neurodevelopment and neuronal function. We suggest that ET can play a role, as for metal toxins. A review presents molecular processes that have been implicated in the illness [98]. Among the various factors is cell signaling. Cell adhesion molecule (CAM) pathway genes are associated with autism [99]. The CAM pathway is important for normal cell signaling. Biological components are part of an integrated network that permeates all aspects from gene regulation to cell signaling and neuronal activity [100]. The concept has been applied to various illnesses including autism. An epidemiologic investigation was performed on genetic and environmental factors concerned with autism [101]. Cell signaling was among the various contributing items. The expression abnormalities of genes in autism were evaluated and their roles in cell signaling events are addressed [102].
Other factors
The effect of fetal stem cell transplantation on autism was investigated [103]. No adverse effects were found in the treated children. Statistically significant favorable differences were noted. The results may be of therapeutic value. Important brain function in human can be attributed to polyunsaturated fatty acids of which docosohexaenoic acid (DHA) has particular importance [104]. Many developmental disorders, such as autism, are causally related to lower levels of DHA. We believe that extended conjugation in DHA may play a part in ET and ROS involvement.
Acknowledgment
Editorial assistance by Thelma Chavez is acknowledged, as well as literature searchers by Darlene Nowak and Linda Muroi.
References
- Kovacic P, Becvar LE (2000) Mode of action of anti-infective agents: focus on oxidative stress and electron transfer. Curr Pharmaceut Des 6(2): 143-167.
- Kovacic P, Osuna JA (2000) Mechanisms of anti-cancer agnets: emphasis on oxidative stress and electron transfer. Curr Pharmaceut Des 6(3): 277-309.
- Kovacic P, Jacintho JD (2001) Mechanism of carcinogenesis. Focus on oxidative stress and electron transfer. Curr Med Chem 8(7): 773-796.
- Kovacic P, Jacintho JD (2001) Reproductive toxins. Pervasive theme of oxidative stress and electron transfer. Curr Med Chem 8(7): 863-892.
- Kovacic P, Sacman A, Wu-Weis M (2002) Nephrotoxins: Widespread role of oxidative stress and electron transfer. Curr Med Chem 9(8): 823-847.
- Poli G, Cheeseman KH, Dianzani MU, Slater TF (1989) Free Radicals in the Pathogenesis of Liver Injury. Pergamon New York, USA, pp. 1-330.
- Kovacic P, Thurn LA (2005) Cardiovascular toxicity from the perspective of oxidative stress, electron transfer, and prevention by antioxidants. Curr Vasc Pharmacol 3(2): 107-117.
- Kovacic P, Pozos RS, Somanathan R, Shangari R, O'Brien PJ (2005) Mechanism of mitochondrial upcouplers, inhibitors, and toxins: Focus on electron transfer, free radicals, and structure-activity relationships. Curr Med Chem 12(22): 2601-2623.
- Kovacic P, Cooksy AL (2005) Unifying mechanism for toxicity and addiction of abused drug and electron transfer and reactive oxygen species. Med Hypotheses 64(2): 357-366.
- Kovacic P, Somanathan R (2009) In: Whitacre DE (Ed.), Rev Environ Contam Toxicol, Springer, New York, USA, pp. 41-69.
- Kovacic P, Somanathan R (2008) Ototoxicity and noise trauma: Electron transfer, reactive oxygen species, cell signaling, electrical effects, and protection by antioxidants: Practical medical aspects. Med Hypotheses 70(5): 914-923.
- Halliwell B, Gutteridge JMC (1999) Free Radicals in Biology and Medicine. Oxford University Press, New York, USA, p. 1-897.
- Kovacic P, Somanathan R (2010) Mechanism of conjugated imine and iminium species, including marine alkaloids: electron transfer, reactive oxygen species, therapeutics and toxicity. Curr Bioact Compds 6: 4659.
- Kalkbrenner AE, Windham GC, Serre MC, Akita Y, Wang X et al. (2015) Particulate matter exposure, prenatal and postnatal windows of susceptibility, and autism spectrum disorders. Epidemiology 26(1): 30-42.
- Volk HE, Lurmann F, Penjold B, Hertz-Picciotto I, McConnell R (2013) Traffic-related air pollution, particulate matter and autism. JAMA Psychiatry 70(1): 71-77.
- Windham GC, Zhang L, Gunier R, Croen LA, Grether JK (2006) Autism spectrum disorders in relation to distribution of hazardous air pollutants in the San Framcisco Bay area. Environ Health Perspect 114(9): 1438-1444.
- Nevison CD (2014) A comparison of temporal trends in United Staes autism prevalence to trends in suspected environmental factors. Environ Health 13: 73.
- Roberts AL, Lyall K, Hart JE, Laden JE, Just AC, et al. (2013) Perinatal air pollutant exposure and autism spectrum disorders in the children of nurses health study II participants. Environ Health Perspect 121(8): 978-984.
- Kovacic P, Somanathan R (2014) Nitro aromatic compounds: environmental toxicity, carcinogenicity, mutagenicity, therapy and mechanism. J Appl Toxicol 34(8): 810-824.
- Rossignol DA, Frye RE (2012) A review of research trends in physiological abnormalities in autism spectrum disorders: immune dysregulation, inflammation, oxidative stress, mitochondrial dysfunction and environmental toxicant exposures. Mol Psychiatry 17(4): 389-401.
- Yao Y, Walsh WJ, McGinnis WR, Pratico D (2006) Altered vascular phenotype in autism: correlation with oxidative stress. Arch Neurol 63(8): 1161-1164.
- Kaur K, Chauhan V, Gu F, Chauhan A (2014) Bisphenol A induces oxidative stress and mitochondrial dysfunction in lymphoblasts from children with autism and unaffected siblings. Free Radic Biol Med 76:25-33.
- Kovacic P, Somanathan R, Abadjian MCZ (2015) Natural monophenols as therapeutics, antioxidants and toxins; electron transfer, radicals and oxidative stress. Nat Prod J in press.
- Sajdel-Sulkowska EM, Xu M, McGinninis W, Koibuchi N (2011) Brain region-specific changes in oxidative stress and neurotrophin levels in autism spectrum disorders (ASD). Cerebellum 10(1): 43-48.
- Al-Ayadhi LY, Elamin NE (2013) Camel milk as a potential therapy as antioxidant in autism spectrum disorder (ASD). Evid Based Complement Alteranat Med 2013: 602834.
- Söğüt S, Zorogurlu F, Sivasli E, Yetkin O, Yanik M, et al. (2003) Changes in nitric oxide levels and antioxidant enzyme activities may have a role in the pathophysiological mechanisms involved in autism. Clin Chim Acta 33(1-2): 111-117.
- McGinnis WR (2007) Could oxidative stress from psychosocial stress affect neurodevelopment in autism? J Autism Dev Disord 37(5): 993994.
- Zhang Y, Sun Y, Wang Z, Peng Y, Li R (2012) Down regulating the canonical Wnt/p-catenin signaling pathway attenuates the susceptibility to autism-like phenotypes by decreasing oxidative stress. Neurochem Res 37(7): 1409-1419.
- Meguid NA, Dardir AA, Abdel-Raouf ER, Hashish A (2011) Evaluation of oxidative stress in autism: defective antioxidant enzymes and increased lipid per oxidation. Biol Trace Elem Res 143(1): 58-65.
- Rossignol DA, Frye DA (2014) Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism. Front Physiol 5: 150.
- Napoli E, Wong S, Giulivi C (2013) Evidence of reactive oxygen species- mediated damage to mitochondrial DNA in children with typical autism. Mol Autism 4(1): 2.
- Kern JK, Jones AM (2006) Evidence of toxicity, oxidative stress, and neuronal insult in autism. J Toxicol Environ Health B Crit Rev 9(6): 485-499.
- Ghanizadeh A, Akhondzadeh S, Hormozi M, Makarem A, Abotorabi- Zarchi M, et al. (2012) Glutathione-related factors and oxidative stress in autism, a review. Curr Med Chem 19(23): 4000-4005.
- Ming X, Johnson WG, Stenroos ES, Mars A, Lambert GH, et al. (2010) Genetic variant of glutathione peroxidase 1 in autism. Brain Dev 32(2): 105-109.
- Gebril OH, Meguid NA (2011) HFE gene polymorphisms and risk for autism in Egyptian children and impact on the effect of oxidative stress. Dis Markers 31(5): 289-294.
- Heberling CA, Dhurjati PS, Sasser M (2013) Hypothesis for a systems connectivity model of autism spectrum disorder pathogenesis: links to gut bacteria, oxidative stress, and intestinal permeability. Med Hypotheses 80(3): 264-270.
- Sajdel-Sulkowska EM, Xu M, Koibuchi N (2009) Increase in cerebellar neurotrophin-3 and oxidative stress markers in autism. Cerebellum 8(3): 366-372.
- Ming X, Stein TP, Brimacombe M, Johnson WG, Wagner GC (2005) Increased excretion of lipid per oxidation biomarker in autism. Prostaglandins Leukot Essent Fatty Acids 73(5): 379-384.
- Zoroglu SS, Armutcu F, Ozen S, Gurel A, Sivasli E, et al. (2004) Increased oxidative stress and altered activities of erythrocyte free radical scavenging enzymes in autism. Eur Arch Psychiatry Clin Neurosci 254(3): 143-147.
- James SJ, Cutler P, Melnyk S, Jernigan S, Janak L, et al. (2004) Metabolic biomarkers of increased oxidative stress and impaired methylation capacity in children with autism. Am J Clin Nutr 80(6): 1611-1617.
- James SJ, Melnyk S, Jernigan S, Cleves MA, Halsted CH, et al. (2006) Metabolic endophenotype and related genotypes are associated with oxidative stress in children with autism. Am J Med Genet B Neuropsychiatr Genet 141B(8): 947-956.
- Ghezzo A, Visconti P, Abruzzo PM, Bolotta A, Ferreri A, et al. (2013) Oxidative stress and erythrocyte membrane alterations in children with autism: correlation with clinical features. PLoS One 8(6): e66414.
- McGinnis WR (2004) Oxidative stress in autism. Altern Ther Health Med 10(6): 22-36.
- Chauhan A, Chauhan V (2006) Oxidative stress in autism. Pathophysology 13(3): 171-181.
- McGinnis WR (2005) Oxidative stress in autism. Altern Ther Health Med 11(1): 19.
- Rossignol DA, Genuis SJ, Frye RE (2014) Environmental toxicants and autism spectrum disorders: a systematic review. Transl Psychiatry 4: e360.
- von Ehrenstein OS, Aralis H, Cockburn M, Ritz B (2014) In utero exposure to toxic air pollutants and risk of chilhood autism. Epidemiology 25(6): 851-858.
- Smaga I, Gawlik M, Moniczewski A, Krzek J, Przegalinski E, et al. (2015) Oxidative stress as an etiological factor and a potential treatment target of psychiatric disorders Part 2 depression, anxiety, schizophrenia and autism. Pharmacol Rep 67(3): 569-580.
- Faber S, Boggess A, Fahrenholz T, Kern JC, Kingston HM (2015) A cleanroom sleeping environment's impact on markers of oxidative stress, immune dysregulation, and behavior in children with autism spectrum disorders. BMC Complement Altern Med 15: 71.
- Han Y, Dai W, Yang SH, Gao L, Su YY, et al. (2015) Abnormal trans sulfuration metabolism and reduced antioxidant capacity in Chinese children with autism spectrum disorders. Int J Dev Neurosci 46: 27-32.
- Porokhovnik LN, Passekov VP, Gorbachevskaya NL, Sorokin AB, Veiko NN, et al. (2015) Active ribosomal genes, trasnslational homeostasis and oxidative stress in the pathogenesis of schizophrenia and autism. Psychiatr Genet 25(2): 79-87.
- Niufar NN, Haycock FL, Wesemann JL, MacStay HVL, Kovacic P (2002) Reduction potentials of conjugated aliphatic ketones, oximes and imines: correlation with structure and bioactivity. Rev Soc Quim 46: 307-312.
- Rose S, Fyre RE, Slattery J, Wynne R, Tippett M, et al. (2014) Oxidative stress induces mitochondrial dysfunction in a subset of autism lymphoblastoid cell lines in a wall-matched case control cohort. PloS One. 9(1): e85436.
- Yassa HA (2014) Autism: a form of lead and mercury toxicity. Environ Toxicol Pharmacol 38: 1016-1024.
- Alabdali A, Al-Ayadhi L, El-Ansary A (2014) A key role for an impaired detoxification mechansim in the etiology and severity of autism spectrum disorders. Behav. Behav Brain Funct 10: 14.
- Geier DA, King PG, Sykes LK, Geier MR (2008) A comprehensive review of mercury provoked autism. Indian J Med Res 128(4): 383-411.
- Adams JB, Baral M, Geis E, Mitchell J, Ingram J, et al. (2009) The severity of autism is associated with toxic metal body burden and red blood cell glutathione levels. Journal of Toxicology doi:10.1155/2009/532640.
- Adams JB, Audhya T, McDonough-Means S, Rubin RA, Quig D, et al. (2013) Toxicological status of children with autism vs. neurotypical children and the association with autism severity. Biol Trace Elem Res 15(12): 171-180.
- Geier DA, Kern JK, King PG, Sykes LK, Geier MR (2012) Hair toxic metal concentrations and autism spectrum disorder severity in young children. Int J Environ Res Public Health 9(12): 4486-4497.
- Blaurock BE, Amin OR, Dessoki HH (2012) Toxic metals and essential elements in hair and severity of symptoms among children with autism. Maedica (Buchar) 7(1): 38-48.
- Hegazy HG, Ali EH, Elgoly AH (2015) Interplay between pro- inflammatory cytokins and brain oxidative stress biomarkers: evidence of parallels between butyl paraben intoxication and the valproic acid brain physiopathology in autism rat model. Cytokine 71: 173-180.
- Cannell JJ (2014) Paracetamol, oxidative stress, vitamin D and autism spectrum disorders. Int J Epidemiol 43(3): 974-975.
- Nataf R, Skorupka C, Amet L, Lam A, Springbett A, et al. (2006) Porphyrinuria in childhood autistic disorder:implications for environmental toxicity. Toxicol Appl Pharmacol 214(2): 99-108.
- St Hilaire S, Ezike VO, Stryhn H, Thomas MA (2012) An ecological study on childhood autism. Int J Health Geogr 11: 44.
- Herbert MR (2010) Contributions of the environment and environmentally vulnerable physiology to autismspectrum disorders. Curr Curr Opin Neurol 23(2): 103-110.
- Yochum CL, Wagner GC (2009) Autism and Parkinson's disease: animal models and a common etiology ical mechanism. Chinese J Physiol 52: 236-249.
- Grandjean P, Landrigan PJ (2014) Neurobeahavoural effects on developmental toxicity. Lancet Neurol 13: 330-338.
- Adams JB, Baral M, Geis E, Mitchell J, Ingram J, et al. (2009) Safety and efficacy of oral DMSA therapy for children with autism spectrum disorders: part A-medical results. BMC Clin Pharmacol 9: 16.
- Rossignol DA (2007) Hyperbaric oxygen therapy might improve certain pathophysiological findings in autism. Med Med Hypotheses 68(6): 1208-1227.
- Kern JK, Jones AM (2006) Evidence of toxicity, oxidative stress, and neuronal insult in autism. J Toxicol Environ Health B Crit Rev 9(6): 485-499.
- Damodaran LP, Arumugam G (2011) Urinary oxidative stress markers in children with autism. Redox Rep 16(5): 216-222.
- Denis PA (2014) Could the novel paradigm for NO (nitric oxide) be involved in Autism? J Psychol Abnorm Child doi.org/10.4172/2329- 9525.1000112.
- Yao Y, Walsh WJ, Mc Ginnis WR, Pratico D (2006) Altered vascular phenotype in autism. Arch Neurol 63(8): 1161-1164.
- Miles JH (2011) Autism spectrum disorders-a genetic review. Genet Med 13(4): 178-294.
- Lichtenstein P, Carlstrom E, Rastam M, Gillberg C, Anckarsater H (2010) The genetics of autism spectrum disorders and related neuropsychiatric disorders in childhood. Am J Psychiatry 167(11): 1357-1363.
- Herbert MR, Russo JP, Yang S, Roohi J, Blaxill M, et al. (2006) Autism and environmental genomics. Neurotoxicology 27(5): 671-684.
- Rzhetsky A, Bagley SC, Wang K, Lyttle CS, Cook EH, et al. (2014) Environmental and state-level regulatory factors affect the incidence of autism and intellectual disability. PLoS Comput Biol 10(3): e1003518.
- Polyak A, Kubina RM, Girirajan S (2015) Comorbidity of intellectual disability confounds ascertainment of autism: implications for genetic diagnosis. Am J Med Genet B Neuropsychiatr Gene 168(7): 600-608.
- Garener H, Spiegelman D, Buka SL (2009) Prenatal risk factors for autism: comprehensive meta-analysis. Br J Psychiatry 195(1): 7-14.
- Geschwind DH, Levitt P (2007) Autism spectrum disorders: developmental disconnection syndromes. Curr Opin Neurobiol 17(1): 103-111.
- Hallmayer J, Cleveland S, Torres A, Phillips J, Cohen B, et al. (2011) Genetic heritability and shared environmental factors among twin pairs with autism. Arch Gen Psychiatry 68(11): 1095-1102.
- Persico AM, Bourgeron T (2006) Searching for ways out of the autism maze: genetic, epigenetic and environmental clues. Trends Neurosci 29(7): 349-358.
- Gadad BS, Hewitson L, Young KA, German DC (2013) Neuropathology and animal models of autism: genetic and environmental factors. Autism Research and Treatment 2013 (2013): 2.
- Klei L, Sanders SJ, Murtha MT, Hus V, Lowe JK, et al. (2012) Common genetic variants, acting additively, are a major source of risk for autism. Mol Autism 3(1): 9.
- Marchant GE, Robert JS (2009) Genetic testing for autism predisposition: ethical, legal and social challenges. Houston J Health Law Policy 9: 203-235.
- Carroll LS, Owen MJ (2009) Genetic overlap between autism, schizophrenia and bipolar disorders. Genome Med 1(10): 102.
- Klauck SM (2006) Genetics of autism spectrum disorder. Eur J Human Gen 14: 714-720.
- Hall L, Kelley E (2014) The contribution of epigenetics to understanding genetic factors in autism. Autism 18(8): 872-881.
- Sandin S, Lichtenstein P, Kuja HR, Larsson H, Hultman CM, et al. (2014) The familal risk of autism JAMA 311(17): 1770-1777.
- Geggel L (2014) Environment, genetics may contribute equally to autism risk. Simons Found Autism Res Initiative.
- Tordjman S, Somogyi E, Coulon N, Kermarrec S, Cohen D, et al. (2014) Gene, environment interactions in autism spectrum disorders: role of epigenetic mechanisms. Front Psychiatry 5: 53.
- Deth R, Muratore C, Benzecry J, Power CVA, Waly M (2008) How environmental and gentic factors combine to cause autism: a redox/ methylation hypothesis. Neurotoxicology 29(1): 190-201.
- Wise J (2015) Genetic factors are "substantial” in aetiology of autism, study of UK twins finds. BMJ 35: h1212.
- DeStefano F, Chen RT (1999) Negative association between MMR and Autism. Lancet 353(9169): 1987-1988.
- Taylor B, Miller E, Farrington CP, Petropoulos MC, Favot MI, et al. (1999) Autism and measles, mumps, and rubella vaccine: ni epidemiologic evidence for a casual association. Lancet 353(9169): 2026-2019.?
- Sathyanarayana Rao TS, Andrade C (2011) The MMR vaccine and autism: sensation, refutation, retraction, and fraud. Indian J Psychiatry 53(2): 95-96.
- Blaylock RL, Strunecka A (2009) Immune-glutamatergic dysfunction as a central mechanism of the autism spectrum disorders. Curr Med Chem 16(2) 157-170.
- Pescosolido MF, Yang U, Sabbagh M, Morrow EM (2012) Lighting a path: genetic Studies pinpoint neurodevelopmental mechanisms in autism and related disorders. Dialogues Clin Neurosci 14(3): 239-252.
- O'Dushlaine C, Kenny E, Heron E, Donohoe G, Gill M, et al. (2011) Molecular pathways involved in neuronal cell adhesion and membrane scaffolding contribute to schizophrenia and bipolar disorder susceptibility. Mol Psychiatry 16(3): 286-292.
- Broderick G, Craddock TJ (2013) Systems biology of complex symptom profiles: capturing interactivity across behavior, brain and immune regulation. Brain Behav Immun 29: 1-8.
- Hertz PI, Croen LA, Hansen R, Jones CR, Van de Water J, et al. (2006) The CHARGE study: an epidemiologic investigation factors of genetic and environmental factors contributing to autism. Environ Health Perspect 114(7): 1119-1125.
- Zhang JY, Middleton FA, Sagvolden T, Faraone SV (2012) Differential expression of SLC9A9 and interacting molecules in the hippocampus of rat models for attention deficit/hyperactivity disorders. Dev Neurosci 34(2-3): 218-227.
- Bradstreet JJ, Sych N, Antonucci N, Ivakova O, Matyashchuk I, et al.(2014) Efficacy of fetal stem cell transplantation in autism spectrum disorders: an open-labeled pilot study. Cell Transplant 23(Suppl 1): S105-S112.
- Gharami K, Raja SC, Das M, Das S (2015) Essential role of docosahexaenoic acid towards development of a smart brain. Neurochem Int 89: 51-62.






























