Isolation and Identification of Staphylococcus aureus from Dairy Farms in Bishoftu Town, Ethiopia
Ararsa Duguma*, Abebe Wirtu1, Fufa Abunna1 and Ararsa Duguma2
1Addis Ababa University, College of Veterinary Medicine and Agriculture, Ethiopia
2Haramaya University, College of Veterinary Medicine, Ethiopia
Submission: February 23, 2018; Published: March 20, 2018
*Corresponding author: College of Veterinary Medicine, PO Box 138, Ethiopia, Tel: 251926260484; E-mail: ararsad@jfahoo.com
How to cite this article: Ararsa Duguma, Abebe Wirtu, Fufa Abunna, Ararsa Duguma. Isolation and Identification of Staphylococcus aureus from Dairy Farms in Bishoftu Town, Ethiopia. JOJ Pub Health. 2018; 3(1): 555604. DOI: 10.19080/JOJPH.2018.03.555604.
Abstract
Across-sectional study was conducted from November, 2014 to April, 2015 in selected dairy farms in Bishoftu Town to isolate and identify Staphylococcus aureus from milk samples of mastitis positive cows. The samples were taken from CMT positive cows and, isolation and identification of an organism was based on morphological, cultural and biochemical characteristics. A total of 46 lactating cows and 128 quarters were tested for mastitis using CMT, of which 13 (28.26%) sub-clinical mastitis at cow level and 21(16.40%) was at quarter level. Those CMT positive milk samples were collected and cultured parallel on both Blood agar and Nutrient agar. Out of the total 21 sub-clinical mastitis 18 samples were well grown on both Medias and sub cultured on nutrient agar for farther identification using Primary and secondary biochemical tests. Accordingly, 16 milk samples were shown typical yellow pigmentation on mannitol salt agar which is selective media for genus Staphylococcus. Finally, 9(42.85%) Staphylococcus aureus were identified on purple agar with the result of deep yellow pigmentation. Hence, implementing hygiene conditions, creation of awareness on control and prevention of subclinical mastitis in dairy farms and conducting drug sensitivity test for Staphylococcus aureus is recommended.
Keywords: Bovine; Subclinical mastitis; CMT; Staphylococcus aureus isolation; Bishoftu "
Introduction
In Ethiopia livestock represents a major national resource and form an integral part of the agricultural production system. The country has the largest livestock population of any African country with estimated 52.13 million and dairy cows are representing around 7.2 million [1].
Milkproduced from these animals provides an importantdietary source for the majority of rural as well asconsiderable number of the urban and per-urbanpopulation. However; milk production often does not satisfy the countries requirement, due to a multitude of factors, out of which disease of the mammary glands known as mastitis is among the various factors contributing to reduced milk production. According to thereports of [2], the total annual national milkproduction in Ethiopia ranges from 797, 9000 to 1,197,500 metric tons raw milk equivalents. Out of the totalnational milk production, between 85 and 89 percent iscontributed from cattle. However, this amount is by farbelow the national demand for milk and milk products inthe country, given the considerable potential for dairy farms income and employment generation from highvalue dairy products.
Development of the dairy sector in Ethiopia can contribute significantly to poverty alleviation and nutrition in the country [3]. Nevertheless, the quality and quantity of milk in the country deteriorates due to various causes. Mastitis imposes a serious problem cause reduction in milk production and remains one of the most economically important diseases for the dairy industry worldwide irrespective of the species of animals [4,5]. Mastitis can cause devastating effects to farmers because of the serious economic losses and the danger that the bacterial contamination of milk from affected cows may render it unsuitable for human consumption [6]. Mastitis, inflammation of the mammary gland, is a highly prevalent problem in dairy cattle and is one of the most important threats affecting the world's dairy industry [7]. Disease of Bovine mastitis can be clinical or subclinical. Clinical cases of mastitis are characterized by the presence of one or more of symptoms such as abnormal milk, udder swelling and systemic signs including elevated temperature, lethargy and anorexia, and sub clinical mastitis are those in which milk production decreases and lowered milk quality [8].
Once cows become infected they pose significant problems. Staphylococcal mastitis is the commonest and economically the greatest concern wherever dairy farming is practiced. Staphylococcus aureus is the etiological agent more commonly associated to the disease and is normally related to both subclinical and chronic infections leading to severe economic loss to dairy farms [9]. This disease, has received little attention in Ethiopia, especially the sub clinical form which is mainly caused by S. aureus [10,11]. Efforts have only been concentrated on the treatment of clinical cases. Therefore, the objective of this paper was to isolate and identify Staphylococcus aureus from mastitic cows in Bishoftu town.
Materials and Methods
Study Area
The study was conducted in dairy farms found in Bishoftu Town, which is located in 45 km along South East of Addis Ababa. The area is located at 9°N latitude and 40°E longitude at altitude of 1850 meter above sea level with annual rain fall of 866 mm, of which 84% is in the long rainy season June to September (Bishoftu town administrative office, 2010). The study animals were cross breeds cattle from dairy farms in Bishoftu Town.
Study Design
A cross-sectional study was conducted from November 2014, to April, 2015 in Bishoftu town, to isolate and identify Staphylococcus aureus, the major bacterial causative agent of bovine mastitis. CMT test was primary conducted for screening of positive samples and microbiological examination from strong positive CMT result samples were analyzed in the laboratory.
Milk Sample Collection and Storage
Milk samples were collected according to the after a quarter had been washed with tap water and dried the teat end was swabbed with cotton soaked in70% alcohol [12]. Thus strict aseptic procedures were adopted when collecting milk samples in order to prevent contamination with microorganisms present on the body of animal and from the barn environment. A Total 128 milk samples were collected from 46 sub clinical mastitis of cows at quarter level. Approximately 10 ml of milk samples were collected into sterile universal bottles after discarding the first three milking streams. Milk sampling and screening will be performed for each quarter of the sample taken. Collected samples were transported using ice-box to Addis Ababa University, College of Veterinary Medicine and Agriculture, Microbiology Laboratory. As soon as arrival to the laboratory it stored at 4°C for a maximum of 24 h until cultured on standard bacteriological media [13].
California Mastitis Test
The California Mastitis Test (CMT) was conducted to diagnose the presence of subclinical mastitis and it was carried out according to procedures given by [14]. A squirt of milk from each quarter of the udder was placed in each of four shallow cups in the California Mastitis Test paddle and an equal amount of the California Mastitis Test reagent was added. A gentle circular motion was applied in a horizontal plane. Positive samples show gel formation within a few seconds. The result was scored based on the gel formation and categorized as negative if there was no gel formation, or positive if there was gel formation. If at least one quarter was positive by the California Mastitis Test, then the cow was considered positive. Therefore, a cow was considered mastitic if one or more quarters were CMT positive with or without isolation of microorganisms [15].
Bacteriological Examination
The refrigerated samples were thawed by allowing equilibrating to room temperature for not less than 30 minutes before inoculation of the culture media. A loopful of each mastitis milk sample was streaked on nutrient and sheep blood agar containing 7% heparinized sheep whole blood. Bacteriological examination was done according to the [14]. Both Nutrient Agar and Blood agar plates streaked with the milk sample were incubated aerobically at 370C and examined for growth after 24 - 48h. Staphylococcusspp. colonies are usually white, opaque and up to 4mm in diameter. The colony of bovine strain of S.aureus was golden yellow. S.aureus and S. intermedius usually produce both alpha (α) and beta (β) haemolysin. Primary isolation and identification of the bacteria were made after sub-culturing of those colony which shows the same growth on both blood agar and nutrient agar to examine clear colony morphology (size, shape and colour) of the colonies, presence or absence of haemolysis or type of haemolysis on blood agar, gram's staining reaction, 3%KOH test, catalase test, oxidative fermentative test and oxidase test. Known genera were also sub-cultured on selective and differential media such as Mannitol salt agar and those grown on manitol salt agar and shown yellow pigmentation were sub cultured on nutrient agar for coagulase test.
The tube coagulase test was conducted by 0.5 ml of rabbit plasma in a small (5ml) test tube and an equal 0.5ml of an overnight broth culture of the Staphylococcus mixed and the tube was rotated gently to mix the contents and then incubated at 370C and examined every 2-4 hours to seen the clumping or clot formation.
Results
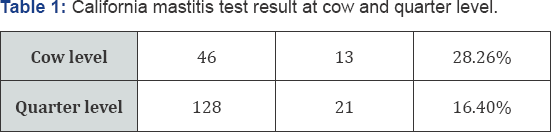
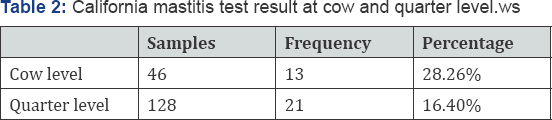
From a total of 46 lactating cows and 128 teat quarters examined, 13(28.26%) and 21(16.40%) were showed CMT positive at cow and quarter level respectively (Table 1). CMT positive samples were cultured parallel on both blood agar and Nutrient agar at 37 0c for 18 to 24 hrs. On Nutrient agar they tend to be white, circular, entire convex colonies. On Blood Agar Staphylococcus aureus shows hemolysis of the agar in the area around the colony. Their morphology has shown grape like characteristics during grams staining. Out of the total 21 sub- clinical mastitis 18 samples were well grown on both Media and sub cultured on nutrient agar for further identification using Primary and secondary biochemical tests. Accordingly, 76.2% (16/21) milk samples were shown typical yellow pigmentation on mannitol salt agar which is selective media for genus Staphylococcus. A 14 isolates of Staphylococcus coagulase positives in rabbit plasma has shown clumping and cultured on purple agar. Finally, 42.85% (9/21) Staphylococcus aureus were identified on purple agar by observing result of deep yellow pigmentation (Tables 2,3).

Discussion
Mastitis is a complex disease commonly caused by bacteria and the difference in results could be due to variations in herd size, management practices, proportion of exotic gene inheritance, agro-climates and other risk factors might have contributions to the observed differences in proportion of mastitis among the findings of other scholars. The present study was undertaken to investigate Staphylococcus aureus from sub- clinical mastitis in selected dairy farms of Bishoftu town. The CMT is of great value in attracting attention to sub clinically infected quarter which may show no clinical abnormality. CMT positive milk sample result was taken for isolation and identification of Staphylococcus aureus. CMT screening result showed that from a total of 128 milk samples at quarter level and 46 at cow level, 21 (16.40%) and 13(28.26%) were CMT positive respectively. The present report is lower than findings of dfferent scholars from different areas of Ethiopia [10,16-18]. This variation will be due to difference in herd size, management practices, genetic difference and enviromental factors. From bacteriologically positive samples 76.2% (16/21) were shown typical yellow pigmentation on mannitol salt agar which is selective media for genus Staphylococcus. The present isolates of Staphylococcus spp. was higher than the previous report of Addisalem, et al. [19] and Wubishet, et al. [18], which was 31.19 and 36.9% respectively. From further laboratory confirmation 9(42.85%) isolates of staphylococcus aureus were identified. The predominant or high proportion of Staphylococcus aureus in the present study was supported by the finding of [11,10]. The result of this study is lower than the findings of Kerro, et al. [20] who reports 40% at cow and 16% at quarter level. High prevalence of Staphylococcus aureus is due to its contagious nature and has been adapted to survive in the udder and establish chronic and subclinical infections. From there it is shed into the milk, which serves as a source of infection for healthy cows during the milking process [21].
Conclusion
The overall high prevalence of mastitis in the present study signifies the effect of mastitis in Bishoftu dairy farms. Staphylococcus aureus was mastitis inducing pathogens detected from subclinical mastitis in this study. Primary and secondary bacterial identification tests were employed for isolation and identification of confirms that high isolation rate showed little attention and invisible loss from subclinical mastitis. Therefore, implementing hygiene conditions and creation of awareness should be considered to reduce and prevent subclinical mastitis in dairy farms including future study on determination of sensitivity test for Staphylococcus aureus to available drugs to select an appropriate effective one in treating the cases.
Acknowledgment
The authors would like to thank Addis Ababa University, College of veterinary medicine and Agriculture for financial, logistic and other material support. Farm owners of the study area would well acknowledged for their voluntary participation and supports during sample collection.
References
- Central Statistical Agency (CSA) (2012) Livestock and Livestock Characteristics Agricultural Sample Survey. Statistician Bulletin 2: 215-245.
- FAO (2003) The Technology of Traditional Milk Production in Developing Country. Animal Production and Health Paper 85: 9-24.
- Mohamed AM, AE Simeon, A Yemesrach (2004) Dairy development in Ethiopia. International Food Policy Research Institute, EPTD Discussion, Washington, USA, pp.123.
- Korhonen H, L Kaartinen (1995) Changes in the composition of milk induced by mastits. In The bovine udder and mastitis. Sandholm MT Honkanen mammary gland Vet Res 37: 369-400.
- Bradley AJ (2002) Bovine Mastitis An Evolving disease. Vet J 163: 1-13.
- Quinn PJ, Markey BK, Carter ME, Donelly WJ, Leonard FC (2002) Veterinary Microbiology and Microbial Disease. Blackwell Science Ltd a Blackwell Publishing Company 465- 475.
- Wallenberg GJ, JT Vanior (2002) Viral infection and bovine mastitis. J Vet Microbiol 88: 27-45.
- Bradley AJ, Green MJ (2001) Aetiology of clinical mastitis in six Somerset dairy herds. Vet Rec 148: 683-686.
- Kubota M, T Hayashi, K Iwasaki, H Ohtsuka, M Kohiruimaki (2007) Rapid and Effective Method for Separation of Staphylococcus aureus from Somatic Cells in mastitis milk. J Dairy Sci 90: 4100- 4107
- Hundera S, Z Ademe, A Sintayehu (2005) Dairy cattle mastitis in and around Sebeta, Ethiopia. Intern J Appl Vet Med 3(4): 1525-1530.
- Mekonnen H, S Workineh, M Bayleyegn, A Moges, K Taddele (2005) Antimicrobial susceptibility profile of mastitis isolates from cows in three major Ethiopian dairies. Med Vet 176: 391-394.
- National Mastitis Council (NMC) (1990) Microbiological procedures for the diagnosis of bovine udder infection (3ri edn.); Madison Wisconsin National Mastitis Council Inc p.7-8.
- Quinn PJ, ME Carter, B Markey, GR Carter (1994) Clinical Veterinary Microbiology. Mosby Publishing London 327-344.?
- Quinn PJ, ME Carter, B Markey, GR Carter (2004) Clinical Veterinary Microbiology. Mosby Publishing London 43-55.
- Abera M, Demie B, Aragaw K, Regassa F, Regassa A (2010) Isolation and identification of Staphylococcus aureus from bovine mastitic milk and their drug resistance patterns in Adama town, Ethiopia. J Vet Med Animal Health 2(3): 29-34.
- Lakew M, Tolosa T, Tigre W (2009) Prevalence and major bacterial causes of bovine mastitis in Asella South Eastern Ethiopia. Trop Anim Health Prod 41: 1525-1530.
- Mekibib B, Furgassa M, Abuna F, Megersa B, Regassa A (2010) Bovine mastitis prevalence risk factors and major pathogens in dairy farms of Holeta town Central Ethiopia. Veter World 9: 397- 403.
- Wubishet Z, Ararsa D, Alemayehu L (2013) Bovine Mastitis in selected districts of Borena zone, southern Ethiopia Bull. Anim Hlth Prod Afr 61: 285-291.
- Addisalem T, Mersha C (2012) Study on the Occurrence of Bovine Mastitis in Addis Ababa Dairy Farms and Associated Risk Factors. Advan Biol Res 6: 151-158.
- Kerro O, Tarekegn F (2003) Bovine mastitis in Veterinary Medicine, School of Graduate Studies.selected areas of Southern Ethiopia. J Tropi Ani Hlth and Prod 197-205.
- Radostits M, Gay C, Hinchelitt W, Constable D (2007) Veterinary Medicine A Text Book of the Disease of cattle Horses Sheep Pigs and Goats. (10th edn.); Grafos, SA Arte Sobre Papel Spain, pp. 823-835.






























