Behavioral and Metabolic Risk Factors for Cardiovascular Disease among the School Adolescents of Nepal
Manita Pyakurel1*, Anup Ghimire2, Paras Pokharel2, Rishi Bhatta3 and Ramesh Parajuli4
1Department of Community Medicine, Nepalgunj Medical College, Nepal
2Department of Community medicince, BPKIHS,Nepal
3Department of ENT, Nepalgunj Medical College, Nepal
4Department of ENT, Chitwan Medical College, Nepal
Submission: March 20, 2017; Published: March 30, 2017
*Corresponding author: Manita Pyakurel, Lecturer, Dept of Community Medicine, Nepalgunj Medical College, Nepal,Email: bhattamanita@gmail.com
How to cite this article: Manita P, Anup G, Paras P, Rishi B, Ramesh P. Behavioral and Metabolic Risk Factors for Cardiovascular Disease among the School Adolescents of Nepal. JOJ Pub Health. 2017; 1(3): 555565. DOI:10.19080/JOJPH.2017.01.555565
Abstract
Background: Cardiovascular disease (CVD) is increasing in European and Asian population. Prevalence of cardiovascular risk factors in adolescents is not estimated in Nepal.
Objectives: The aim of this study was to find out the prevalence of Behavioral and metabolic risk factors.
Methods: A cross sectional study was done among 736 school going adolescents. A Systematic random sampling was done to select the sampling unit. Blood Pressure, waist circumference measured as an anthropometric measurement. Biochemical tests included fasting blood sugar (FBS), triglyceride (TG) and high density lipoprotein (HDL). MS was defined based on National Cholesterol Education Programme (NCEP, 2003) criteria. Analysis was done using SPSS 17 version.
Results: At least one risk factor was present in 567(76.9%). Unprotective HDL was the commonest metabolic risk factors. Significant association was shown between stress and MS among females. Family history found to be significantly associated with MS among Males.
Conclusion: Metabolic risk factors are prevalent in this age group. This suggests the need for preventive actions to reduce future burden of CVD. Tracking and correction of modifiable risk factors help for early detection and primary prevention of CVD.
Keywords: Hypertension; Metabolic syndrome; Obesity
Introduction
Non communicable diseases (NCDs) are slowly progressing diseases. The four common NCDs are cardiovascular diseases, cancers, chronic respiratory diseases and diabetes mellitus. NCDs occur in all age groups [1]. Globally, NCDs are attributed to twenty nine million deaths annually. Distribution of various risk factors associated with high mortality rates are high blood pressure (16.5%) and high blood glucose (6%). Among the behavioral risk factors, tobacco use (9%), lacking physical activity (6%) and overweight and obesity (5%) are the major risk factors for global mortalities [2,3]. Notably, there are two types of risk factors for NCDs; modifiable behavioral and metabolic risk factors. Modifiable behavioral risk factors include tobacco use, harmful use of alcohol, unhealthy diet and physical inactivity. These behavioral risk factors lead to metabolic changes like raised blood pressure, overweight and obesity, hyperglycemia and hyperlipidemia [1].
In South Asia, over the centuries a great transition has resulted in major changes in availability of food, lifestyle, physical activity, and dietary habit. Dietary acculturation is associated with higher prevalence of MS in South Asians [2]. Nepal is facing a surge in the burden of NCDs which is similar to other developing nations of South East Asia [4]. In Nepal, the first NCDs risk factors’ study revealed high prevalence of behavioral risk factors [5]. Similarly, metabolic risk factors are also found in an increasing trend in Nepal due to lack of knowledge for physical activity, presence of hereditary factors and obesity [6]. Therefore, the risk factors of NCDs are prevalent among general population and also applicable for young population.
Adolescent age is defined as age from 10 to 19 years. Adolescent age is appropriate age for tracking CVD as there are evidences of formation of atherosclerosis at an early age [7-10]. The growing interest in relationship between behavioral risk factors and metabolic risk factors and the lack of prior research findings on cardiovascular risk factors in this age group of Nepal, implies a need to study about cardiovascular risk factors among adolescents. Tracking of NCDs at an early age is very important to prevent their occurrence at a later age [11]. Thus, the aim of this study is to assess the prevalence of behavioral, genetic and metabolic risk factors for CVDs among the adolescents in Nepal.
Materials and Methods
Study Population
A school based cross sectional study was conducted among 736 adolescents in Nepalgunj municipality of Banke district, from September 2014 to February 2015. Out of 7 public and 4 private schools, 4 public and 2 private schools comprising 50% from each group were selected using simple random sampling. The number of required participants was calculated using probability proportional to size among all the schools. Out of 760 students, 5 were excluded because they did not meet the inclusion criteria. Out of the 755 participants, 6 were absent from the school, 12 were not fasting and 1 was not fit for blood test.
Behavioral Factors
We used WHO STEPS questionnaire for data collection after adjusting it according to the local setting of Nepal [12]. Pretesting was done in 10% of estimated sample in a similar population and modifications were done accordingly. Dietary habits comprised of; salt intake, fruits consumption, vegetables intake and fast food intake per week. Minimum required score was calculated for all four parts and added. The total score was 23. Score equal to or more than 23 was coded as 0(healthy group) and less than 23 as 1(unhealthy group). Physical activities were classified as heavy/moderate/low physical work and exercise for entertainment hours per week walk/cycle per week, sleep hours and rest hour per week. Minimum required score was calculated for all four parts and added. The total score was 98 for physical activity. Score equal to or more than 98 was coded as 0(physically active) and less than 98 as 1(physically inactive). Sleep hours was defined by National heart lung and blood institute [13-15]. Stress was defined as stress at any point of time in life which caused disturbance(s) in sleep.
Genetic factors
The questionnaire contained questions about family history of diabetes mellitus, hypertension, and congenital heart diseases.
Anthropometric Measurements
Blood pressure was measured with standard mercury sphygmomanometer with adequate cuff size. Systolic blood pressure was taken by first heart sound (kortokoff phase I) anddiastolic pressure was recorded at level when sound disappeared (kortokoff phase V).Two readings were taken on right arm in the interval of at least five minutes. Waist circumference was measured using a non-elastic measuring tape with calibration of one millimeter as minimum over unclothed abdomen at smallest diameter between coastal margin& iliac crest. Tape measure was horizontal. Respondents were relaxed with arms held loosely by the side. Measurements were taken at the end of normal expiration. Both arterial hypertension and abdominal obesity were categorized according to IDF criteria [16].
Biochemical tests
Five milliliters of venous blood sample was drawn by sterile disposable syringe. Blood samples were collected after the participants fasted overnight for about 10-12 hours and brought to the biochemistry laboratory of Nepalgunj medical college within half an hour. Automated biochemistry analyzer (BS-380, Mindray) was used to analyze the serum levels of triglyceride (TG), high density lipoprotein cholesterol (HDL-C) and fasting blood sugar.
Diagnostic criteria
MS was categorized as positive and negative according to IDF criteria i.e. among the five metabolic risk factors: obesity, hypertension, increased FBS, increased TG and decreased HDL. Presence of three MRFs was known as positive for MS.
Statistical Analysis
Data was analyzed using SPSS 18(SPSS Inc. ChicagoIL). Between the two categorical data (socio demographic variables and MS, behavioral risk factors and MS, genetic risk factors and MS) Pearson’s chi square was applied. Results with p value <0.05 was considered statistically significant. Variables with p value <0.2 in chi square test were analyzed in binary logistic regression presented with beta coefficient with 95% CI.
Ethics statement
Study was approved by the institutional review board of Nepalgunj Medical College. Informed written consent was taken from the legal guardians or teachers of the participants. Written permissions were taken from selected schools.
Results
Mean age of the study participant was 15.22 ±1.79 years. Major religion was Hindu (83.8%) and major ethnic group was of upper caste (30.4%) (Table 1). Waist circumference was significantly larger (P<0.05) among the adolescent with MS as compared with those without MS. Mean SBP, DBP, TG and FBS were significantly higher (p<0.05) among those with MS as compared those without MS. No statistical differences were noted in mean HDL-cholesterol between the two groups. Among the behavioral risk factors, adolescents who consumed tobacco were 62(8.4%). Cigarette was the mostly used tobacco item 41(66.1%) Most of the cigarette smokers smoked filter cigarette 53(85.4%): non-filter cigarette users were 9(14.6%). Consumption less than 5 cigarettes per day were 35(85.4%). The number of passive smokers was 61(8.3%). Alcohol consumers were 51(6.9%). Most of the alcohol consumers consumed for less than 4years 34(66.7%) whereas 17(33.3%) consumed for ≥ 4 years. Beer was the mostly consumed beverage 32 (62.8%). Among the alcohol consumers, 25(49.02%) consumed sometimes in a year, whereas 21(41.2%) drank every month. Among the total alcohol consumers 51(6.9%), 27(52.9%) consumed alcohollessthan250mlperintake, whereas 24(47.06%) consumed more than 250 ml per intake.
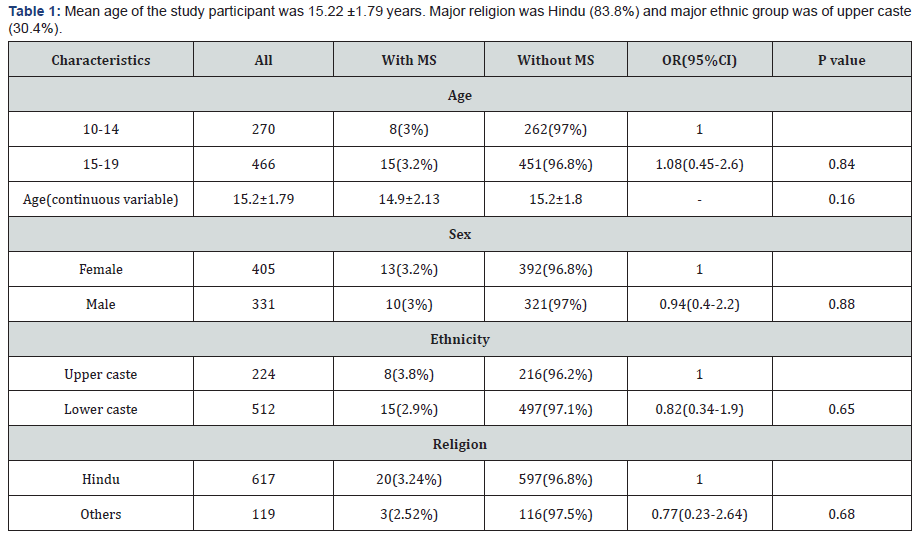
There were 726(98.6%) adolescents with unhealthy dietary habit. Non refined oil consumers were 264(35.9%) and nonvegetarian were 560(76.1%). Total 591(80.3%) were physically inactive adolescents. Also 345(46.9%) reported stress at any point of time. Among the genetic factors, family history of chronic diseases were present among 547(74.3%) and 14(1.9%) were diagnosed with congenital heart disease. Among the total 736 adolescents, 566 (76.9%) had at least one risk factor and 1(0.1%) had four risk factors. Low HDL was the most common metabolic risk factor. Prevalence of higher TG and adiposity were higher in females then males. Arterial hypertension was higher among males compared to females whereas high blood sugar was equal among both sexes (Figure 1).

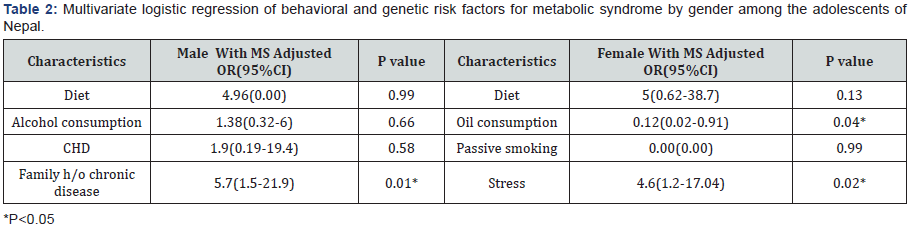
Bivariate analysis between socio demographic variable and MS shows significant association with p value >0.05.There is a significant association between stress and MS among females with OR of 4.7(95% CI, 1.27-17.28) and p value of <0.05. Also, there was a significant association between oil consumption and MS among females with OR of 0.12(95%CI, 0.02-0.94) and p value of 0.02.Among male adolescents’ families, history of chronic diseases was significantly associated with MS with OR of 6.66(95%CI, 1.82-24.35) and p value of 0.001. Result of multivariate analysis shows, male adolescents with positive family history has 5.7 times higher odds of (95% CI: 1.5-21.9) MS as compared to their counterparts with negative family history (Table 2). Among the female adolescents, non refined oil consumption showed 0.12 times protective odds of (95% CI: 0.02-0.91) MS as compared to the refined oil consumers. Whereas stressful female adolescents have 4.6 times higher odds of (95% CI: 1.08-16.4) MS compared to the non-stressful female.
Discussion
Prevalence of MS in this study is (3.1%.IDF) which is consistent with previous studies conducted among non-Hispanic adolescents of African origin in NHANES study in the United States(2.5%,ATP III), Kashmir (3.8%,ATP III), Jammu(2.6%,ATP III),two studies from China(6.6%-IDF,3.7%-US adolescent criteria) and Turkey (2.2%-ATP III) [8,17-22]. In our study MS is more prevalent in late adolescents with 2.03%, which is similar to Chinese and Indian adolescents [19,20]. Prevalence of MS was higher in females (1.7%) as compared to males (1.3%) which is supported by the study from urban India(female-22.3% vs. Male-16.6%) [23]. It is not affirmed by other studies having higher prevalence in males [8,22-24]. In our study, there is a higher prevalence of MS (1.4%) in disadvantaged minority groups called Janajati followed by upper caste (1%). MS differs according to race and ethnicity [25] The minority groups are more exposed to environmental factors than the advantaged groups of adolescents which lead to higher MS in minority group of adolescents [26,27].
Among the MRFs, the most common MRF was decreased high density lipoprotein (99%) which was similar to the study from Jammu, Kashmir and Teheran [17,20,28]. The extent of decreased HDL was found to be higher as compared with Indian, Turkish and Teherani (71%, 29%, and 41.6%) adolescents [8,23]. It shows the need for further study in the future for the evidence of HDL-C Dyslipidemia. Prevalence of increased blood sugar (3.8%) was least common and least prevalent in our study which was similar with the study from Teheran (1%). The prevalence of increased triglyceride in our study was 8.9% which was supported by the South Indian study (8.2%) [29]. But it was higher than the study from Jammu and Kashmir (3.4% and 3.8%) [17,20]. Likewise 7.2% prevalence of hypertension was consistent with the study from Jammu(2.7%) but in opposition to study the from Kashmir with no cases of hypertension [17,20]Prevalence of obesity (6.3%) was consistent with the study from Jammu India(5.6%) [20].
The prevalence of behavioral risk factors for CVD is increasing in Nepal [30,31]. Our study revealed relatively higher prevalence of unhealthy dietary habits (98.6%) compared to a study from Pakistan (64%) [32], which is not surprising because of the rapidly increasing sedentary lifestyle and increased availability of fast foods in Nepalgunj [33]. About one fourth of adolescents had positive family history which was comparable to Pakistani adolescents for Hypertension (23%) and (16%) for Diabetes [32]. Additionally 1.9% prevalence of verbally reported CHD was explored from the study which was inconsistent with the study from Kathmandu valley (0.13%) which suggests the need for further study [34]. In our study, there is no significant association between socio-demographic variables and MS. This is consistent with the study from US [18]. However a study from Saudi Arabia shows sex as a significant variable for MS [35]. This shows the need for further study on the relationship between socio-demographic profile and MS in Nepal. Among the genetic risk factors, significant association was found between family history and MS which is favored by studies from Beijing and Nanjing [36].
Our study found, oil consumption to be a protective significant predictor for MS which is not supported by Framingham nutrition study [37]. In addition, stress was found to be a significant predictor for MS which is similar to NHANES study [38]. This study defines stress as stress at any point of time, but it needs further quantification to explain the relation of stress with MS. Moreover, male adolescents with a positive family history was a significant predictor for MS; which was similar to the from the studies from Beijing, Nanjing and Kashmir [17,36]. Although each predictor can be managed to some extent, it is prudent to prevent the multiple factors. They should be treated in early life to promote the health from the childhood.
Limitations of the study
Limitations of this study include the biochemical test results data which were based on one measurement and limited dietary questionnaire for understanding dietary habit. The study result can only be generalized to similar type of population and lifestyle but can’t be generalized to other adolescents.
Conclusion
In our study, prevalent behavioral risk factors were consumption of non -refined oil, unhealthy dietary habit and physical inactivity. Among the genetic risk factors, prevalence of 1.9% CHD and positive association of family history with MS were alarming. More than three fourth of the adolescents have at least one risk factor of MS Dyslipidemia was the most common risk factor affecting the adolescents. Male with positive family history, non-refined oil consumer female were important risk factors for identifying adolescents at risk for later CVD onset. The result suggests that preventive measures, including consumption of refined oil may be warranted for these adolescents.
Acknowledgement
We thank all the participants of this study. Nepalgunj Medical College team is acknowledged for providing biochemistry laboratory facility. Professor Surya Niraula is acknowledged for statistical support.
References
- WHO (2013) Noncommunicable disease: fact sheet. Division of Global Health Protection, CDC, USA.
- Garduno Diaz S, Khokhar S (2013) South Asian dietary patterns and their association with risk factors for the metabolic syndrome. Journal of Human Nutrition and Dietetics 26(2): 145-155.
- Yusuf S, Reddy S, Ounpuu S, Anand S (2001) Global burden of cardiovascular diseases part II: variations in cardiovascular disease by specific ethnic groups and geographic regions and prevention strategies. Circulation 104(23): 2855-2864.
- Bhandari GP, Angdembe MR, Dhimal M, Neupane S, Bhusal C (2014) State of non-communicable diseases in Nepal. BMC public health 14: 23.
- Nepal S (2003) surviellance of risk factors for non communicable diseases in Nepal. Nepal Health Research Council, Government of Nepal, Nepal.
- Yadav K, Wagle R (2013) Knowledge and Attitude Regarding Major Risk Factors of Cardiovascular Diseases among 15-19 Year Old Students of Kathmandu District. Health Prospect 11: 7-10.
- Andersen LB, Wedderkopp N, Hansen H, Cooper A, Froberg K (2003) Biological cardiovascular risk factors cluster in Danish children and adolescents: the European Youth Heart Study. Preventive medicine 37(4): 363-367.
- Agirbasli M, Cakir S, Ozme S, Ciliv G (2006) Metabolic syndrome in Turkish children and adolescents. Metabolism 55(8): 1002-1006.
- Lambert M, Paradis G, O loughlin J, Delvin E, Hanley J, et al. (2004) Insulin resistance syndrome in a representative sample of children and adolescents from Quebec, Canada. International journal of obesity 28(7): 833-841.
- Wikibook (2013) Human physiology/Cardiovascular system.
- Population HNa (2011) Non-Communicable Diseases (Ncds)-Nepal’s Next Major Health Challenge. Development SAH, NCDs policy brief Nepal Bank TW, Nepal.
- WHO (2013) Chronic diseases and health surveillance. WHO Report, Geneva, Switzerland.
- WHA57 (2004) 17 Global strategy on diet, physical activity and health. WHO Report, Geneva, Switzerland, p. 38-55.
- WHO (2013) Diet, nutrition and the prevention of chronic diseases. Report of a Joint WHO/FAO Expert Consultation. Geneva, Switzerland, p. 23-25.
- National Heart L, and Blood Institute (2012) How much sleep is enough? US Department of health & Human Services, USA.
- Zimmet P, Alberti KGM, Kaufman F, Tajima N, Silink M, et al. (2007) The metabolic syndrome in children and adolescents an IDF consensus report. Pediatric diabetes 8(5): 299-306.
- Andrabi SM, Bhat MH, Andrabi SR, Kamili MM, Imran A, et al. (2013) Prevalence of metabolic syndrome in 8–18-year-old schoolgoingchildren of Srinagar city of Kashmir India. Indian journal of endocrinology and metabolism 17(1): 95-100.
- De Ferranti SD, Gauvreau K, Ludwig DS, Neufeld EJ, Newburger JW, et al. (2004) Prevalence of the metabolic syndrome in American adolescents findings from the Third National Health and Nutrition Examination Survey. Circulation 110(16): 2494-2497.
- Li Y, Yang X, Zhai F, Kok FJ, Zhao W, et al. (2008) Prevalence of the metabolic syndrome in Chinese adolescents. British journal of nutrition 99(3): 565-570.
- Singh N, Parihar RK, Saini G, Mohan SK, Sharma N, et al. (2013) Prevalence of metabolic syndrome in adolescents aged 10-18 years in Jammu, J and K. Indian journal of endocrinology and metabolism 17(1): 133-137.
- Xu H, Li Y, Liu A, Zhang Q, Hu X, et al. (2012) Prevalence of the metabolic syndrome among children from six cities of China. BMC public health 6(12): 13.
- Cook S, Weitzman M, Auinger P, Nguyen M, Dietz WH (2003) Prevalence of a metabolic syndrome phenotype in adolescents: findings from the third National Health and Nutrition Examination Survey, 1988-1994. Archives of pediatrics & adolescent medicine 157(8): 821-827.
- Tandon N, Garg M, Singh Y, Marwaha RK (2013) Prevalence of metabolic syndrome among urban Indian adolescents and its relation with insulin resistance (HOMA-IR). Journal of Pediatric Endocrinology and Metabolism 26(11-12): 1123-1130.
- Sarrafzadegan N, Gharipour M, Sadeghi M, Nouri F, Asgary S, et al. (2013) Differences in the prevalence of metabolic syndrome in boys and girls based on various definitions. ARYA atherosclerosis 9: 70. Arya Atherosclerosis 9(1): 70-76.
- Cappuccio FP (1997) Ethnicity and cardiovascular risk: variations in people of African ancestry and South Asian origin. Journal of human hypertension 11: 571-576.
- Prussian KH, Barksdale Brown DJ, Dieckmann J (2007) Racial and Ethnic Differences in the Presentation of Metabolic Syndrome. The Journal for Nurse Practitioners 3(4): 229-239.
- Kumanyika SK, Grier S (2006) Targeting interventions for ethnic minority and low-income populations. The Future of Children 16(1): 187-207.
- Afkhami Ardekani M, Zahedi Asl S, Rashidi M, Atifah M, Hosseinpanah F, et al. (2010) Incidence and Trend of a Metabolic Syndrome Phenotype Among Tehranian Adolescents Findings from the Tehran Lipid and Glucose Study, 1998-2001 to 2003-2006. Diabetes care 33(9): 2110- 2112.
- Vasan S, Thomas N, Christopher S, Geethanjali F, Paul T, et al. (2011) Anthropometric measurements for the prediction of the metabolic syndrome: a cross-sectional study on adolescents and young adults from southern india. Heart Asia 3(1): 2-7.
- WHO (2013) Non communicable diseases risk factor STEPS survey 2013 Nepal: fact sheet. Nepal Health Research Council, Nepal.
- Giri S, Sharma SR, Timalsina S, Yadav V, Koirala S, et al. (2012) Cardiovascular health risk behaviour among medical students in teaching hospital. J Nepal Health Res council 10(22): 187-191.
- Khuwaja A, Fatmi Z, Soomro W, Khuwaja N (2003) Risk factors for cardiovascular disease in school children-a pilot study. J Pak Med Assoc 53(9): 396-400.
- Subedi P (2001) Urbanization and development. Un Habitat, World cities Report, Kenya pp. 262.
- Bahadur K, Sharma D, Shrestha M, Gurung S, Rajbhandari S, et al. (2002) Prevalence of rheumatic and congenital heart disease inschoolchildren of Kathmandu valley in Nepal. Indian heart journal 55(6): 615-618.
- Mehairi AE, Khouri AA, Naqbi MM, Muhairi SJ, Maskari FA, et al. (2013) Metabolic syndrome among Emirati adolescents: a school-based study. PloS one 8(2): e56159.
- Hu B, Li W, Yang W, Tao C (2009) Multivariate analysis on the risk factors of metabolic syndrome in Beijing and Nanjing. Journal of Diabetes 1: A101.
- Millen BE, Pencina MJ, Kimokoti RW, Zhu L, Meigs JB, et al. (2006) Nutritional risk and the metabolic syndrome in women: opportunities for preventive intervention from the Framingham Nutrition Study. The American journal of clinical nutrition 84(2): 434-441.
- Kinder LS, Carnethon MR, Palaniappan LP, King AC, Fortmann SP (2004) Depression and the metabolic syndrome in young adults: findings from the Third National Health and Nutrition Examination Survey. Psychosomatic medicine 66(3): 316-322.






























