Monitoring of Insecticide Resistance of Anopheles arabiensis Patton To DDT 4%, Deltamethrin 0.05%, Permethrin 0.75% And Bendiocarb 0.1% In River Nile State, Sudan
Osman SOS1, Toto TH2, Abdalmajed MA3*, Jihad A4 and Azrag SR5
1Medical Entomologist, Sudan
2 Medical Entomologist (National Malaria control), Sudan
3 Public health Specialist (KMFI), Sudan
4 Medical Entomologist (ALGadaal Center-Sennar), Sudan
5Department of Zoology Faculty of Science, University of Khartoum (Medical entomologist), Sudan
Submission: September 3, 2019; Published: October 04, 2019
*Corresponding author: Abdalmajed MA, Public health Specialist (KMFI), Sudan
How to cite this article:Osman SOS, Toto TH, Abdalmajed MA, Jihad A, Azrag SR. Monitoring of Insecticide Resistance of Anopheles arabiensis Patton To DDT 4%, Deltamethrin 0.05%, Permethrin 0.75% And Bendiocarb 0.1% In River Nile State, Sudan. JOJ Pub Health. 2019; 5(2): 555656. DOI: 10.19080/JOJPH.2019.05.555656
Abstract
The study purposed to determine insecticide susceptibility status of An. arabiensis in the three sites in River Nile State of Sudan to DDT 4%, Deltamethrin 0.05%, Permethrin 0.75% and Bendiocarb 0.1%. The insecticide susceptibility tests were implemented using the WHO standard procedure under optimum conditions and by using impregnated papers with recommended diagnostic concentrations. The tests were done with three days-old an arabiensis non-blood fed females. One hundred twenty to one hundred fifty females were tested for each insecticide at the diagnostic concentration with 6 replicates of 20 - 25 mosquitoes per test. Control tests consisted of groups of 50 female mosquitoes exposed to papers impregnated with carrier oil without insecticide under the same conditions.
The exposure time was one hour; then mosquitoes were transferred into holding tubes for a 24 hours recovery period after that the mortality was recorded. Results revealed that An arabiensis was susceptible to Bendiocarb with 100% mortality rate in all three sites in River Nile state; Shendi, Alzidab and Al Bawga. Anophele sarabiensis was also susceptible to Deltamethrin in Al zidab (98.8%), Al bawga (98.7%) and resistant in Shendi (81.2%). Anopheles arabiensis showed resistance to Permethrin and DDT in all three sites with mortality rates ranged 31.4%-80.1%. For Bendiocarb KDT50 ranged between 18.1 - 38.5 minutes, while KDT95 ranged between 30.6-94.7 minutes. The fastest KDT50and KDT95recorded were in Shendi area. Whereas for Deltamethrin KDT50 ranged between 20.5 – 24.2 minutes, while KDT95 ranged between 90.6–132.2 minutes. The fastestKDT50 and KDT95 recorded was in Albawga. These results should be taken in consideration by the current vector control interventions in River Nile State.
Keywords:Susceptibility; Anopheles arabiensis; River Nile state
Introduction
Malaria is one of the most common infectious diseases and an enormous public health problem [1]. Globally, an estimated 3.2 billion people are at risk of being infected with malaria and developing disease, and 1.2 billion are at high risk (>1 in 1000 chance of getting malaria in a year). According to the latest estimates, 198 million cases of malaria occurred globally in 2013 and the disease led to 584 000 deaths. The burden is heaviest in the WHO African Region, where an estimated 90% of all malaria deaths occur, and in children aged under five years, who account for 78% of all deaths [1]. A number of 280 million people in Eastern Mediterranean Region where eight countries were at some risk of malaria with a figure of 104 million at high-risk. From them two countries accounted for >90% of the deaths in 2013 where the Sudan alone represent (67%) and Pakistan (24%) [1].
In African region there are several areas of critical concern because of particularly widespread resistance to pyrethroids or to multiple insecticide classes [2]. The situation of malaria in north Sudan, malaria represents around 21% of outpatient consultations and around 30% of inpatient admissions. However, the rates are significantly higher in rural when compared to urban populations. In Sudan Malaria causes considerable mortality, mainly among young children and pregnant women. Possible factors leading to this situation include floods, drought, famine, widely extended irrigated schemes without due consideration to the health component as well as population movement (internal displacement and influx refugees) [3].The situation is further complicated by the spread of insecticide and drug resistance [4]. The current study aimed to investigate insecticide resistance status of An. arabiensis in River Nile State as part of the overall mapping of insecticide resistance in the Sudan.
Materials and Methods
Study area: This study was conducted in River Nile State in northern Sudan during the period March to May 2015 River Nile State is located between Latitudes 16°-22° N and Longitudes 32°- 35° E. It’s bordered from the north by the Arab Republic of Egypt, from the East by Kassala state and Red Sea States, from the South by Khartoum State and from the West by the Northern State.
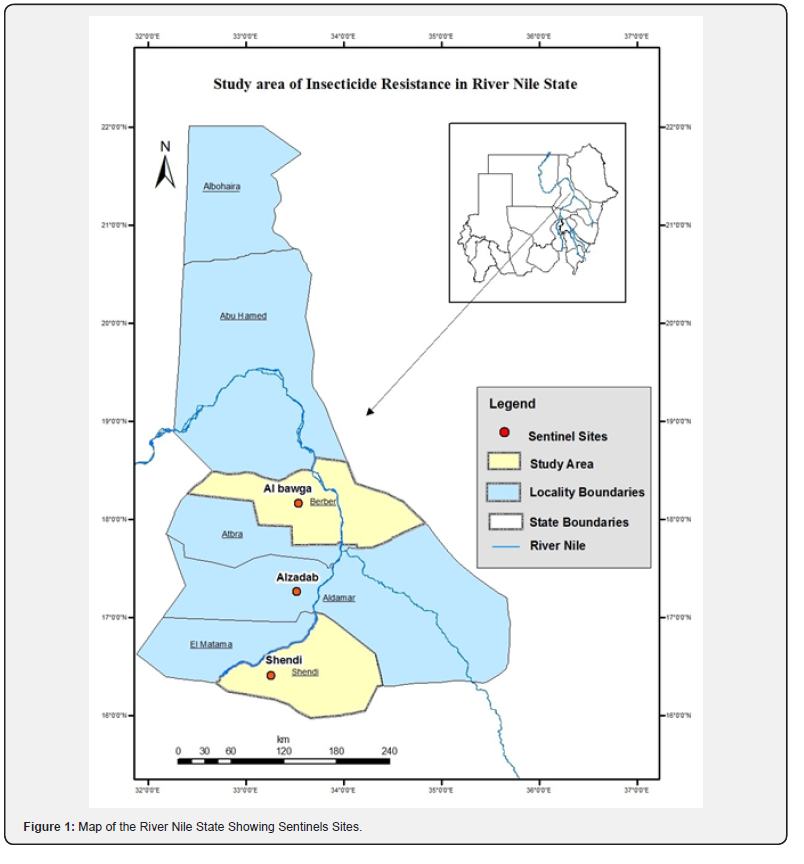
Study Sites: The selection of the study sites was based mainly on WHO criteria (accessibility, representation, availability, distribution and productivity of the breeding sites).Three localities representing irrigated and urban areas were chosen as sentinels sites, which were: Shendi site in Shendi locality(16° 40′ 631″ N, 033° 25′ 536″E; Al zidab site in Aldamar locality( 033° 51′ 783″ E , 17° 26′ 297″ and Al bawga site in Barbar locality( 18°’ 16.103” N, 33° ‘ 53.857” E): is in the northern sector of the study area (Figure 1).
Specimen collection, identification and rearing: Anopheles larvae were collected from different breeding sites using standard larval collection kits including plastic dippers, plastic screened netting, plastic pipettes, plastic buckets, dishes and sorted out from other aquatic organisms. Larvae were kept in plastic buckets and transferred to the laboratory. In the laboratory, larvae were reared and fed on rice powder. When pupation of reared larva occurred, pupae were sorted out by pipette and replaced in paper cups and inserted into the cages to emerge. Adults emerged from pupation during a period of two to three days. The emerging adults were fed only on 10% sugar solution (sucrose) and kept until tests started. Larvae were identified using morphological features according to the keys of [5]. When sufficient number was obtained as recommended by the WHO, the bioassay tests were carried out [6].
Insecticides susceptibility tests: Insecticide susceptibility tests were conducted using the WHO standard procedures [7]. Using test kits for adult mosquitoes Bioassay tests were done under optimum conditions (temperature 25Co±2 and 80%±10 RH). Impregnated papers with recommended diagnostic concentrations of 4% DDT, 0.5% Deltamethrin, 0.75% Permthrin, and 0.1% bendiocarb were used, Tests were carried out with one to three days-old An. arabiensis, non-blood-fed female mosquitoes.
One hundred twenty to one hundred fifty female mosquitoes were tested for each insecticide at the diagnostic concentration, with 6 replicates of 20-25 mosquitoes per test. Control tests consisted of a group of fifty mosquitoes exposed to papers impregnated with carrier oil without insecticide under the same conditions. At the end of exposure time (one hour), mosquitoes were transferred into holding tubes for recovery period during which time provided 10%sucrose solution and allowed a 24- hour recovery period after which mortality was recorded. During the exposure time, numbers of knocked down mosquitoes were recorded after (10, 15, 20, 30, 40, 50) and 60 minutes of exposure.
Statistical analysis: Data was analysed using SPSS statistical and EXCEL programme. Descriptive statistics as percentages was used to describe mortality rates and Probit statistical analysis was used to determineKdT50, KdT95.The resistant status of mosquito samples was determined according to the WHO criteria (WHO, 2013) as follows: mortality rates between 98%-100% indicate full susceptibility; mortality rates between 90%-97% require further investigation and mortality rates < 90%, the population is considered resistant to the tested insecticides. If the control mortality is between 5-20%, the percentage mortality should be corrected by Abbott’s formula.
Results
Species composition
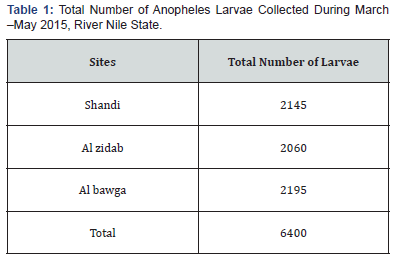
A total of 6400 Anopheles larvae were collected from the three sites from different types of larval habitats (Table 1). All specimens were identified morphologically as An. gambiae complex. As may studies showed that An. arabiensis is the only member of the An. gambiae complex in northern Sudan we accordingly assigned An. arabiensisto all larvae specimens found during this study
Susceptibility status of an arabiensis in river Nile state
A total of 1640 females An. arabiensis were exposed to the diagnostic concentrations of DDT (4%), Deltamethrin (0.05%), Permethrin (0.75%), and Bendiocarb (0.1%). A total of 72 replicates were conducted, in each replicate 25 females An. arabiensis were used. Control tests included 600 An. arabiensis females in 24 replicates.
Mortality rates
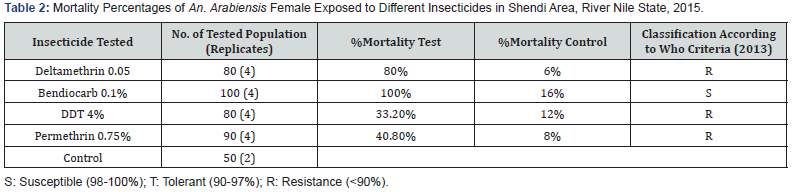
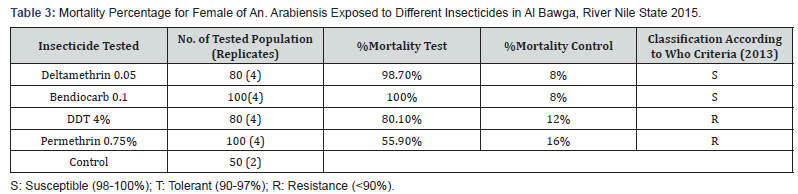
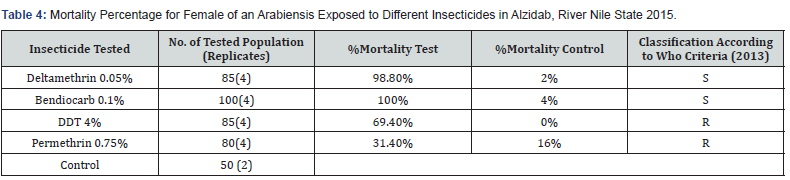
Based on the WHO criteria, bioassay results showed the an arabiensis was susceptible to Bendiocarb (0.1%) in Shendi, Albawga and Alzidab with 100% mortality rates in three sites. For Deltamethrin 0.05%Al bawga and Alzidab were susceptible 98.7% and 98.8% mortality rates respectively. However, An. arabiensis population from Shendi was resistance with 80% mortality rate. High rates of insecticide resistance were reported for DDT and Permithrin in all three study sites with mortality rates ranged between 80.1%-33.2% mortality rates for DDT. However, higher resistance rates with mortality rates ranged 55.9%-31.4% were reported using Permithrin shown in Table 2-4.
Results of KDT50 and KDT95
Bendiocarb 0.1%(KDT50) and (KDT95): The fastest KDT50 of An. arabiensis against Bendiocarb 0.1 was reported in Shendi area30.6 minutes (95% 28.3-33.8) followed by Albawga area36.6minutes (95% 34.3-39.2) and Alzidab area 38.5minutes (95% 36.4- 40.7). Fastest KDT95 was reported in Shendi area 30.6 minutes (95% 28.3-33.8), followed by Al zidab81.5minutes (95% 73.1-93.7) and Albawga area 94.7minutes (95% 82.6-112.9) shown in Table 5. Probit transformed responses of An. arabiensis exposed to Bendiocarb 0.1% (Table 5).
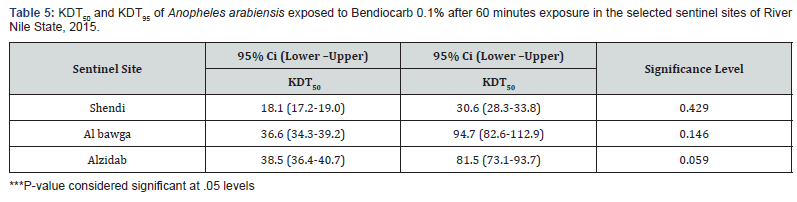
Deltamethrin 0.05%(KDT50) and (KDT95): The fastest KDT50 of An arabiensis against Deltamethrin 0.05% was reported in AlBawga area 20.5 minutes (95% 11.4-28.9) followed by Shendi area 24.0minutes(95% 18.8-29.6) and Alzidab area 24.2minutes (95% 21.8-26.7),The fastest KDT95 was found in Albawga area 90.6 minutes (95% 53.2-547.9) followed by Alzidab area 123.2 minutes(95% 97.2—170.8) and Shendi area 132.2minutes(95%84.2-327.8) shown in Table 6 .Probit transformed responses of An. arabiensis exposed Deltamethrin 0.05%shown in Table 6.
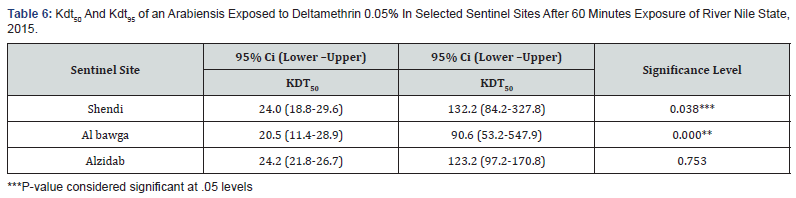
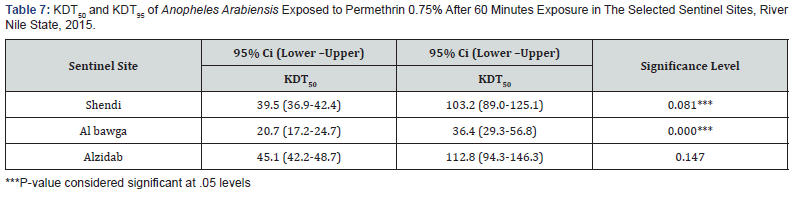
Permethrin 0.75% (KDT50) and (KDT95): The fastest KDT50 of An. arabiensis against Permethrin was found in Al Bawga area 20.7minutes (95% 17.2-24.7) followed by Shendi area 39.5minutes(95% 36.9-42.2) and Alzidab area 45.1minutes (95% 42.2-48.7), The fastest KDT95 was found in Al-bawga area 36.4 minutes (95% 29.3-56.8), followed by Shendi 103.2minutes (95% 89.0-125.1) and Al zidabarea 112.8minutes (95%94.3-146.3) shown in Table 7.
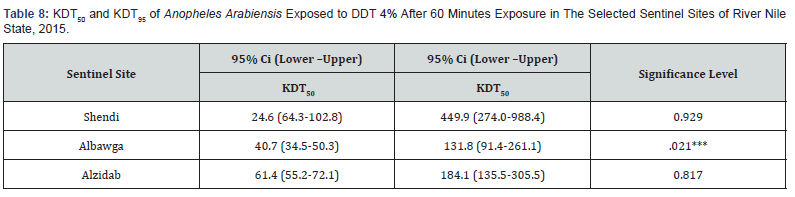
DDT 4% KDT50 and (KDT95): Shendi area also was registered the fastest KDT50 of An. arabiensis against DDT 4%24.67minutes (95% 18.8-29.6) followed by Albawga area40.7minutes (95% 34.5-50.3) and Al zidab area 61.47minutes (95% 55.2- 72.1). The fasts KDT95 was reported in Shendi area449.9 minutes (95% 274.0-988.0) followed by Al zidab184.1 minutes (95% 135.5-305.5)and Al-bawga area 131.8minutes (95% 91.4-261.1) shown in Table 8, Probit transformed responses of An. Arabiensis exposed DDT 4% shown in Fig 3.6 ,3.7 shown A comparison of Knockdown time 50%and Knock down 95.
Knockdown resistance gene detection: Samples were tested for mosquitoes of state stations in the microbiology laboratory of the National Malaria Research & Training Centre in Sinner area to compare the results in the method of testing sensitivity and genetic testing of the samples for detection of kdr mutations, genomic DNA was isolated from 136 mosquitoes according to the method of [7]. Two separate PCR reactions were run, one to detect alleles of the leucine-phenylalanine substitution, the other, to detect wild-type susceptible alleles following the methods described in [8,9]. The occurrence of kdr was confirmed by direct sequencing of the 293 base pair fragments of the sodium channel gene amplified using Agd1 and Agd2 primers.
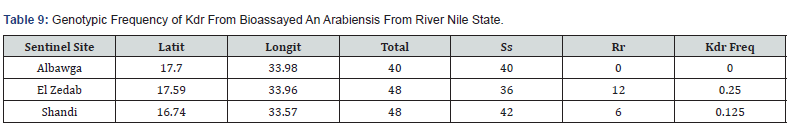
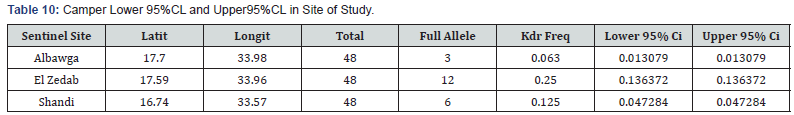
KDR gene detection: One hundred thirty-six An. arabiensis specimens from River Nile State were examined for the occurrence of 1014 F kdr mutation. The frequency of genotypes of tested mosquitoes on each site depicted in (Table 9) the allelic and genotypic Geno typic frequency of kdr (Table 10).
Discussion
Control of Anopheline mosquito vectors of malaria by the use of insecticides has been shown to have a significant impact on both morbidity and mortality of this disease. This positive impact can, under certain circumstances, be compromised. Some of these events that lead to a negative, or less than desirable, impact are poor coverage, application with sub-lethal dosages and development of resistance by the vectors to the applied insecticides. Evidence of insecticide resistance in different settings necessitates surveillance studies to allow prompt detection of resistance should it arise and thus enable its management [10]. Concurrently, studies on the prevalence, distribution and relative densities of the vectors are very essential.
The results obtained in this study, regarding the Anopheline species composition showed that only An. arabiensis prevailed in the study area during the observation period where a total of 6400 Anopheles larvae were collected from the three sites from different types of breeding places and were all identified as An. Gambia complex previous studies done on River Nile State showed that An. Arabiensis is only members of the Gambia complex present in that area [11]. Accordingly, we considered that Anopheles species found on all Anopheles arabiensis. Based on WHO criteria for characterizing insecticide resistance susceptibility, where susceptibility is defined by Mortality rates between 98%- 100% indicate full susceptibility. Mortality rates between 90%- 97% require further investigation. Mortality rates < 90%, the population is considered resistant to the tested insecticides [7]. The findings of this study showed that an arabiensis from the River Nile State was resistant to Permethrin 0.75% in Shendi, Albawga and Alzidab Localities.
However, An. arabiensis was full susceptible to Bendiocarb 0. 1%, in all three sites with recorded 100% mortality rates. Bendiocarb was not used for vector control in River Nile state and that may explain the sensitivity to this insecticide. The Gambia Control Project was extended to River Nile State in 2001 The larvicide Temephos was used; concurrently, Malathion was first used, followed by Deltamethrinin 2003 up to now. Deltamethrin 0.05%was resistance in Shendi (80%) while it was susceptible in Al bawgaand Alzidab. The low efficacy of Permethrin in all sites can be explained in terms of the extensive domestic and agricultural use of this pesticide in the locality and used this insecticide in the long time for control major. Also, the high resistance to DDT can be explained by the long use of DDT especially in the agricultural sector.
The first record for DDT resistance in Sudan was from El Gunaid sugar cane area in 1970 [12]. This is in agreement with studies done in Central Sudan in Gezira and Sinnar States [9,13]. Similar study was carried out in 2007 in the Northern State, which neighbouring the River Nile State, against eight insecticides three Pyrethroids (Deltamethrin, Permethrin, Lambda cyhalothrin),Organochlorines (DDT), two Organophosphates (Malathion and Fenitrothion) and two Carbamates (Bendiocarb , Propoxur) and the results showed no evidence of resistance in An. arabiensis to seven out of eight insecticides tested [14]. Another study carried out in Khartoum town, which bordering the state from the south, in 2007 showed that: Anarabiensis was susceptible to (Bendiocarb, Propoxur ,Fenitrothion ,Deltamethrin and Lambda-Cyhalothrin) with the exception of suspected resistance to DDT (total mortality rate 96.9%) and Permethrin (97.4%), and confirmed resistance to Malathion (69.1%) [14].
Agricultural use of pesticides considered to be one of the major factors of insecticide resistance in malaria vectors. Considering the expansion of urban agriculture, which is often resulted to intensive use of regulated insecticide which, had serious impact on the ecology and resistance levels in An. arabiensis population [14-16]. The KDT50 and KDT95 values are usually considered as indicators for imminent development of resistance to insecticides and can be considered as base-line data for future observations on the development of resistance by the vector to the insecticides used in the state.
The results obtained in this study have serious implications for the malaria control programmes not only in River Nile State, but also the national level, since the control depends largely on the use of pyrethroid insecticides for indoor residual spraying and for insecticide-treated bed nets. At present only pyrethroids are used in the treating of bed nets. It is not known however, how this finding will affect the impact of ITNs since killing the vectors is not the only method of protection with this strategy.
Conclusion
It can be concluded that; An arabiensis in River Nile State is only susceptible to Bendiocarb with recorded 100% mortality ratesin all three sites of study: Shendi , Al zidab, Al bawga, while Deltamethrin was reported resistance in Shendi with mortality rate (80s%) and susceptible in Al damar and Barbar localities. In addition, evidence of insecticides resistance to DDT and Permethrin was observed in all three site shedi, Al bawga and Al zidab. The current study suggested that the Gambiae Control Project could adopt using Deltamethrin in the River Nile State except in Shendi area. Hence, further coordination is needed between integrated vector control and management of plant protection and pesticides for better management of insecticide resistance.
References
- Kamau, Vulule (2006) Status of insecticide susceptibility in Anopheles arabiensis from Mwewa rice irrigation scheme, Central Kenya. Malaria Journal 5: 46.
- Abdalla H, Matambo TS, Koekemoer LL, Mnzava AP, Hunt RH, et al. (2008) Insecticide susceptibility and vector status of natural populations of Anopheles arabiensis from Sudan. Trans R Soc Trop Med Hyg (102)3: 263-271.
- Abdel Nur OM, Nugud AD (1994) Key of identification of mosquitoes. Department of Medical Entomology. National Health Laboratory, Ministry of Health, Sudan.
- Ageep TB, Cox J, Hassan MM, Knols BG, Benedict MQ, et al. (2009) Spatial and temporal distribution of the malaria mosquito Anopheles arabiensisin northern Sudan: influence of environmental factors and implications for vector control. Malar J (8): 123.
- Azrag RS (2008) Microsatellite and mitochondrial genetic differentiation of Anopheles arabiensis Patton (Diptera: Culicidae) along the river Nile. University of Khartoum.
- Collins FH, Mendez MA, Rasmussen MO, Mehaffey PC, Besansky NJ, et al. (1987) A ribosomal RNA gene probe differentiates member species of Anopheles gambiae Am J Trop Med Hyg 37(1): 37-41.
- Elsyed EM (2013) Resistance of arabiensis Patton Mosquito to DDT, Fenitrothion, Deltamethrin, Permethrin and Bendiocarb in Shendi, Aldamar, Atbara and Abu Hamad localities, River Nile State, Sudan. Partial dissertation of Master of Science. University of Gezira.
- Haridi AM (1972a) Partial exophily of Anopheles gambiae B in Khashm Elgirba area in eastern Sudan. Bulletin of the World Health Organization 46(1): 39-46.
- Himeidan YE, Dukeen MY, El-Rayah AI (2004) Anopheles arabiensis: abundance and insecticide resistance in an irrigated area of eastern Sudan. East Mediterranean 10(1-2): 167-174.
- Malik EM, Mohamed TA, Elmardi KA, Mowien MM, Elhassan AH, et al. (2006) From chloroquine to artemisinin-based combination therapy: The Sudanese experience. Malar J (5): 65.
- Matambo TS, Abdalla H, Brooke BD (2007) Insecticide resistance in Anopheles arabiensis and association with the kdr mutation. Medical and Veterinary Entomology 21: 97-102.
- Ranson, H, Jensen, B, Vulule JM, Wang X, Hemingway J (2000) Identification of a novel mutation in the voltage-gated sodium channel gene of Anopheles gambiae associated with resistance to pyrethroid insecticides. Insect Molecular Biology 9: 491-497.
- Rose RI (2001) Pesticides and public health: integrated methods of mosquito management. Emerg Infect Dis 7: 17-23.
- Seidahmed OM (2007) Insecticides Susceptibility Status of the Malaria Vector Anopheles arabiensis Patton at the African Continental Fringe in Sudan. Department of Medical Entomology. Notional health laboratory, Sudan (unpublished report).
- WHO (2014) World Malaria Report? World Health Organization.
- WHO (2013) Test procedures for insecticide resistance monitoring in malaria vector mosquitoes? Geneva, World Health Organization.






























