COVID-19 Vaccination Hesitancy: Preliminary Questionnaire Survey of Knowledge and Attitudes in Libya
Abdusalam Mahmoud1*, Abdurraouf Said2, Abdurrazag Nami3, Halla Elshwekh4 and Ahlam Ellafi5
1Department of Preventive Medicine, Faculty of veterinary Medicine, University of Tripoli, Libya
2Department of Anaestheisa and Intensive care, faculty of Medical Technology, University of Tripoli
3Department of Biology, Faculty of Arts and Sciences, Kasr Khiar, AL-Mergib University, Libya
4Biotechnology Research Center (BTRC), Tripoli, Libya
5Department of Histology and Genetics, Faculty of Medicine, University of Tripoli, Libya
Submission: June 14, 2021; Published: June 25, 2021
*Corresponding author: Abdusalam Sharef Mahmoud, Department of Preventive Medicine, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya
How to cite this article:Abdusalam M, Abdurraouf S, Abdurrazag N, Halla E, Ahlam E. Covid-19 Vaccination Hesitancy: Preliminary Questionnaire Survey of Knowledge and Attitudes in Libya. JOJ Pub Health. 2021; 5(5): 555674. DOI: 10.19080/JOJPH.2021.05.555674
Abstract
Background: The pre-vaccination survey is considered an important tool for assessing and evaluating knowledge, attitudes and determine vaccine hesitancy. Throughout many decades the world had faced a challenge in fighting against the endemic, epidemic, and pandemic diseases that have been emerging and re-emerging special those that constitute a great potential risk on humanity’s life. Still, after more than one year and half of scientific milestones of COVID-19 pandemic, there are many countries scarifying and struggling to mitigate the impacts of SARS-CoV-2 among their societies.
Objectives: Our study was conduct to address, assessing and evaluate COVID-19 vaccine literacy (VL) prior to and during the vaccination campaign in Libya.
Material and Methods: The cross-sectional questionnaire survey was conducted during April 2021 one week prior COVID-19 vaccination campaign in Libya. All relevant data was obtained and collected by online and face-to-face interview. The sampling method with an online Microsoft form questionnaire resulted in collecting only 33 valid questionnaires. Further, the survey carried out also during vaccination campaign however, any participants who had already taken the COVID-19 vaccine were not indorsed in questionnaire survey.
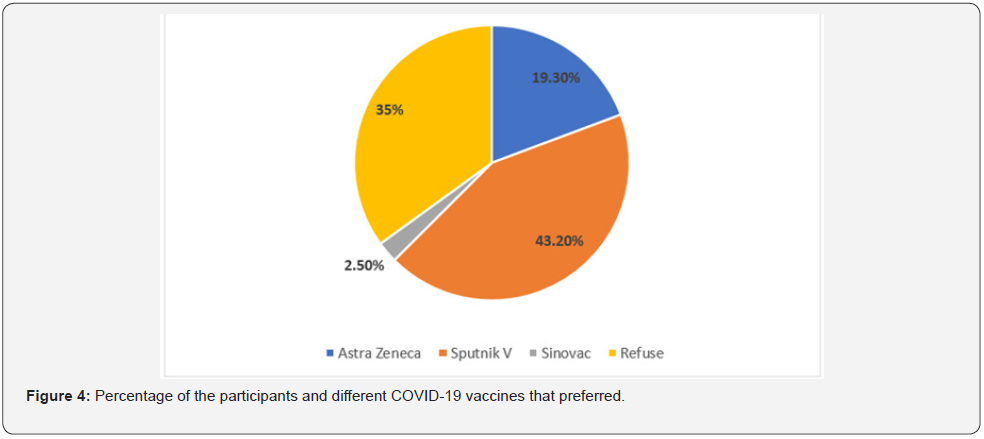
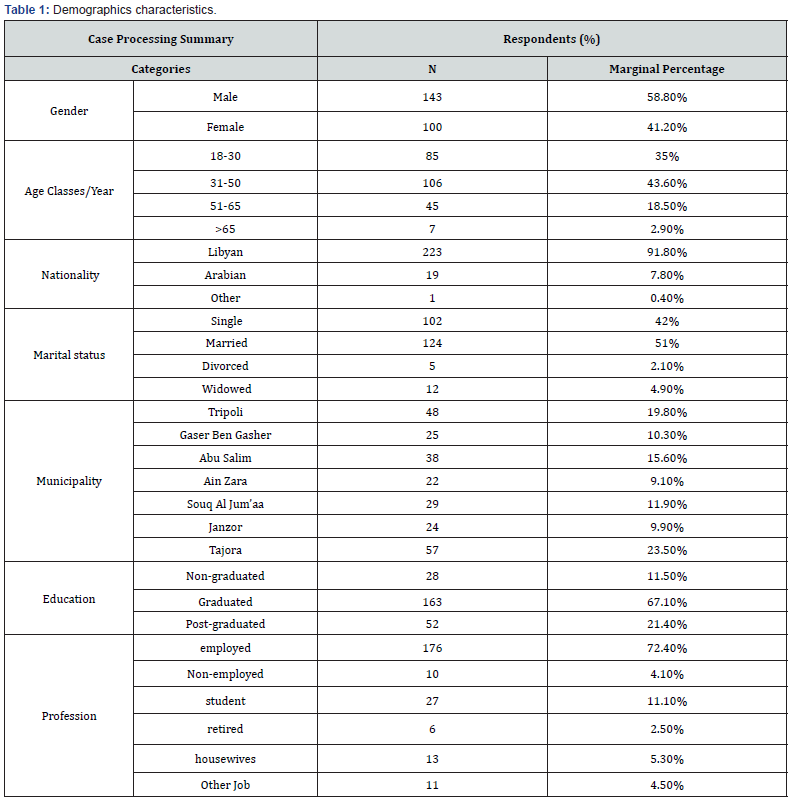
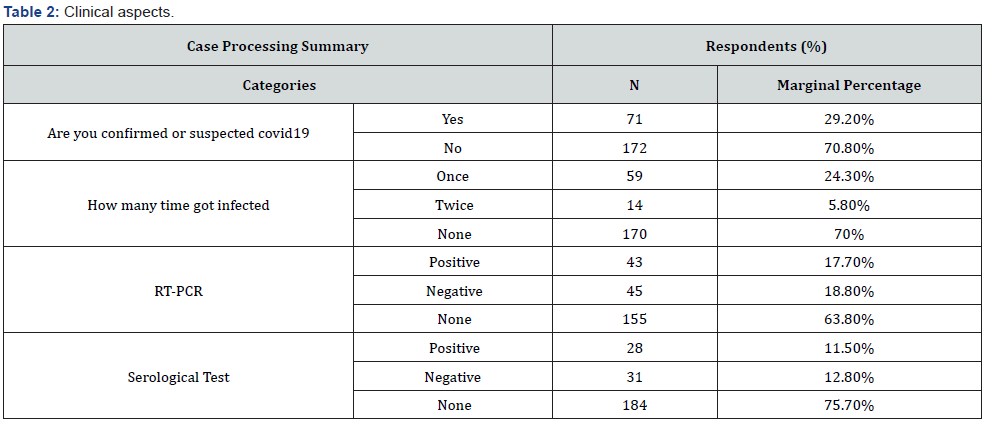
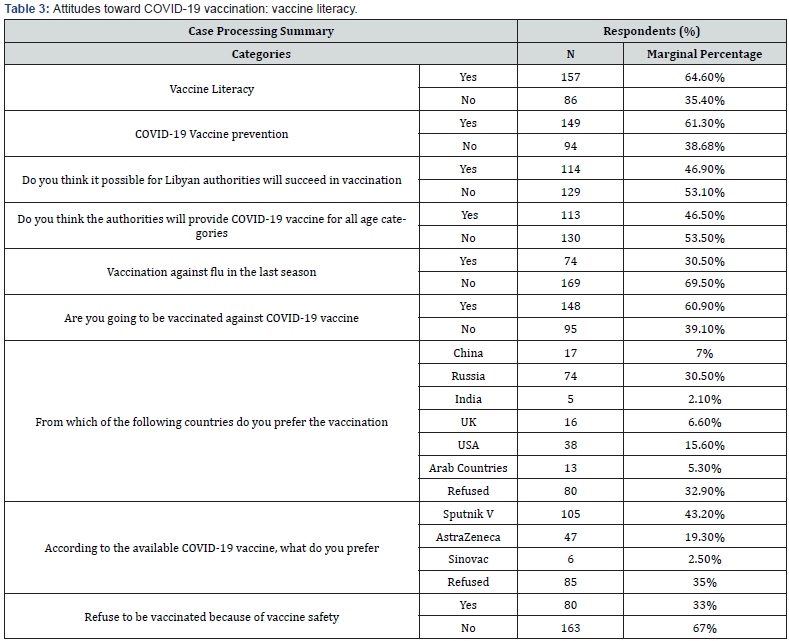
Results: A total of 243 participants were involved in this study. The overall response rate (45.6%). This study enrolled (58.8%) and (41.2%) of participants from male and female respectively. Considering the age classes, the results revealed that (35%, 18-30 year), (43.6%, 31-50 year), (18.5%, 51-65 year) and (>65 year, 2.9%). The results revealed that, 29.2% (95% Confidence Interval (CI): 25%-33.44%) of the respondents were confirmed or suspected COVID-19 infection, while and 70.8% (95% CI: 66.56%-75%) participants were not sure if they had contracted the virus. Regarding the recurrent COVID-19 infection, our questionnaire survey reported that, at least 5.8% (95% CI: 3.60%-7.92%) of the respondents had got infected twice by SRAS-CoV-2. Regarding the currently available COVID-19 vaccines (Sputnik V, AstraZeneca and Sinovac) in the country, the vast majority (43.2%) of the participants, they would like to prefer the Sputnik V, followed by (19.3%) AstraZeneca and (2.5%) Sinovac vaccines, while (35%) of the respondents refused to be vaccinated by three of these vaccines that have been provided by the Libyan healthy authorities under the supervision of NCDC. A proximately 33% (95% CI: 28.56%-37.28%) of the respondents refused to be vaccinated due to mistrust in the vaccine safety. Clearly, the vaccine safety was the main cause for the refuse and delay time of the vaccination from the most of the population.
Conclusion: Our study highlights that, the vaccine hesitancy is underestimated issue in Libya, and clearly there is misleading and misinformation about SARS-CoV-2 from unauthorized media sources. According to the metadata analysis of high efficacy, safety and successes of COVID-19 vaccines for mitigate and minimize both the case fatality rate (CFT) and transmission rate across the entire of the global, still there is a controversies around potential safety concerns of the COVID-19 vaccines. Urgently, the Libyan healthy authority needs to enforce the public health education about vaccine safety, and Libyan healthy authorities should be implement an effective vaccination strategies that complement with international standard criteria for the vaccination policy to improve the vaccination efficacy and the vaccine coverage rate. Consequently, The Libya national deployment and vaccination plan for the COVID-19 vaccine should implement National Immunization Technical Advisory Group (NITAG) in every municipality for collecting and processing information, and producing periodic reports during COVID-19 vaccination campaign.
Keywords: COVID-19 Vaccines; Vaccine Literacy; COVID-19
Introduction
The coronavirus disease-19 (COVID-19) emerged in Wuhan, China in late 2019. The COVID-19 is caused by severe acute respiratory syndrome (SARS-CoV-2). Consequently, SARS-CoV-2 spread and slipover outside China leading to a pandemic throughout the world resulting in public and socioeconomic impacts with high morbidity and mortality rate that exceeds the peak of the records in the humanitarian history. Globally, until writing this paper, the COVID-19 pandemic has caused 170,426,245 and 3,548,628 of confirmed cases and death respectively of the SARS-CoV-2, and as of 31 May 2021, a total of 1,579,416,705 vaccine doses have been administered [1]. Throughout many decades the world had faced a challenge in fighting against the endemic, epidemic, and pandemic diseases that have been emerging and re-emerging especially those that constitute a great potential risk on humanity’s life. Still, after more than one year of scientific milestones of COVID-19 pandemic, there are many countries scarifying and struggling to mitigate the impacts of SARS-CoV-2 among their societies. And also due to shortage of their public health capacities, fragility of health system and weakness of surveillance system likely in the almost African Countries [2,3]. Conversely, the countries that have met high standard level of public health capacities were able to mitigate and combat COVID-19 pandemic. However, one of the biggest challenges throughout medical and science history is how to implement blended strategy of prevention and control to combat of COVID-19 pandemic. Therefore, the vaccination strategies and policies are considered of great importance for the prevention, control and eradication of many pandemic diseases [4,5]. Consequently, the vaccination policy is a milestone for success of any vaccination campaign. Moreover, vaccination literacy is considered the main task and basic key clues for any vaccination campaign or programs especially those implemented in case of emergency during the epidemic or pandemic either on the regional or international level [6,7]. The Libyan authorities have been implemented vaccination strategies against many transmissible infectious diseases for the past couple of years. And the country is considered one of the most African Countries that strictly following the scheduled vaccination. There are different types of vaccines have been used for a long time as compulsory programmes and routinely applied as vaccines preventable diseases. The Libyan National Centre for Disease Control (NCDC) under the supervision of Libyan health authorities has been approved and authorized a list of vaccines preventable diseases (e.g. Bacillus Calmette-Guérinis (BCG), Oral Polio vaccine (OPV), Influenza vaccine, a combination of HEXA vaccines i.e Diphtheria, Tetanus, Whooping cough, Polio, Hepatitis B, and Haemophilus Influenzae type b (Hib), etc.) that, primarily used against most endemic and epidemic diseases [8]. In spite, vaccination policy well implemented and financially supported by Libyan authorities, still, the country facing great challenging in the deployment of vaccines. Notably, Libya has considered one of the many countries that had no pandemic preparedness plan concerning the vaccine deployment and monitoring toward COVID-19 pandemic, therefore, instability of the country led to impacts on the capacity of the public health infrastructure. Consequently, the Libyan government until beginning of April 2021 was not received the vaccine batches that has been distributed and implemented worldwide through the United-Nations-co-led COVAX initiative. Since 30 March 2020, WHO declared COVID-19 as public health emergency of international concern (PHEIC), Consequently, WHO and partners work together on the response and implementation of international projects for supporting vaccine production and safety for different countries started for vaccine manufacture for combat of SARS-CoV-2. The most African countries are relying on COVAX vaccines. Comparatively, according to the WHO data about 1% of the 1.3 billion of the global COVID-19 vaccines have been distributed in Africa. In addition, there is a slow rollout of COVID-19 vaccines in many African Countries, and almost have already used all of their COVAX doses. Urgently, WHO, United Nation and their partners are requested to support African Countries to mitigate COVID-19 specially those countries lack of fund and weakness of public health capacities [9]. Libya likes most of the African Countries, there are insufficient reporting and shortage of the data regarding the side effect of the vaccine, and moreover lack of trained professionals and hesitancy among the population that make almost countries faced a potential risks and challenges of COVID-19 vaccine deployments. Therefore, developing a vaccine against COVID-19 and maintaining the highest standards of safety is the main priority of the WHO. The vaccination strategy and policy are involved in the pre-vaccination and post-vaccination survey and efficacy. However, the pre-vaccination survey is considered an important tolls for assessing and evaluating knowledge, attitudes and determine vaccine hesitance [6]. Notably, the vaccine literacy in Libya is neglected and missed, therefore, we conducted this pilot questionnaire survey to address, assessing and evaluate COVID-19 literacy prior to and during the vaccination campaign in the Tripoli area, Libya.
Material and Methods
Study design and study area
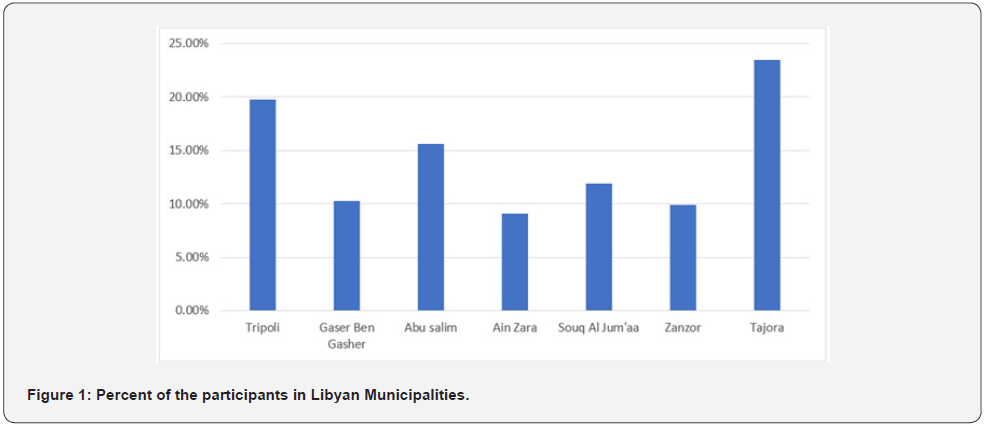
The cross sectional study survey was conducted during April 2021 in Seventh Libyan Municipalities (Tripoli, Gaser ben-Gasher, Ain Zara, Souq Al Jum’aa, Abu Salim, Janzor and Tazora) Figure 1. Online Microsoft form questionnaire and selfadministered structured questionnaire was designated and subjected to pretesting before submission and distribution among the population to obtain the relevant data. The questionnaire data collection tool was prepared in both, English and Arabic languages. The questionnaire composed of three parts, part-I (general information or demographics), part-II (Clinical aspects) and part-III (Vaccine Literacy).
Ethical respects
The ethical considerations were conducted throughout the study to obtaining all the relevant information by informed consent from all participants prior to the starting of the study. Nevertheless, during this study we followed all the criteria considering basic ethical principles in the questionnaire survey, like, providing anonymity and confidentiality.
Questionnaire Survey
This questionnaire survey was conducted one week prior to the COVID-19 vaccination campaign in Libya. The COVID-19 massive vaccination campaign was conducted on 17th April 2021. On first March 2021, the Libyan healthy authority, under supervision of NCDC in Libya implemented software for registration through an online link: https://www.eservices.ly. All relevant data was obtained and collected by online and face-toface interviews. Unexpectedly, the total number of participants in an online Microsoft form questionnaire resulted in collecting limited number of only 33 valid questionnaires, and out of (500) a self-administered structured distributed questionnaire resulted in collecting 210 completed answered questionnaires.
Statistical analysis
All obtained questionnaire data was analysed by SPSS version 22. A descriptive analyses frequency and percent were measured for numerical data and number and percent for qualitative data. For each proportion the percent and 95% CI were calculated (Figures 2 & 3).
Results
Demographics characteristics
Of the (533) participants, a total of 243 participants were involved in this study. The overall response rate (45.6%). This study enrolled (58.8%) and (41.2%) of participants from male and female respectively. Considering the age classes, the results revealed that (35%, 18-30 year), (43.6%, 31-50 year), (18.5%, 51-65 year) and (>65 year, 2.9%). Regarding the nationalities participants, the Libyan were represents (91.8%), while (7.3%) and (0.4%) were from Arabian and other the nationality respectively. The result showed that different proportion (19.8%, 10.3%, 15.6%, 9.1%, 11.9%, 9.9% and 23.5%) of the participants belonging to seventh Libyan municipalities, Tripoli, Gaser Bengasher, Abu Salim, Ain Zara, Souq Al Jum’aa, Janzor and Tajora respectively.
Clinical Aspects
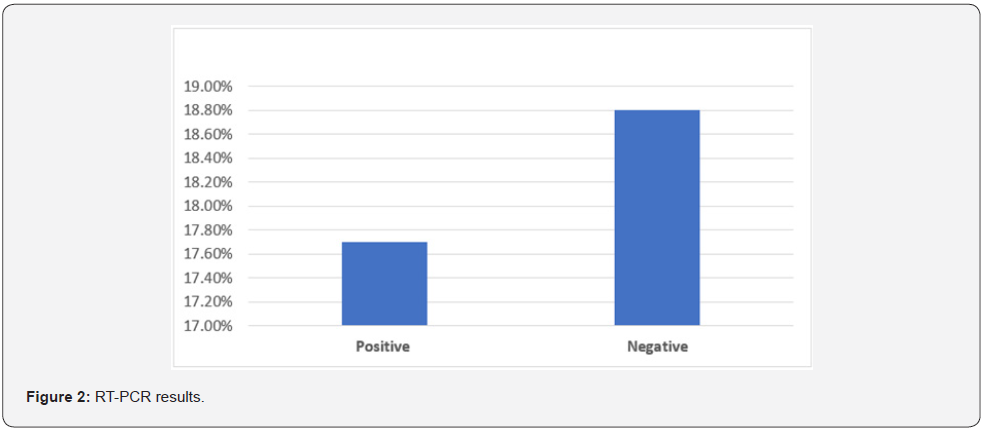
The results revealed that, 29.2% (95% CI: 25.00%-33.44%) and 70.8% (95% CI: 66.56%-75.00%) of the respondents were confirmed or suspected to be positive and negative of COVID-19 respectively. Of 88 of the respondents was subjected to screen by RT.PCR, the results revealed that 48.86% (95% CI: 39.32%- 58.41%) of the participants were positive to SARS-CoV-2 infection. Regarding the recurrent COVID-19 infection, our questionnaire survey reported that, at least 5.8% (95% CI: 3.60%-7.92%) of the respondents had got infected twice by SRAS-CoV-2.
Vaccine literacy
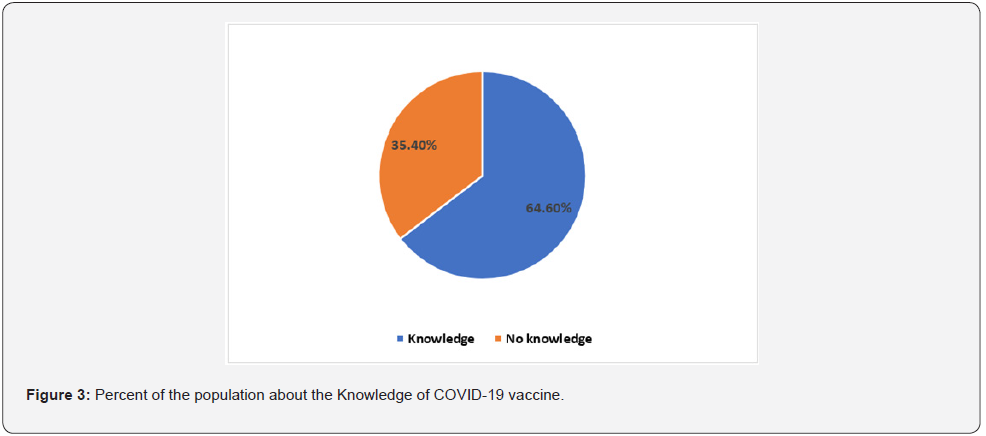
The results showed that 64.6% (95% CI: 60.17%-69.04%) of the participants had significance proportion of VL. The results reported 61.3% (95% CI: 56.80%-65.83%) a relatively higher proportion of the participant’s beliefs about vaccination would be provide prevention against SARS-CoV-2 infection. About 53.1% (95% CI: 48.46%-57.71%) of the participants they not beliefs and mistrust of the Libyan authorities will succeed in vaccination campaign against COVID-19. According to our presumptive results approximately 39.1% (95% CI: 34.57%-43.62%) of the participants are not going to be vaccinated against COVID-19 vaccine. Regarding the previous history of vaccination, if the participants had vaccinated against flu in the last season, the results showed that (30.5%) and (69.5%) had got vaccinated and not vaccinated respectively. A relative percent (35%) of the participants had accordance and agreement with Russian vaccines, followed by USA (15.6%), China (7%), UK (6.6%), Arab Countries (5.3%), and India (2.1%), while (32%) of the respondents refuse at all to be vaccinated by COVID-19 vaccines from list previous mentioned countries. Regarding the available COVID-19 vaccines in the country, the vast majority (43.2%) of the participants, they would like to prefer the Sputnik V, followed by (19.3%) AstraZeneca and (2.5%) Sinovac vaccines, while (35%) of the respondents refused to be vaccinated by three of these vaccines (Figure 4). A proximately 33% (95% CI: 28.56%-37.28%) of the respondents refused to be vaccinated due to mistrust in the vaccine safety (Tables 1-3).
Discussion
This present study results provide preliminary basic data about COVID-19 vaccination literacy among the Libyan population. However, there is a shortage information of the vaccination literacy in our country. To best our knowledge this first-based questionnaire study attempts to address, assess and evaluate COVID-19 vaccination literacy in Libya. Our study results reported variable data of COVID-19 vaccination literacy among Libyan population at least in surveyed municipalities. The present study showed a relatively high proportion (64.6%) of knowledge about vaccines, therefore, the most respondents have a good knowledge of vaccines in general. Along similar line, the most respondents misbeliefs about vaccination, if would be provide prevention against SARS-CoV-2 infection. In addition more than half of the participants misbeliefs and mistrust of the Libyan authorities, if they will succeed in vaccination campaign against COVID-19. In line with numerous studies reported lacking of the principle knowledge and inadequate attitudes about COVID-19 vaccines could be potentially impacts on vaccination campaign success [10,11,12]. In fact, the instability of the country during the last ten year could be considered one of the main factors for the misbeliefs, anti-vaccines beliefs and mistrust of the respondents toward the vaccination. Consequently, about 2/3 of the participants are not going to be vaccinated against COVID-19 vaccine. Notable, the present study reported a relative numbers of individual they have no intention and unwillingness to be vaccinated against SARS-CoV-2. Regardless, the type and manufactures companies of the COVID-19 vaccines, there were many Libyan citizen refusing to be vaccinated because of mistrust in vaccine safety and vaccination policies in the country. And there are different reasons behind the refusal of the vaccination against COVID-19 vaccines and that could be influence the vaccination literacy and attitudes [10,13]. Comparatively, our results showed that 35% of the respondents they have intention to be vaccinated with the Russian vaccine (Sputnik V), while a relatively accordance and agreement with other vaccines are belonging to USA (15.6%), China (7%), UK (6.6%), Arab Countries (5.3%), and India (2.1%), and (32%) of the respondents were in disagreement and refusal at all to be vaccinated by COVID-19 vaccines from list previous mentioned countries. The relatively high proportion of agreement with the Sputnik V vaccine (Gam-COVID-Vac) might be variable according to different factors that could be influence on the vaccination attitudes and acceptance among the Libyan citizens [14]. However, 92% efficacy have been reported for the Sputnik V vaccine according to data analysis of the Sputnik V vaccine against COVID-19 phase III clinical trials at the National Research Centre for Epidemiology and Microbiology [14,15]. According to the currently vaccines (Sputnik V, AstraZeneca and Sinovac), that have been provided by the Libyan healthy authorities under the supervision of NCDC, the results reported that the vast majority (43.2%) of the respondents have willing to be vaccinated by the Sputnik V as compare by other two type of vaccines. In line with previous studies, our study reported a significant mistrust of vaccine safety and that was potential influence on the COVID-19 vaccines acceptance [12,14]. Clearly, the vaccine safety was the main cause for the refuse and delay time of the vaccination from the most of the population. In spite many of the companies declared the safety milestone that required by the U.S. Food and Drug Administration (FDA) for Emergency Use Authorization (EUA) and other regulatory agencies around the world has been performed. And those companies have got the approval for the emergence uses of COVID-19 vaccines with of higher achievement efficacy rate and safety of some vaccines have been reported during the massive vaccination campaign around the entire world, still, there are inattentiveness and frightened from some unusual side effects have been reported from some vaccines [16]. Likely, unusual blood clotting during the massive COVID-19 vaccination campaign of AstraZeneca (Vaxzevria) and Johnson & Johnson (Ad26.COV2.S) vaccines that have been reported in some country [17,18,19,20]. However, still there is a scarcity of data and information regarding the main specific risk factors that might be linked with unusual clotting disorder in people vaccinated with Vaxzevria [16,18]. In addition, the another factors are the lack of the internet access in rural area, shortage of internetwork operating system and cost of internet access were considered the most obstacles and difficulties that have been left almost of communities out the vaccination campaign. The misleading and false information (fake news) from unauthorized media advices regarding the COVID-19 vaccines is another factor influencing vaccine literacy. Expectedly, the vaccination rate is much lower than that required, only less 8% of the population received a vaccination. There was delayed in the receiving of the vaccine batches have made difficulty in vaccination strategies in Libya. Therefore, the vaccines literacy is considered the potential factors for successes any vaccination campaign among the societies. Despite, our study have made a snapshot regarding COVID-19 vaccine hesitancy among the Libyan population at least in surveyed municipalities. Notably, our online survey has some limitations, and the response rate was very limited. Consequently, online questionnaires resulted in collecting a low number of valid questionnaires. Apparently, the instability in the country causes some difficulties to certain areas to have internet access and makes it difficult to respond to our online questionnaires. Conversely, a self-administered structured distributed questionnaire was more reliable, applicable, credible, and valuable at least in surveyed municipalities. In fact, the few limitations of this study are the short time of study and response rate by the online survey was incomplete because of instability and irregular supply for both electricity and internet access in Libya during this era. Also, the comparison of age and gender groups of respondents is disadvantaged when one of the pair of groups is small. The last limitation was regarding vaccine safety, limited choices of COVID-19 vaccines in Libya, the appearance of severe side effects related to AstraZeneca, and Sinovac was not approved by FDA yet, these made the respondents chose the sputnik.
Conclusion
Our study highlights that, the vaccine hesitancy is underestimated issue in Libya, and clearly there is misleading and misinformation about SARS-CoV-2 from unauthorized media sources. According to the metadata analysis of high efficacy, safety and successes of COVID-19 vaccines for mitigate and minimize both the case fatality rate (CFT) and transmission rate across the entire of the global, still there is a controversies around potential safety concerns of the COVID-19 vaccines. Urgently, the Libyan healthy authority needs to enforce the public health education about vaccine safety, and Libyan healthy authorities should be implement an effective vaccination strategies that complement with international standard criteria for the vaccination policy to improve the vaccination efficacy and the vaccine coverage rate. Consequently, The Libya national deployment and vaccination plan for the COVID-19 vaccine should implement NITAG in every municipality for collecting and processing information, and producing periodic reports during COVID-19 vaccination campaign.
Acknowledgments
The Authors would like to thank all medical staffs and people at seventh Libyan municipalities for their cooperation during study period.
Authors’ Contributions
Study Design: Abdusalam Mahmoud, Ahlam Ellafi. Computational framework and analysed the data: Abdusalam Mahmoud. Contributed to the interpretation of the results: Abdusalam Mahmoud, Ahlam Ellafi, Abdurraouf Said, Abdurrazag Nami, Halla Elshwekh. Abdusalam Mahmoud: took the lead in writing the manuscript. All authors provided critical comments, feedback and helped shape the research, analysis and manuscript.
Conflict of Interests
The authors declare that there is no conflict of interest.
Funding
The study not funded and the authors have not received any fund.
References
- World health Organization (WHO) (2021).
- Margolin E, Burgers WA, Sturrock ED, Mendelson M, Chapman R, et al. (2020) Prospects for SARS-CoV-2 diagnostics, therapeutics and vaccines in Africa. Nature Reviews Microbiology (18): 690-704.
- Nkengasong JN, Mankoula W (2020) Looming threat of COVID-19 infection in Africa: act collectively, and fast. Lancet 14, 395(10227): 841-842.
- World Health Organization (WHO) (2021) Operational guidance: COVID-19 immunization service delivery modalities. Copenhagen: WHO Regional Office for Europe.
- European Centre for Disease Prevention and Control (ECDC) Objectives of vaccination strategies against COVID-19. Technical report 23 Apr 2021.
- Al-Jayyousi GF, Sherbash MAM, Ali LAM, El-Heneidy A, Alhussaini NWZ, et al. (2021) Factors Influencing Public Attitudes towards COVID-19 Vaccination: A Scoping Review Informed by the Socio-Ecological Model. Vaccines 9(6): 548.
- Biasio LR, Bonaccorsi G, Lorini C, Pecorelli S (2021) Assessing COVID-19 vaccine literacy: a preliminary online survey. Hum Vaccin Immunother 17(5): 1304-1312.
- National Centre for Disease Control (NCDC) (2021).
- World Health Organization. (WHO) (2021).
- Chen T, Dai M, Xia S, Zhou Y (2021) Do Messages Matter? Investigating the Combined Effects of Framing, Outcome Uncertainty, and Number Format on COVID-19 Vaccination Attitudes and Intention. Health Commun 1-8.
- Lazarus JV, Ratzan SC, Palayew A, Gostin LO, Larson HJ, et al. (2021) A global survey of potential acceptance of a COVID-19 vaccine. Nat Med 27(2) 225-228.
- Wang J, Jing R, Lai X, Zhang H, Lyu Y, et al. (2020) Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China. Vaccines 8(3): 482.
- Johnson NF, Velásquez N, Restrepo NJ, Leahy R, Gabriel N, et al. (2020) The online competition between pro- and anti-vaccination views. Nature 582: 230-233.
- Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, et al. (2021) Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 397(10275): 671-681.
- The Gamaleya National Centre. The first interim data analysis of the Sputnik V vaccine against COVID-19 phase III clinical trials in the Russian Federation demonstrated 92% efficacy.
- Douxfils J, Favresse J, Dogne J, Lecompte T, Susen S, et al. (2021) Hypotheses behind the very rare cases of thrombosis with thrombocytopenia syndrome after SARS-CoV-2 vaccination. Thromb Res 203: 163-171.
- Voysey M, Clemens CA, Madhi AS, Weckx LY, Folegatti PM, et al. (2021) Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 397(10269): 99-111.
- European Medical Agency (EMA) An overview of Vaxzevria and why it is authorised in the EU. Vaxzevria0F (COVID-19 Vaccine (ChAdOx1-S [recombinant])) EMA/290514/2021.
- Shimabukuro TT (2021) Thrombosis with thrombocytopenia syndrome (TTS) following Janssen COVID-19 vaccine.
- Muir KL, Kallam A, Koepsell SA, Gundabolu K (2021) Thrombotic thrombocytopenia after Ad26.COV2.S vaccination. N Engl J Med 384(20): 1964-1965.






























