Seasonal Changes in the Frequency of False-Negative Rapid Diagnostic Tests for Malaria based on Histidine-rich Protein 2 (HRP2)
Ousmane A Koita1*, Lansana Sangaré1, Aliou Sissako1, Vincent Sanogo1, Chacka Coulibaly1, Pamela P Badjogo1, Youssouf Diarra1 and Donald J Krogstad2
1Laboratoire de Biologie Moléculaire Appliquée, Université des Sciences, des Techniques et des Technologies de Bamako, Mali
2Tulane School of Public Health and Tropical Medicine, New Orleans, LA
Submission: September 9, 2023; Published: October 10, 2023
*Corresponding author: Ousmane Koita, Professor, Laboratoire de Biologie Moléculaire Appliquée, Université des Sciences, des Techniques et des Technologies de Bamako, Mali
How to cite this article: Ousmane A K, Lansana S, Aliou S, Vincent S, Chacka C, et al. Seasonal Changes in the Frequency of False-Negative Rapid Diagnostic Tests for Malaria based on Histidine-rich Protein 2 (HRP2). JOJ Pub Health. 2023; 8(2): 555732. DOI: 10.19080/JOJPH.2023.08.555732
Abstract
Aim: Rapid diagnostic tests (RDTs) for Plasmodium falciparum infection provide an essential alternative in areas where skilled microscopists are not available and therefore have a central role in the diagnosis of malaria and assessment of malaria control. The sensitivities of HRP2-based RDTs are reported to be 93-98% with false-negative rates of 5-8%. The study aimed to evaluate the seasonality related to the sensitivity of HRP2 – based RDT in the commune of Dioro (Mali).
Subjects and methods: We performed RDTs and thick smears to detect P. falciparum from 691 persons who sought diagnosis and treatment because they had fever or chills between May 2012 and June 2013. The RDTs used were based on histidine-rich protein 2 (HRP2).
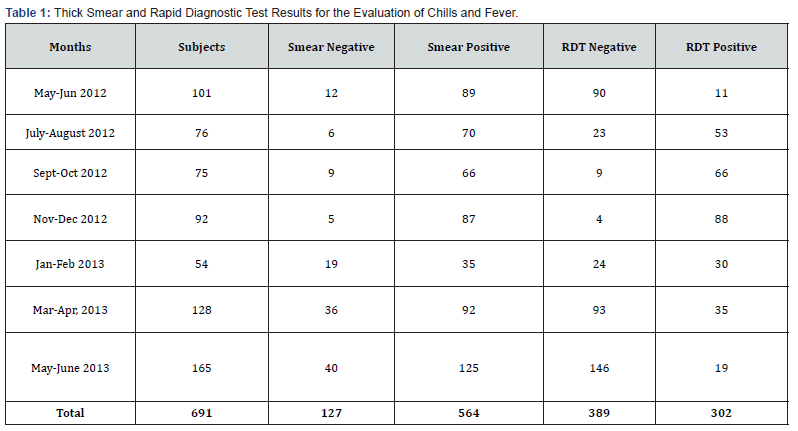
Results: The frequency of false-negative RDTs was highest in the dry season (peaked at 86-88% in May-June of 2012 and 2013), fell to 20-25% with the onset of seasonal rains in July-August and was lowest (1-2%) at the peak of the transmission season between September and November.
Conclusion: False-negative RDTs are a serious problem in the diagnosis of individual patients and the assessment of malaria control programmes. These results indicate HRP2-based RDTs in the Sahel region of Mali are most sensitive during the peak transmission (rainy) season and least sensitive in the low transmission (dry) season. They suggest that better mass diagnostic strategies will be necessary during the dry season to identify and treat infected persons and thus prevent the return of transmission when rains resume, and vector populations increase.
2. Keywords:Malaria; Diagnosis; RDT; HRP2; Sensitivity
Introduction
Both the diagnosis of illness in symptomatic subjects and the success of malaria control programmes depend on rapid diagnostic tests (RDTs) for P. falciparum infection [1,2] The rationales for using these RDTs are to reduce the time required for the preparation and reading of blood smears and identify persons and communities infected with asexual Plasmodium falciparum parasites in areas where there are few experienced microscopists [1,2]. Most of the RDTs used to diagnose P. falciparum infection are based on the detection of histidine-rich protein 2 (HRP2) produced by P. falciparum-parasitized red blood cells [3–6]. In comparison to other antigen detection tests, the detection of HRP2 is more sensitive because the parasite produces greater amounts of HRP2 than other proteins and because HRP2 is transported from the parasite to the red cell surface and then released into the bloodstream [7,8] Although RDTs based on aldolase (genus-specific) and lactate dehydrogenase (genus and species [falciparum] specific) are available, they are less sensitive than HRP2-based tests and are not specific for P. falciparum [1,2].
HRP2-based RDTs are used to diagnose uncomplicated P. falciparum malaria because they have been reported to be sensitive (90-98%) for the detection of Plasmodium falciparum bloodstream infection in persons with symptoms. Because a number of studies have found excellent sensitivities for HRP2-based RDTs, [9–11] we expected to find similar sensitivities (≥ 90%) for P falciparum infection by thick smear when we screened subjects with fever and chills at the Dioro Hospital in Mali. However, during May and June of 2012, we found sensitivities < 20% for HRP2-based RDTs in symptomatic subjects with positive thick smears. This report describes the seasonal changes observed in the frequency of false-negative RDTs between May 2012 and June 2013 using RDTs from the Mali National Malaria Control Programme (NMCP), which were within their recommended dates (before expiration) and had positive readings for the control strip containing HRP2 provided by the manufacturer.
Methods
Patient Population
Subjects invited to participate in this study were children 2 to 15 years of age with fever, chills or similar symptoms who were brought to the hospital by their parents or guardians for diagnosis and treatment of acute uncomplicated malaria.
Screening of Potential Study Participants
HRP2-based RDTs: Screening was based on the standard of care for persons with symptoms of acute malaria in Mali; it included an HRP2-based RDT, a thick smear for asexual P. falciparum parasites and a blood hemoglobin level. HRP2-based RDTs were performed as recommended in the package insert for the test kits supplied by the Mali NMCP (Bioline™, First Response®, Paracheck®). Based on finger sticks performed after cleaning the fingertip with alcohol, blood was applied to the cassette using a capillary tube (5 ml, Bioline, First Response) or a loop provided with the kit (Paracheck) and read by two independent observers in a well-lit environment after incubation at ambient temperature for 15-20 minutes. Readings were positive if the P. falciparum positive control line (C) and the sample being tested (T) were both positive. Readings were negative if the control line (C) was positive, and the sample being tested (T) was negative. Tests yielding a negative control line (C) were invalid and were repeated.
Thick Smears for P. falciparum: A separate drop of blood from the finger stick was applied to the surface of a freshly cleaned glass slide, spread evenly in a circle 2 cm in diameter and allowed to dry before staining with Giemsa. After rinsing and drying, stained slides were read using oil immersion (1,000 X) magnification to count the number of asexual P. falciparum parasites per 300 white blood cells 12 and to determine whether other malaria parasite species were present. Each slide was read by 2 independent observers: slides for which there was disagreement on the parasite count (differences ≥ 20%), on the presence of asexual P. falciparum parasites or on the presence of species other than P. falciparum were reviewed by a third (senior) microscopist whose reading was entered into the data base as final. Otherwise, the two independent readings were averaged and entered as the baseline asexual parasite count at the time of diagnosis. Investigators who read the microscopic slides were blinded to (did not have access to) information about the RDT result, Hb level or the patient’s history or physical examination.
Hemocue Hemoglobin (Hb) Determinations: Another drop of blood from the finger stick was applied to a Hb 201 Microcuvette provided in the test kit and read using the Hb 201 DM analyzer (spectrophotometer) available from the manufacturer (Hemocue AB – Ängelholm, Sweden). Readings were standardized using the low and high Hb controls recommended by the manufacturer and by using the internal electronic self-test (EQC) provided with the analyzer each day before the testing of patient samples.
Clinical examination: Subjects 2 to 15 years of age with fever, chills, or other symptoms consistent with malaria were invited to participate in a prospective study of the treatment provided by the NMCP and MOH for uncomplicated P. falciparum malaria. Inclusion criteria for this study included: ≥ 2,000 asexual P. falciparum parasites per ml on the thick smear, 2] diagnosis of uncomplicated P. falciparum malaria, 3] written informed consent from participants’ parents or guardians and the assent of subjects ≥ 14 years of age. Exclusion criteria included one or more of the following: 1] P. falciparum asexual parasite counts < 2,000 per ml, 2] parasites other than P. falciparum on the thick smear (P. malariae, P. ovale, P. vivax), evidence for severe or complicated malaria, including ≥ 200,000 asexual P. falciparum parasites per ml, impaired consciousness, coma or recent seizures, blood Hb level < 7 gms per dL on screening, hypersensitivity to either of the medications used to treat uncomplicated P. falciparum malaria (artemether, lumefantrine) and 5] acute or chronic potentially confounding illnesses other than uncomplicated P. falciparum malaria.
Results
Seasonal Variation in the Sensitivity of HRP2-based RDTs
The results reported here are based on 691 specimens from subjects 2-15 years of age who came to the Dioro Hospital in Mali requesting diagnosis and treatment for malaria between May 2012 and June 2013. Because HRP2-based antigen testing is recommended for the evaluation of symptomatic patients when microscopy is not available [1,2] the results of HRP2-based antigen testing performed during the 14 months from May 2012 to June 2013 were compared to the microscopy for the same specimens. This comparison indicated that the sensitivity of HRP2-based RDTs varied seasonally Table 1, Figure 1. Based on microscopy as the gold standard, [12-14] the sensitivity of HRP2-based RDTs was highest during the peak transmission or rainy season (98% [109/111] in October-November 2012) and at its nadir during the low transmission or dry season (13% [37/231] in May-June 2012 and April-June 2013).
Seasonal Variation in Asexual Parasite Densities
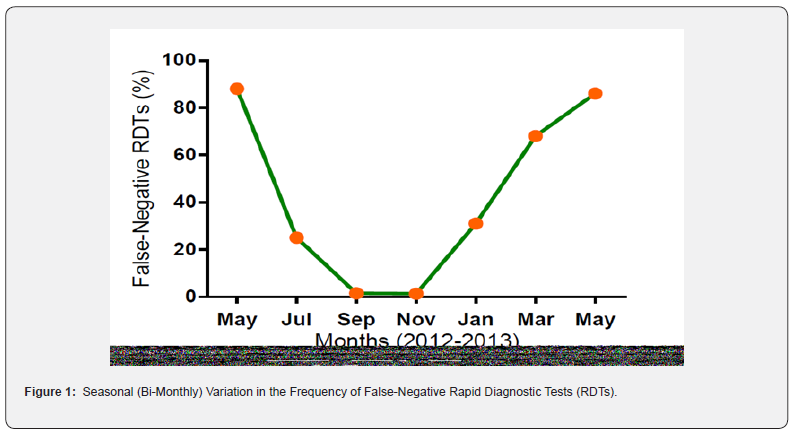
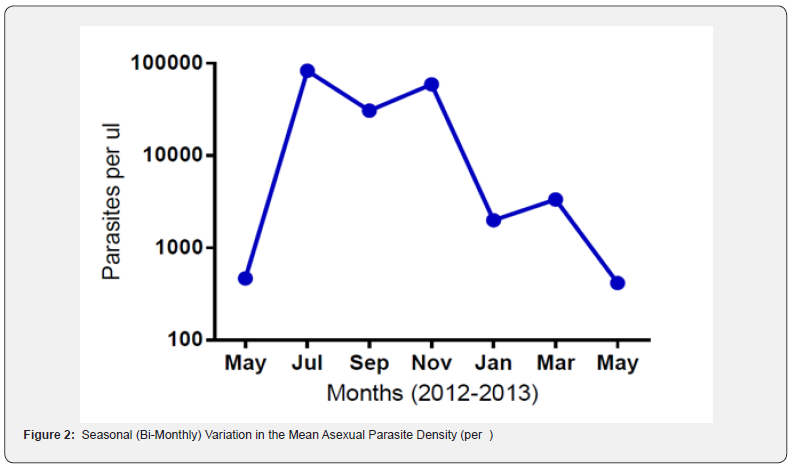
Based on microscopy, we examined the seasonal variations observed in mean asexual parasite counts for infected (smearpositive) subjects. Mean asexual parasite densities peaked during the transmission season (between July and December) and were lowest (at their nadir) at the end of the dry season Figure 2, Table 2. When mean asexual parasite densities during the transmission season were compared to those in the dry season, parasite densities were higher during the transmission season Table 3, p < 0.001, t=6.41, SEdiff=5498). Seasonal Variation in the Frequency of False-Negative RDT Results: Controlling for Confounding by Concomitant Seasonal Changes in Asexual Parasite Densities.

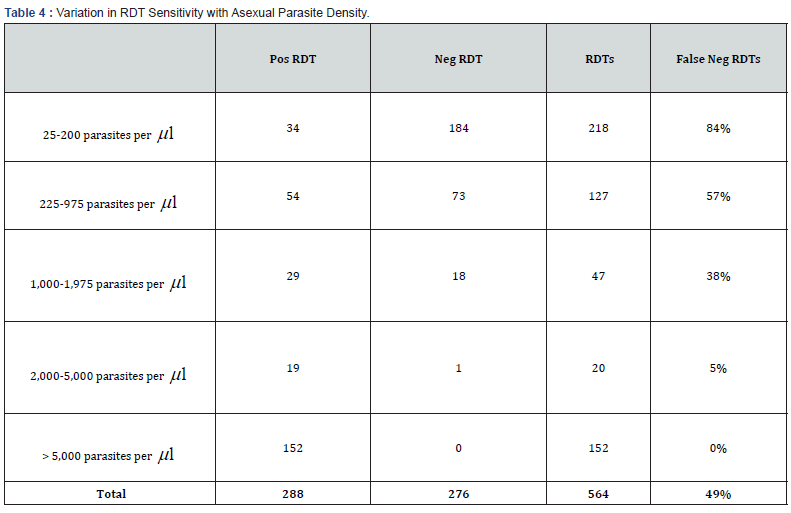
Because the sensitivity of HRP2-based RDTs is lower for specimens with fewer asexual parasites, [15] it was necessary to control for confounding by seasonal variation in the mean parasite counts Figure 2 and Table 3 to examine the effects of seasonal variation on the frequency of false-negative RDTs. This was accomplished by stratifying the RDT results for individual specimens according to their asexual parasite counts. Thus, seasonal differences in the frequency of false-negative RDTs identified by these comparisons cannot be explained by seasonal differences in the parasite counts (Panels A-C, Table 4. Although virtually all RDT results were true positives at parasite densities > 2,000 per ml during the transmission season (166/167), there were so few samples with > 2,000 parasites per ml during the dry season (n=5) it was not possible to test whether increased numbers of false-negative RDTs were observed during the dry season with samples containing > 2,000 asexual parasites per ml (Panel D, Table 4).
Quality Control Issues potentially related to False-Negative RDT Results
Faulty or Expired Rapid Diagnostic Test (RDT) Cassettes
The RDT cassettes used in this study were provided by the Mali NMCP, were prepared by manufacturers participating in the WHO Quality Control Programme (First Response®, Paracheck®, SD Bioline™) and were used before their dates of expiration. The Mali investigators who performed this testing had been trained to accept only data from RDTs with clearly positive results for the “C” (control) band with Plasmodium falciparum HRP2 antigen and to discard and repeat tests for which the “C” (control) band was negative or uninterpretable.
Possible False Positive Thick Smears
The thick smears for specimens that yielded false-negative RDT results (positive thick smear, negative RDT) have each been reviewed by an independent microscopist. The results of that review indicate that each of those slides was positive for asexual P. falciparum parasites.
Discussion
The major finding from these studies is the marked seasonal variation observed in the frequency of false-negative HRP2-based RDTs in Dioro. The frequency of false-negative test results in persons with positive thick smears for P. falciparum peaked at 86-88% (sensitivity of 12-14%) during the dry season (May-June of 2012, April-June of 2013). In contrast, the sensitivity of HRP2- based RDTs was high during the transmission/rainy season (98- 99% true positive RDTs). These results indicate that the sensitivity of HRP2-based RDTs was excellent during the active transmission (rainy) season, but unacceptably low during the dry season.
Because these seasonal changes are so striking, one logical question is why they have not been recognized previously because HRP2-based RDTs have been used for more than a decade? [10,11,13,15] In part, the answer is that false-negative HRP2-based RDTs have in fact been known for some time and - as a result - have been reported from Asia, South America and sub-Saharan Africa [16–22] What has not been reported previously is that changes in the frequency of false-negative RDTs may be markedly seasonal. In fairness, we failed to recognize the importance of seasonality in our initial studies, presumably because we obtained most of those samples during the rainy (transmission) season [3] We discovered the pronounced seasonality of these changes only later when we screened children with fever and chills from May 2012 to June 2013 to examine the efficacy of the treatment recommended for children with uncomplicated P. falciparum malaria in Mali by the NMCP and Ministry of Health [23]
A second question is whether this problem is restricted to Mali or is more widespread (e.g., whether it could be present in all P. falciparum-endemic areas?). Although large numbers of falsenegative RDTs in persons with positive smears are uncommon, recent publications have noted false-negative RDTs in subjects with positive blood smears in sub-Saharan Africa, South America and Southeast Asia [16–20] Thus, it is clear that the challenges posed by false-negative HRP2-based RDTs are not unique to Mali [23].
From a clinical perspective, the importance of these observations cannot be overstated. HRP2-based RDTs are at the center of the currently recommended approach to the diagnosis and treatment of uncomplicated malaria in human subjects [1,2,10,11,13,23,24]. They are particularly important in areas where microscopy is not available or requires delays that interfere with prompt treatment. In terms of clinical practice, people with positive RDTs can be treated rapidly (within an hour or less of diagnosis). Conversely, persons with negative RDTs are examined for other causes of their illness and are typically not treated with antimalarials.
The use of RDTs as the focal point of the approach to patients with chills and fever in malaria-endemic areas has a simple logic that can be enormously helpful in guiding the diagnosis and treatment of persons who may have malaria. However, to the extent that false-negative HRP2-based RDTs are as common elsewhere as they are in Dioro, this approach will need to be re-examined (because it rests on HRP2-based RDTs). Please note also that this strategy (treating only subjects with positive RDTs) will be counterproductive if large numbers of persons with P. falciparum malaria are not treated because they have false-negative RDTs.
From a public health or malaria control perspective, falsenegative RDTs have the potential to produce problems. This is because the goal of malaria elimination is to interfere with transmission and thus to prevent new infections and diseases in previously uninfected individuals. Thus, the time of greatest importance in areas with seasonal malaria is likely to be near the end of the dry season when the numbers of infected persons may be increasing rapidly. Unfortunately, this is also a time when parasite densities are often low Figure 2 and false-negative HRP2- based RDTs may be more common Figure 1.
From a theoretical perspective, three questions should be
considered:
First: Is the high frequency of false-negative RDTs during
the dry season restricted to Mali or is it a potential concern in
all countries with active transmission of P. falciparum malaria?
Previous reports of smear-positive specimens with false-negative
RDTs or deletions of hrp2 from sub-Saharan Africa, South America
and Asia suggest that this question should be considered in a
number of malaria-endemic countries. Preliminary discussions
with colleagues from Senegal and The Gambia suggest that this
problem is not universal in West Africa although they have not yet
performed studies during the dry season.
Second: In areas where false-negative HRP2-based RDTs
interfere with the diagnosis and treatment of infected (smearpositive)
individuals, how should the role of HRP2-based RDTs
change? For example, in the short-term (before alternative
diagnostic approaches are available), would it be reasonable to
continue using HRP2-based RDTs during the transmission season
when one would expect sensitivities ≥ 95%? In addition, what
would be the impact of using RDTs that test for both parasite LDH
and HRP2 on sensitivity during the dry season?
Third: What biologic mechanisms are responsible for these
effects, and do they (can they) explain the occurrence of falsenegative
HRP2-based RDTs during the dry season? Based on
previous studies, it seems plausible that deletions, spontaneous
point mutations, rearrangements and other mechanisms may each
be involved in some of the many hrp2-deficient isolates that have
been identified. Finally, it is not clear whether parasites lacking
HRP2 are more common during the dry season or are simply more
apparent because the numbers of other parasites (which have
HRP2 and produce the HRP2 protein) are lower.
Author contribution
Lansana Sangaré, Aliou Sissako, Vincent Sanogo, Chacka Coulibaly, Pamela P Badjogo, Youssouf Diarra collected and analyzed the data Ousmane A Koita and Donald J Krogstad contributed to study design as well as the writing of the manuscript. All authors agreed on this final version of the manuscript to be submitted.
Funding
These studies were supported by a Cooperative Agreement to develop an International Center of Excellence for Malaria Research in West Africa from the National Institute of Allergy and Infectious Diseases (U19 AI 089696) and by the University of Bamako in Bamako, Mali
Declaration
Ethics approval: Ethical review and approval was obtained from the Internal Review Board of the Institut National de Recherche en Santé Publique (INRSP, Ministry of Health and Public Hygiene, FWA 00000892). Parents or guardians of study participants were asked to provide written informed consent.
Consent to participate individual consent/assent was obtained from parents or relatives of each study participant based on the directives of the Ethics committee.
Conflict of interests/competing interests
The author reports no conflicts of interest in this work.
References
- Abba K, Deeks JJ, Olliaro PL, Cho MN, Sally MJ, et al. (2011) Rapid diagnostic tests for diagnosing uncomplicated falciparum malaria in endemic countries. Cochrane Database Syst Rev (7): CD008122.
- Bell D, Wongsrichanalai C, Barnwell JW (2006) Ensuring quality and access for malaria diagnosis: how can it be achieved?. Nat Rev Microbiol 4(9):682-695.
- Koita OA, Doumbo OK, Ouattara A, Lalla KT, Aoua K, et al. (2012) False-negative rapid diagnostic tests for malaria and deletion of the histidine-rich repeat region of the hrp2 gene. Am J Trop Med Hyg 86(2): 194-198.
- Maltha J, Gamboa D, Bendezu J, Luis S, Lieselotte C, et al. (2012) Rapid diagnostic tests for malaria diagnosis in the Peruvian Amazon: impact of pfhrp2 gene deletions and cross-reactions. PloS One 7(8): e43094.
- Kumar N, Pande V, Bhatt RM, Naman KS, Neelima M, et al. (2013) Genetic deletion of HRP2 and HRP3 in Indian Plasmodium falciparum population and false negative malaria rapid diagnostic test. Acta Trop 125(1): 119-121.
- Houzé S, Hubert V, Le Pessec G, Le Bras J, Clain J (2011) Combined deletions of pfhrp2 and pfhrp3 genes result in Plasmodium falciparum malaria false-negative rapid diagnostic test. J Clin Microbiol 49(7): 2694-2696.
- Baker J, Gatton ML, Peters J, Ho MF, McCarthy JS, et al. (2011) Transcription and expression of Plasmodium falciparum histidine-rich proteins in different stages and strains: implications for rapid diagnostic tests. Plos One 6(7): e22593.
- Desakorn V, Dondorp AM, Silamut K, Wirichada P, Duangjai S, et al. (2005) Stage-dependent production and release of histidine-rich protein 2 by Plasmodium falciparum. Trans R Soc Trop Med Hyg 99(7): 517-524.
- Hopkins H, Bebell L, Kambale W, Dokomajilar C, Rosenthal PJ, et al. (2008) Rapid diagnostic tests for malaria at sites of varying transmission intensity in Uganda. J Infect Dis 197(4): 510-518.
- Mouatcho JC, Goldring JPD (2013) Malaria rapid diagnostic tests: challenges and prospects. J Med Microbiol 62(Pt 10):1491-1505.
- Maltha J, Gillet P, Jacobs J (2013) Malaria rapid diagnostic tests in endemic settings. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis 19(5): 399-407.
- Payne D (1998) Use and limitations of light microscopy for diagnosing malaria at the primary health care level. Bull World Health Organ 66(5): 621-626.
- White NJ, Pukrittayakamee S, Hien TT, Faiz MA, Mokuolu OA, et al. (2014) Malaria. Lancet Lond Engl 383(9918): 723-735.
- Murphy SC, Shott JP, Parikh S, Etter P, Prescott WR, et al. (2013) Malaria diagnostics in clinical trials. Am J Trop Med Hyg 89(5): 824-839.
- Abeku TA, Kristan M, Jones C (2008) Determinants of the accuracy of rapid diagnostic tests in malaria case management: evidence from low and moderate transmission settings in the East African highlands. Malar J 7: 202.
- Batwala V, Magnussen P, Nuwaha F (2010) Are rapid diagnostic tests more accurate in diagnosis of plasmodium falciparum malaria compared to microscopy at rural health centres? Malar J 9: 349.
- Shakely D, Elfving K, Aydin Schmidt B, Mwinyi IM, Ulrika M, et al. (2013) The usefulness of rapid diagnostic tests in the new context of low malaria transmission in Zanzibar. PloS One 8(9): e72912.
- Trouvay M, Palazon G, Berger F, Béatrice V, Denis B, et al. (2013) High performance of histidine-rich protein 2 based rapid diagnostic tests in French Guiana are explained by the absence of pfhrp2 gene deletion in falciparum. PloS One 8(9): e74269.
- Ishengoma DS, Francis F, Mmbando BP, John P AL, Pamela M, et al. (2011) Accuracy of malaria rapid diagnostic tests in community studies and their impact on treatment of malaria in an area with declining malaria burden in north-eastern Tanzania. Malar J 10: 176.
- Wurtz N, Fall B, Bui K, Aurélie P, Mansour F, et al. (2013) Pfhrp2 and pfhrp3 polymorphisms in Plasmodium falciparum isolates from Dakar, Senegal: impact on rapid malaria diagnostic tests. Malar J 12: 34.
- Pati P, Dhangadamajhi G, Bal M, Ranjit M (2018) High proportions of pfhrp2 gene deletion and performance of HRP2-based rapid diagnostic test in Plasmodium falciparum field isolates of Odisha. Malar J 17(1): 394.
- Funwei R, Nderu D, Nguetse CN, Bolaji NT, Catherine OF, et al. (2019) Molecular surveillance of pfhrp2 and pfhrp3 genes deletion in Plasmodium falciparum isolates and the implications for rapid diagnostic tests in Nigeria. Acta Trop 196: 121-125.
- Baltzell KA, Shakely D, Hsiang M, et al. (2013) Prevalence of PCR detectable malaria infection among febrile patients with a negative Plasmodium falciparum specific rapid diagnostic test in Zanzibar. Am J Trop Med Hyg 88(2):289-291.
- McMorrow ML, Masanja MI, Kahigwa E, Abdulla SMK, Kachur SP (2010) Quality assurance of rapid diagnostic tests for malaria in routine patient care in rural Tanzania. Am J Trop Med Hyg 82(1): 151-155.






























