Appraisal of In-Vitro Antimicrobial, Thrombolytic and Membrane Stabilizing Potentials of Chloroform Extract of Avicennia Marina Leaves and Barks: An Approach to Develop Novel Drug
Shalahuddin M*, Saddam H, Mohammad Nurul Amin, Ibrahim Md, and Farhana S
Department of pharmacy, Atish Dipankar University of Science and Technology, Bangladesh.
Submission:May 09, 2017; Published: May 30, 2017
*Corresponding author: Md. Shalahuddin Millat, Lecturer, Department of pharmacy, Atish Dipankar University of Science and Technology, Banani, Dhaka-1213, Bangladesh Tel:+8801722598057, Email: millat404pharma@gmail.com
How to cite this article:Md. Shalahuddin M, Md. S Hussain, M N Amin, Md. Ibrahim, F Sultana. Appraisal of In-Vitro Antimicrobial, Thrombolytic and Membrane Stabilizing Potentials of Chloroform Extract of Avicennia Marina Leaves and Barks: An Approach to Develop Novel Drug. Nov Appro Drug Des Dev. 2017; 1(2): 555560. DOI: 10.19080/NAPDD.2017.01.555560
Abstract
The present study was aimed to assess ethno medicinal value of the plant. We tried to oversee in-vitro antimicrobial, thrombolytic and membrane stabilizing potentials of chloroform extracts of Avicennia marina leaves and barks. A. marina is an ethno medicinal plant belongs to the family of Acanthaceae. Crude chloroform extracts of A. marina (leaves & barks) dose dependently used for in vitro antimicrobial, thrombolytic and membrane stability screening. Chloroform extracts of A. marina at higher concentration evolved moderate antimicrobial activities against Gram negative (-ve) bacteria and were resistant against Gram positive (+ve) bacteria during assessment through disc diffusion method. During analysis of thrombolytic activity, 10mg/ml concentration of the extracts most significantly showed 16.33±0.49% lysis of clot (p<0.001), where streptokinase was used as standard & distilled water was treated as negative control. Again, Acetyl salicylic acid was used as standard in membrane stabilizing study whereas chloroform extracts of A.marina leaves and barks dose dependently showed inhibition of hemolysisat 10mg/ml, which was found 18.84±0.99% and 26.24±1.04% respectively for both hypotonic solution and heat induced condition of hemolysis of erythrocyte membrane.
Keywords: A. marina; Antimicrobial; Thrombolytic; Membrane stabilizing
Introduction
To cure all malady of mankind, nature always acts as a great source of succor by providing different redress from its plants, animals, and other sources. Different substances containing medicinal value are comprised from many species of plant kingdom, which are yet to be examined. A large number of plants are constantly being veiled due to the presence of their possible medicinal value [1]. Allowable means of health care for poor and marginalized peoples are provided by medicinal plants. Different parts such as; leaves, stems, barks, flowers and underground parts of medicinal plants are most often used for traditional medicines. Traditional medical practitioners are using medicinal plants for disease-treating formulations and treatment from ancient time [2]. Major components of indigenous medicines are constructed from medicinal plants. Bacterial transits including; respiratory transits, gastrointestinal transits, skin transits, urinary tract & surgical wound transits causing mild to life threatening illnesses. Due to promiscuous use of antibiotics, the advancing of resistance of pathogens against antibiotics has become a difficult consequence in the recent years [3-8]. In developed countries, the main causes of morbidity and mortality are deep vein thrombosis, pulmonary emboli, strokes and heart attacks etc. [9]. Different thrombolytic agents are used as thrombolytic therapy to dissolve clots including; streptokinase, alteplase, anistreplase, urokinase, and tissue plasminogen activator (TPA). Inflammation, one of the most important paranormal illness and a freaky biological reaction of vascular tissues to exaggerated stimuli. In response to bodily injury, inflammation occurs as a part of non-specific immune response [10,11]. Medicinal plants play a vital role in the treatment of several diseases. Avicennia marinaisan an ethno medicinal plant, commonly known as grey mangrove or white mangrove which is available in Bangladesh, Indonesia, Malaysia, Africa, Brazil, and Central America. The present study was designed to explore in vitro antimicrobial, thrombolytic &membrane stabilizing potentials of chloroform extracts of A. marina leaves and barks.
Materials and Methods
Collection of plant materials
The leaves and barks of A.marina were accumulated during November, 2016 from Sonadia Deep, CoxsBazar, Bangladesh. A.marina is locally known as Tella mada or Moricha baen. Its accession number was confirmed by the taxonomist of National Herbarium Institute, Mirpur, Dhaka, Bangladesh, which is- 38311.
Extraction
The collected leaves and barks of A. marina were properly washed with water. Later they were chopped, dried, and powdered. About 500g of the powdered materials were soaked in 1.5 litre of chloroform at room temperature for two weeks. Then the solution was filtered using filter cloth and Whatman’s filter paper and concentrated with a rotary evaporator. It rendered a brown granular. The brown granular was designated as crude chloroform extract.
Antimicrobial activity
Antibacterial screening was determined in the presence of both gram positive (+ve) & gram negative (-ve) bacteria. The experimental bacterial strains were collected as pure cultures from the National Institute of Biotechnology, Savar, Dhaka, Bangladesh. Kirby-Bauer’s Disc diffusion method with minute modifications was used to determine the antibacterial activities of chloroform extracts of A. marina leaves and barks [12].
Thrombolytic activity
Standard drug streptokinase (SK): Marketed obtainable lyophilized Altepase (Streptokinase) vial (Bea¬con pharmaceutical Ltd.) of 15,00,000 I.U., was aggregated and 5ml sterile distilled water was mixed properly. This suspension was used as a standard from which 100μl (30,000 I.U) was used for in vitro clot lysis assay method described by Prasad et al. [13].
Preparation of test sample: The plant extracts were dissolved in chloroform and shaken vigorously on a vortex mixer to prepare different concentration (2, 4, 6, 8 and 10mg/ml respectively) of the test sample. The suspension was kept overnight and decanted to remove the soluble supernatant, which was filtered through a 0.22 micron syringe filter. Thus test samples were prepared for thrombolytic screening.
Thrombolytic potential analysis
Thrombolytic potentials of chloroform extract of A. marina leaves and barks was carried out by the method of Prasad et al. [13]. 5ml of venous blood were drawn from healthy volunteers without a history of oral contraceptive or anticoagulant thera¬py, which were distributed in five different pre weighed sterile micro cen¬trifuge tubes (0.5ml/tube) and incubated at 37°C for 45 minutes. After clot formation, the serum was completely removed without disturbing the clot and each tube having clot was again weighed to determine the clot weight (clot weight = weight of clot containing tube - weight of tube alone). Each micro-centrifuge tube containing clot was properly labeled and 100μl of the test samples from each doses (2, 4, 6, 8 and 10mg/ml respectively) were added to the tubes accordingly [13]. As a positive control, 100μl of streptokinase (SK) and as a negative control, 100μl of distilled water were separately added to the control tubes. All the tubes were then incubated at 37°C for 90 minutes and observed for clot lysis. After incubation, the fluid was removed and tubes were again weighed to observe the difference in weight after clot disruption. Difference obtained in weight taken before and after clot lysis was expressed as percentage of clot lysis as shown below:
% of clot lysis = (wt. of released clot / wt. of clot before treatment) × 100
Membrane stabilizing activity
In vitro membrane stabilizing activity of the extractives was performed by using hypotonic solution and heat-induced haemolysis of erythrocyte membrane by the method developed by Shinde et al. [14] and modified by Sikder et al. [15].
Statistical analysis
The present data were represented as Mean±SD, while one way analysis of variance (ANOVA) followed by Dunnett ‘t’ test, using SPSS software of 10 version, was considered for analyzing the significance level of calculated values (P<0.05).
Results
Antimicrobial activity
The antibacterial effects of crude chloroform extracts of A. marina leaves and barks have been imparted in Table 1. Chloroform extracts of A. marina leaves and barks displayed moderate activity against gram negative (-ve) bacteria and were resistant against gram positive (+ve) bacteria.
Here, (+++)=highly active; (++)=moderately active; (+)=slightly active; (-)=No activity against microorganism. ***= P<0.001, **=P<0.01, *=P<0.05 (one way ANOVA tests followed by Dennett’s t-test on each group P<0.05 were considered statistically significant).
Thrombolytic activity
The effects of chloroform extracts of A. marina leaves and barks on in-vitro clot lysis are tabulated in Table 2. From Table 2, it is pronounced that, when 100μl of streptokinase (30,000 I.U.) was used as a positive control, the percentage of clot lysis was 45.49% whereas, water (negative control) exhibited negligible clot lysis (6.03%). The percentage of clot lysis was dose dependently increased, whereas10mg/ml concentration most significantly showed 13.22% lysis of clot (p< 0.001).
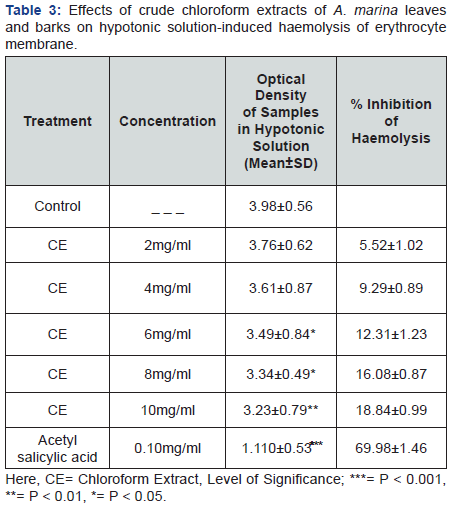
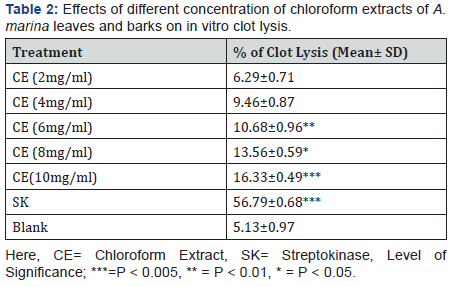
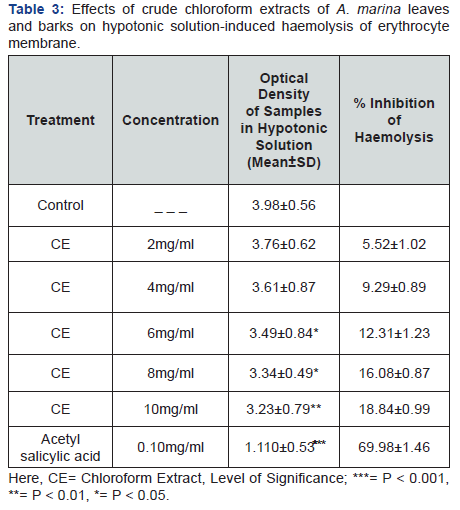
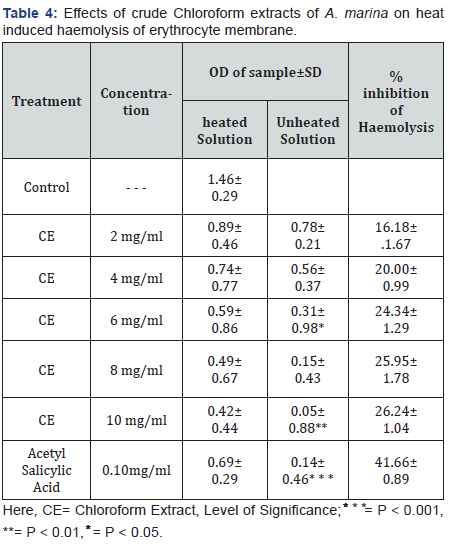
Membrane stabilizing activity
Crude chloroform extracts of A. marina leaves and barks dose dependently increased in membrane stabilizing potential which are represented in Table 3 & 4, whereas 10mg/ml concentration more significantly showed 10.11% & 27.76% inhibition of haemolysis respectively by hypotonic solution and heat induced haemolysis of erythrocyte membrane. Acetyl salicylic acid was used as standard in membrane stabilization. ASA (0.10mg/mL) revealed 69.98%& 71.97% inhibition of haemolysis respectively induced by hypotonic solution and heat induced haemolysis correspondingly.
Discussion
Recent strategies for discovering novel drugs from unexplored natural resources recommended marine plants as an important source of potentially useful chemicals [16]. Phyto chemical screening and determination of biological activities are essential steps for exploration. Employing a wide range of bioassays [17] is valuable not only for preliminary biological characterization of new compounds, but also an essential guidance of the chemical isolation procedures [18]. Antibacterial agents are showing their activity against microorganisms by inhibiting cell wall synthesis [19,20], they accumulate in plasma membrane of bacterial and causing depletion of energy [21]. Through interfering in the permeability of cell membrane, antibacterial agents changes the structure and function of key cellular constituents, resulting in mutation, cell damage, and finally leads to death [22]. The medicinal properties of the Avicennia marina lie in a several chemical group such as tannins, flavonoids, glycosides carbohydrates, organic acids and terpenes [23]. Many parts of the Avicennia marina plant especially leaves and barks possess antibacterial properties due to presence of tannins and flavonoids [24,25]. This Plant also synthesize huge amount of aromatic compound among which phenols or their oxygen substituted derivatives are predominant [26]. These compounds provide protection against microbes for the plant [27]. This is great to Avicennia marina plant extract showed to have phyto chemicals responsible for antibacterial effects [28]. May be that is why the crude chloroform extracts of Avicennia marina surprisingly showed significant antibacterial activity against Gram negative (-ve) bacteria. Very recently phyto pharmacological investigation able to create a new field to discover plant derivative drugs and renew the attention in herbal medicines, where 30% of the pharmaceuticals are prepared from plants derivatives [29] Some severe outcomes such as stroke and myocardial in ¬fraction manifested due to the failure of hemostasis and consequent for¬mation of blood clots in the circulatory system. Fibrinolytic agents such as urokinase, tissue plasminogen activator and streptokinase used for clinical intervention for pathological development of blood clots. Many research works have been undertaken to discover antithrombotic (anti¬coagulant and anti platelet) effect of plants and natural food sources in order to prevention of coronary events and stroke [30]. In the present study chloroform extract of Avicennia marina showed significant thrombolytic activity, this effect may be possible due to phyto constituents such as tannin, alkaloid, and saponin present in the plant extracts affecting acti-vation of plasminogen both by fibrin-dependent and fibrinindependent mechanisms similar to Streptokinase which causes extra production of plasmin which breaks down fibrin the major constituent of thrombi, to dissolve unwanted blood clots. Several studies support our present find¬ings [31]. Present study evidenced that the chloroform extract of the plant dosedependently protects the human erythrocyte membrane against lysis induced by hypotonic solution and heat induced condition. During inflammation, phagocytes release many lysosomal enzymes and hydrolytic components to the extracellular space, which assists a variety of disorders by inducing damages of the surrounding organelles and tissues [32]. Studies evidence that non-steroidal anti-inflammatory drugs act through stabilization of lysosomal membranes by inhibiting these lysosomal enzymes. Again, lysis of the RBC membranes accompanied by the oxidation when exposed to harmful substances such as hypotonic medium, heat, etc through lysis of hemoglobin [33]. Thus mechanism of anti-inflammatory activity of the plant extract is assessed by considering their potentials in inhibition of hypotonicity and heat induced RBC membrane lysis, because human RBC membranes are considered similar to lysosomal membrane components [34]. One can also assumed that the possible mode of action of the extract and standard anti-inflammatory drugs may be connected with binding to the erythrocyte membranes through consequent alteration of surface charges of cells. Some research works were able to reveal the name of some responsible chemical components present in the extracts, which are well known for their anti-inflammatory activity [35]. Both in vitro and in vivo studies in experimental animals showed that the flavonoids exert stabilizing effects largely only so somes [36] while tannin and saponins are also capable of stabilizing the erythrocyte membrane with an ability of binding with cations and other bio molecules [37]. Our present research reveals that plant chloroform extracts showed potent RBC membrane stabilization activity with a good protection against both hypotonic solution induced condition may be this due to the presence of phyto constituent like flavonoid
Conclusion
This study corroborated that the crude chloroform extracts of A. marina (leaves & barks) at higher concentration bloomed moderate in-vitro thrombolytic & membrane stabilizing activities. It also manifested middling antimicrobial activities against gram negative (-ve) bacteria and were resistant against gram positive (+ve) bacteria.
Acknowledgement
The authors would like to express their heartfelt gratitude, indebtedness and profound appreciation to all honorable teachers and staffs of the department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh for their cordial support.
References
- Evans WC (1997) Trease and Evan’s Pharmacognosy. (14th edn), W.B. Saunders Company Limited, USA, p. 3.
- Hossan S, Agarwala B, Sarwar S, Karim M, Jahan R, et al. (2010) Traditional Use of Medicinal Plants in Bangladesh to Treat Urinary Tract Infections and Sexually Transmitted Diseases. Ethnobotany Research & Applications 8: 61-74.
- Kunin CM (1993) Resistance to Antimicrobial Drugs- A Worldwide Calanity. Ann Intern Med 118(7): 557-561.
- Kunin CM (1983) Antibiotic resistance - a world health problem we cannot ignore. Ann Intern Med 99(6): 859-860.
- Burke JP, Levy SB (1985) Summary report of worldwide antibiotic resistance: international task forces on antibiotic use. Rev Infect Dis 7(4): 560-564.
- Kunin CM, Lipton HL, Tupasi T, Sacks T, Scheckler WF, et al. (1987) Social, behavioral, and practical factors affecting antibiotic use worldwide: report of Task Force. Rev Infect Dis 9(3): S270-S285.
- Cohen ML (1992) Epidemiology of drug resistance: implications for a post-antimicrobial era. Science 257(5073): 1050-1055.
- Neu HC (1992) The crisis in antibiotic resistance. Science 257(5073): 1064-1073.
- Nicolini FA, Nichols WW, Mehta JL, Saldeen TG, Schofield R, et al. (1992) Sustained reflow in dogs with coronary thrombosis with K2P, a novel mutant of tissue plasminogen activator. J Am Coll Cardiol 20(1): 228-235.
- Perianayagam JB, Sharma SK, Pillai SK (2006) Anti-inflammatory activity of Trichodesma indicum root extract in experimental animals. J Ethnopharmacol 104(3): 410-414.
- Anosike CA, Obidoa O, Ezeanyika LU (2012) Membrane stabilization as a mechanism of the anti-inflammatory activity of methanol extract of garden egg (Solanum aethiopicum). DARU 20(1): 76.
- Bauer AW, Kirby WMM, Sheriss JC, Turck M (1966) Antibiotic susceptibility testing by standardized single method. Amer J clin path 45(4): 493-496.
- Prasad S, Kashyap RS, Deopujari JY, Purohit HJ, Taori GM, et al. (2007) Effect of Fagonia arabica (Dhamasa) on in vitro thrombolysis. BMC Complement Altern Med 7: 36.
- Shinde UA, Phadke AS, Nair AM, Mungantiwar AA, Dikshit VJ, et al. (1999) Membrane stabilizing activity - a possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia 70(3): 251-257.
- Sikder MAA, Kaiser MA, Rashid MA, Millat MS, Sultan A (2012) In vitro membrane stabilizing activity, total phenolic content, cytotoxic, thrombolytic and antimicrobial activities of Calliandra surinamensis (Wall.). J Pharmacog Phytochem 1(3): 45-50.
- Harvey A (2000) Strategies for discovering drugs from previously unexplored natural products. Drug Discov Today 5(7): 294 -300.
- Khafagi I, Abdel KM, Sami E, El GH (1999) Are exogenous antibiotic safe to use in plant tissue culture media? 6th International Conference of Union Arab Biologists 8-11 Nov. 1999. Journal of Union Arab Biologists 7(B) Botany: 249-260.
- Claeson P, Bohlim L (1997) Some aspects of bioassay methods in natural-product research aimed at drug lead discovery. Trends Biotechnol 15(7): 245-248.
- Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12(4): 564-582.
- Marcucci MC, Ferreres F, García VC, Bankova VS, De Castro SL, et al. (2001) Phenolic compounds from Brazilian propolis with pharmacological activities. J Ethnopharmacol 74(4): 105-112.
- Skrinjar MM, Nemet NT (2009) Antimicrobial effects of spices and herbs essential oils. APTEFF 40: 195-209.
- Kim DW, Son KH, Chang HW, Bae K, Kang SS, et al. (2004) Antiinflammatory activity of Sedum kamtschaticum. J Ethnopharmacol 90(2-3): 409-414.
- Khafagi I, Gab AA , Salama W, Fouda M (2003) Biological activities and phytochemical constituents of the gray mangrove Avicennia marina (Forssk.) Vierh. Egyptian Journal of Biology 5: 62-69.
- Chung KT, Wong TY, Wei CI, Huang YW, Lin Y (1998) Tannins and human health: a review. Crit Rev Food Sci Nutr 38(6): 421-464.
- Scalbert A (1991) Antimicrobial properties of tannin. Phyto chemistry 30(12): 3875-3883.
- Geissman TA (1963) Flavonoid compounds, tannins, lignins and related compounds. Elsevier Press, USA.
- Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12(4): 564-582.
- Rahman MA, Hussain MS, Millat MS, Ray MC, Amin MT, et al. (2016) Screenings of In-vitroantimicrobial, cytotoxic and anti-inflammatory activity of crude methanolic extracts of Crinum latifolium (Leaves). J Med Plants Res 10(37): 649-655.
- Ali R, Marjan H, Jannatul FR, Hasanuzzaman M, Islam M (2014) Evaluation of thrombolytic potential of three medicinal plants available in Bangladesh, as a potent source of thrombolytic compounds. Avicenna J Phytomed 4(6): 430-436.
- Hussain MS, Hossain MS, Amin MT, Millat MS (2016) In vitro thrombolytic potentials of methanolic extract of Vigna unguiculata Linn (seed). Journal of Pharmacognosy and Phytochemistry 5(3): 129- 31.
- De P, Chaudhuri D, Tamang S, Chaudhuri TK, Mandal N (2012) In vitro antioxidant and free radical scavenging potential of Clerodendrum viscosum. Int J Pharm Bio Sci 3: 454-471.
- Ackerman NR, Beebe JB (1974) Release of lysosomal enzymes by alveolar mononuclear cells. Nature 247(5441): 475-477.
- Feirrali M, Signormi C, Ciccolili L, Comporti M (1992) Iron release and membrane damage in erythrocytes exposed to oxidizing agents, phenylhydrazine, devicene and iso-uranil. Biochem J 285(pt 1): 295- 301.
- Mounnissamy VM, Kavimani S, Balu V, Drlin QS (2008) Evaluation of anti-inflammatory and membrane stabilizing properties of ethanol extract of Canjera rehedi. Iranian J Pharmacol Therapeut 6: 235-237.
- Vinod RK, Chandrasekhar J, Sudhakar K, Rajeswar T, Sandhya SK, et al. (2010) Membrane stabilizing potency of two Tephrosia species. J Phytol 2: 42-46.
- Van CP (1972) Influence of some hydrosolube substances with vitamin P activity on the fragility of lysosomes in vitro. Biochem Pharmacol 21(11): 1543-1548.
- Khan I, Nisar M, Ebad F, Nadeem S, Saeed M, et al. (2009) Antiinflammatory activities of Sieboldogenin from Smilax china Linn.: experimental and computational studies. J Eethnopharmacol 121(1): 175-177.






























