Negative Pressure Wound Therapy for Lower Limb Amputation Patients - A Case Control Trial
Brendan Chan1, David W Shields2, Helen Findlay2, Ashish Mahendra2 and Sanjay Gupta2*
1University of Glasgow Medical School, University Avenue, UK
2Department of Musculoskeletal Oncology, UK
Submission: April 28, 2018; Published: May 16, 2018
*Corresponding author: Sanjay Gupta, Department of Musculoskeletal Oncology, Glasgow Royal Infirmary, Scotland, UK, Email: sanjay.gupta@@ggc.nhs.uk
How to cite this article: B Chan, D W Shields, H Findlay, A Mahenda, S Gupta. Negative Pressure Wound Therapy for Lower Limb Amputation Patients - A Case Control Trial. JOJ Orthoped Ortho Surg. 2018; 1(4): 555569. DOI: 10.19080/JOJOOS.2018.01.555569
Abstract
Introduction: Limb amputation is a major life changing surgery used in many disciplines as a planned primary procedure or as a salvage procedure in tumours, trauma, and peripheral vascular disease. However, amputation carries a high rate of wound complications such as infection, dehiscence, and impaired healing. These complications may result in further surgical intervention, prolonged hospitalisation, and decreased quality of life for patients. Whilst the use of negative pressure wound therapy (NPWT) in the treatment of chronic wounds has demonstrated improved wound outcomes by increased wound healing, wound shrinkage, and decreasing wound oedema; literature for the use of NPWT in amputations is scarce.
Aim: To evaluate the wound healing outcomes of patients after lower limb amputation treated with and without the use of NPWT. Patients for the intervention group were chosen from an amputation database whom had received NPWT from 2015 to 2017. A control group was established by an independent investigator from a prospective amputation database whilst matching patients in the intervention group for age, gender, type of amputation and method of closure. Outcomes assessed were the presence of wound complications, additional surgical procedures, and drainage volumes.
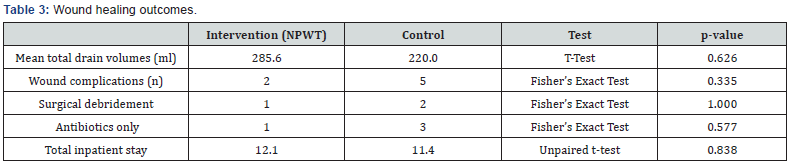
Results: Patients in both the intervention group (n=9) and control group (n=9) were well matched for demographics. The intervention group had a higher yield of mean drainage volumes compared to the control group of 285.6ml and 220.0ml respectively. Additionally, the intervention group demonstrated fewer patients with wound complications compared to the control group at 2 versus 5 patients respectively.
Conclusion: The data presented above demonstrates a positive trend in the reduction of wound complications with the use of NPWT. Despite being statistically insignificant, we believe that there is evidence to suggest the potential of NPWT in amputation therapy. Therefore, a prospective investigation with a larger sample size is warranted for further assessment of the effectiveness of NPWT as routine therapy for amputations.
Keywords: Negative pressure wound therapy; Wound healing; Amputation; Wound infection; Dressings; Sarcoma
Abbreviations: NPWT: Negative Pressure Wound Therapy; VAC: Vacuum Assisted Closure; WOLLF: Wound Management of Open Lower Limb Fractures
Introduction
Amputations are utilised for many indications, as planned primary procedures such as peripheral vascular disease or tumours, or as a salvage procedure for severe trauma or infections. Decisions to amputate in complex cases are usually a multidisciplinary decision with extensive patient and family counselling, preoperatively if possible [1,2]. In trauma, amputation may be considered as management in the presence of a high grade open fracture, severe vascular injury, significant nerve injury, or failed salvage. However due to the nature of trauma, other factors associated to the injury or patient’s physical condition may compound wound healing and amputation success. These factors include the presence of shock or compartment syndrome, extent of the injury, comorbidities, age, fluid status, and level of atherosclerosis [3].
Sarcomas, both bone and soft tissue, are typically treated with a wide surgical resection and adjuvant therapy [4-6]. This group of patients are subject to a higher risk of adverse wound outcomes due to the nature of their disease and adjuvant treatments received. Sarcomas have high mitotic and invasive activity that can interrupt wound healing by inducing a chronic inflammatory environment, delaying wound granulation and maturation [7,8]. Furthermore, the use of adjuvant chemotherapy and radiotherapy can cause damage to proliferative cells and healthy tissue, which causes an additional delay in wound healing [9,10]. Where the tumour is large and high grade, or where limb sparing surgery has failed or is not possible, an amputation may be the most appropriate treatment plan [11,12].
Amputations are life changing operations which are associated with a further increased risk of complications compared to limb sparing procedures and resections. In 2009, Harris et al quote 24.8% of lower limb amputations encounter wound complications by 3 months post-op [13]. In this study, the most common wound complications were infections (34.2%) and wound dehiscence (13.4%). Another study by Jensen et al in 1982 demonstrated 14% of above knee amputation patients suffered from wound complications, with 2% leading to further re-amputation. Comparatively, the same study showed that 40% of below knee amputation patients had wound complications, of which 28% required further re-amputation [14].
Over the last 15 years, negative pressure wound therapy (NPWT) has contributed to improved wound healing outcomes in surgical patients with the initial use to fill voids in chronic wounds. The mode of NPWT in our institute is the Vacuum Assisted Closure (VAC) (ActiV.A.C, KCI San Antonio, Texas). NPWT consists of a semi-occlusive dressing attached to a suction device to create a negative pressure environment thereby promoting wound healing [15]. This is achieved on a macroscopic and microscopic level. Macroscopically, the collapse of the pores of the foam or mesh dressing by 80% transfers centripetal forces to the wound surface causing wound shrinkage [16,17]. This reduces the wound site to sub atmospheric pressure, assisting in the removal of excess fluid in the wound, creating further compression on the wound site and minimising potential deadspace within the wound [18]. Microscopically, NPWT induces undulation of the wound surface which transmits shear and hydrostatic forces to the individual cells via the extracellular fluid and matrix [19]. This provides alteration of the cell shape to promote cell proliferation, migration and differentiation [20]. Additionally, NPWT can assist in optimising neuropeptide and nerve fibre growth in wound healing [21]. It has also been hypothesised that NPWT can reduce bacterial wound infections, however there is insufficient evidence to support this.
Since its advent in 1990, NPWT has been primarily used for tissue loss and open wounds [22]. However, there is an emerging body of evidence indicating the benefit of NPWT in closed wound treatment. In 2006, Stannard et al demonstrated a reduction in wound healing time with drainage of haematomas to 1.6 days from 3.1 days with use of NPWT, and a further reduction in rates of infection of wound sites from 16% to 8% [23]. The same study investigated the use of negative pressure in high risk lower extremity fractures to show a statistically significant reduction in the time taken for the wound to dry from 4.8 days compared to 1.8 days [23]. Another study by Stannard et al in 2012 demonstrated a 1.9 times increase in the risk of wound infection in patients not using NPWT [24].
The use of NPWT in severe open fractures was evaluated by Stannard et al in 2009, quoting that patients on NPWT were only one-fifth as likely to develop deep infections as compared to those without [25]. Presently, the WOLLF trial aims to evaluate the efficacy of NPWT in a pragmatic multicentre randomised controlled trial with patient reported outcomes as their primary outcome measure [26]. A pilot study conducted within our institution by Kong et al showed a clinically and statistically significant improvement in wound complication rates when used following sarcoma resection. Our study demonstrated a lower mean drain volume and a lower proportion of wound complications in the NPWT group [27]. Currently, there is evidence for the benefit of NPWT in reducing wound complications, and for its use in trauma and elective surgery. However, literature on the benefits of NPWT in amputation surgery is scarce.
Aim
We aim to evaluate the wound healing outcomes of sarcoma patients after lower limb amputation treated with and without the use of negative pressure wound therapy.
Method
A case controlled study was conducted on a cohort of predominantly sarcoma patients that received NPWT as routine therapy for amputations between 2015 and 2017. An intervention group containing 9 consecutive patients that received NPWT were matched from a database against a control group of 9 patients who did not receive NPWT. The intervention group was matched on patient age at operation, gender, type of amputation, and method of closure. The control group was constructed at random by an independent investigator from a prospective amputation database. Patients from both groups were grouped by their amputation indication of being sarcoma or non-sarcoma. The non-sarcoma group contained patients with wound breakdown, trauma with secondary osteomyelitis, and bone loss.
Outcomes assessed were; wound complications, additional surgical procedures due to wound breakdown or infection, duration of drain usage, drainage volumes. Additionally, use of adjuvant chemotherapy or radiotherapy before or after surgery was recorded as these interventions are known to influence wound healing. All patients were treated with 40mg enoxaparin subcutaneously as routine post-operative venous thromboembolism prophylaxis. NPWT was applied with a mesh and occlusive dressing attached to a vacuum pump for a minimum of 3 days. The dressing was removed after a satisfactory wound review by the treating surgeon. To assess the non-inferiority between both groups, a Shapiro-Wilk normality test was applied and further statistical analysis performed using MiniTab version 17 (Minitab for Windows, Minitab Inc., State College, PA, USA).
Results
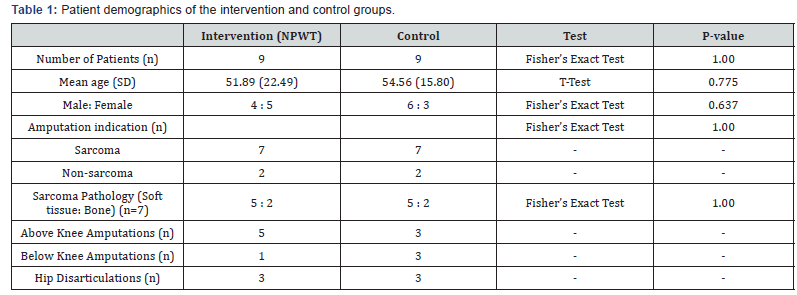
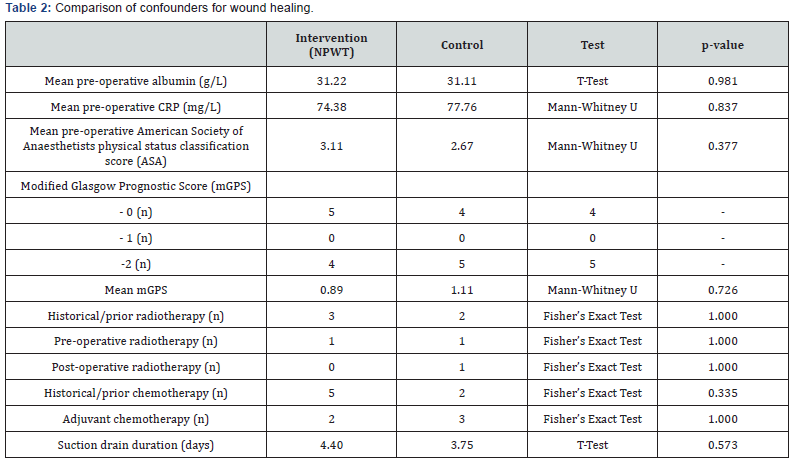
9 consecutive patients from May 2015 to May 2017 received VAC dressings as a routine procedure following amputations. The demographics and outcomes for both groups are compared in the tables below. As outlined in Table 1 & 2, the groups were well matched in terms of patient demographics and confounding factors, with no statistical significance between the intervention and control group. This includes the modified Glasgow Prognostic Score (mGPS) which measures serum albumin and CRP, and has been shown predict survival across multiple cancers. NPWT dressings in the intervention group were utilised for a mean of 5.6 days (range 3-10, median 5), with no adverse reactions encountered. Suction drains were used in patients post-operatively in the intervention and control group for 8 patients and 4 patients respectively, with a similar mean duration of 4.40 days in the intervention group and 3.75 days in the control group. Regarding the outcomes measured, there was no significant difference in the mean volume drained with or without the use of NPWT (285.6ml versus 220.0ml). The intervention group had 2 patients which encountered wound complications compared to 5 patients in the control group, however this did not reach statistical significance (p=0.335).
Discussion
Despite the non-statistical significance in the proportion of patients with wound complications following amputations, there is a trend to suggest the potential for NPWT to improve outcomes which certainly warrants further investigation. This could in part be explained by limited participants, but also by the higher rates of prior chemotherapy and radiotherapy in the intervention group. Quantitatively, the use of NPWT could decrease wound complications in amputation patients by reducing dead space, minimising wound oedema, and presenting a favourable biological environment for wound healing. Wound complications can incur an increase in healthcare expenditure in a multitude of modes; increased hospital stay, usage of antibiotics, and wound breakdown leading to secondary surgical intervention and revisions. [13, 14, 28] In 2005, Ploeg et al investigated morbidity and mortality following lower limb amputation. This investigation found that wound infection was the most common complication in below knee amputations (10.4%) [28]. In the same study, 3 of 7 patients requiring reoperation were due to wound infections [28]. Quality of life is also adversely affected by wound complications, and causes further loss of function or psychological impact on patients with major life changing surgery [27].
The current cost of a reusable ActiVAC unit (KCI, Texas) within our institution is ₤6,500, with each negative pressure dressing costing approximately ₤25 individually. Although the initial implementation of NPWT as routine therapy may increase the individual cost of patient treatment, the potential reduction in wound complications and hence further treatment costs may help to reduce overall costs. Philbeck et al conducted a retrospective cost analysis in 1999, to compare the clinical and cost effectiveness of NPWT therapy on chronic wound therapy in home healthcare. This study saw a mean reduction of USD $8,919 per patient [29]. The current cost for each individual hospital bed is quoted by the Department of Health at ₤400 per day. Furthermore, a study by Daniel de Alcântara Jones et al in 2016 quoted a mean reduction of 7.2 days in patient with NPWT compared to control [30].
Therefore, the reduction of only a small number of bed days would quickly offset the costs of NPWT as a routine therapy [30,31]. There is increasing evidence for the use of NPWT conducted in the field of oncology, trauma, and vascular surgery. Whilst primary outcomes differ between studies, NPWT has displayed positive outcomes via increased rate of wound healing, with a decreased rate of wound complications and adverse events. Future prospective studies and meta-analyses could include a larger cohort of patients to prove the statistical significance of the use of NPWT in sarcoma patients with amputations. Additionally, a cost analysis could be performed to justify an initial investment into the introduction of NPWT as routine therapy.
Conclusion
This study demonstrates that the rate of wound complications in patients following amputation is lower with NPWT compared to control with a definite positive trend. Importantly, no adverse events in relation to the use of NPWT were encountered. Current literature presents increasing evidence that the potential benefits of NPWT could outweigh the increased treatment cost of routine NPWT usage in amputations. Additionally, NPWT may improve outcomes with regards to patient health and quality of life, an outcome which should be addressed in future prospective trials. An appropriately powered randomised control trial involving sarcoma patients within our institution has been ethically approved and seeks to expand upon the hypothesis of the benefits of NPWT.
References
- Tintle L, Keeling C, Shawen L, Forsberg L, Potter M (2010) Traumatic and Trauma-Related Amputations. J Bone Joint Surg Am 92(17): 2852- 2868.
- Lance R (1989) Limb Reconstruction Versus Amputation Decision Making in Massive Lower Extremity Trauma. Clin Orthop Relat Res 243: 92-99.
- Hiatt M, Farmer J, Teasdall R (2000) The Decision to Salvage or Amputate a Severely Injured Limb. J South Orthop Assoc 9(1): 72-78.
- Grimer R, Judson I, Peake D, Seddon B (2010) Guidelines for the Management of Soft Tissue sarcomas. Sarcoma 2010: 1-15.
- Dangoor A, Seddon B, Gerrand C, Grimer R, Whelan J, et al. (2016) UK guidelines for the management of soft tissue sarcomas. Clin Sarcoma Res 6(1).
- Gerrand C, Athanasou N, Brennan B, Grimer R, Judson I, et al. (2016) UK guidelines for the management of bone sarcomas. Clinical Sarcoma Research 6(1).
- Chmell M, Schwartz H (1996) Analysis of variables affecting wound healing after musculoskeletal sarcoma resections. J Surg Oncol 61(3): 185-189.
- Peat B, Bell R, Davis A, O Sullivan B, Mahoney J, et al. (1994) Wound- Healing Complications after Soft-Tissue Sarcoma Surgery. Plast Reconstr Surg 93(5): 980-987.
- Payne W, Naidu D, Wheeler C, Barkoe D, Mentis M, et al. (2008) Wound Healing in Patients With Cancer. Open Access Journal of Plastic Surgery 8(e9).
- Haubner F, Ohmann E, Pohl F, Strutz J, Gassner H (2012) Wound healing after radiation therapy: Review of the literature. Radiat Oncol 7(1): 162.
- Alamanda V, Crosby S, Archer K, Song Y, Schwartz H, et al. (2012) Amputation for extremity soft tissue sarcoma does not increase overall survival: A retrospective cohort study. Eur J Surg Oncol 38(12): 1178- 1183.
- Stojadinovic A, Jaques D, Leung D, Healey J, Brennan M (2001) Amputation for Recurrent Soft Tissue Sarcoma of the Extremity: Indications and Outcome. Ann Surg Oncol 8(6): 509-518.
- Harris A, Althausen P, Kellam J, Bosse M, Castillo R (2009) Complications Following Limb-Threatening Lower Extremity Trauma. J Orthop Trauma 23(1): 1-6.
- Jensen J, Mandrup Poulsen T, Krasnik M (1982) Wound healing complications following major amputations of the lower limb. Prosthet Orthot Int 6(2): 105-107.
- Argenta L, Morykwas M (1997) Vacuum-Assisted Closure: A New Method for Wound Control and Treatment. Ann Plast Surg 38(6): 563- 577.
- Borgquist O, Ingemansson R, Malmsjö M (2011) The Influence of Low and High Pressure Levels during Negative-Pressure Wound Therapy on Wound Contraction and Fluid Evacuation. Plast Reconstr Surg 127(2): 551-559.
- Scherer S, Pietramaggiori G, Mathews J, Prsa M, Huang S, et al. (2008) The Mechanism of Action of the Vacuum-Assisted Closure Device. Plast Reconstr Surg 122(3): 786-797.
- Yang C, Chang D, Webb L (2006) Vacuum-assisted closure for fasciotomy wounds following compartment syndrome of the leg. J Surg Orthop Adv 15(1): 19-23.
- Orgill D, Manders E, Sumpio B, Lee R, Attinger C, et al. (2009) The mechanisms of action of vacuum assisted closure: More to learn. Surgery 146(1): 40-51.
- Scherer S, Pietramaggiori G, Mathews J, Orgill D (2009) Short Periodic Applications of the Vacuum-Assisted Closure Device Cause an Extended Tissue Response in the Diabetic Mouse Model. Plast Reconstr Surg 124(5): 1458-1465.
- Younan G, Ogawa R, Ramirez M, Helm D, Dastouri P, et al. (2010) Analysis of Nerve and Neuropeptide Patterns in Vacuum-Assisted Closure–Treated Diabetic Murine Wounds. Plast Reconstr Surg 126(1): 87-96.
- https://www.dovepress.com/negative-pressure-wound-therapy-clinical-utility-peer-reviewed-fulltext-article-CWCMR
- Stannard J, Robinson J, Anderson E, McGwin G, Volgas D, et al. (2006) Negative Pressure Wound Therapy to Treat Hematomas and Surgical Incisions Following High-Energy Trauma. J Trauma 60(6): 1301-1306.
- Stannard J, Volgas D, McGwin G, Stewart R, Obremskey W, et al. (2012) Incisional Negative Pressure Wound Therapy After High-Risk Lower Extremity Fractures. J Orthop Trauma 26(1): 37-42.
- Stannard J, Volgas D, Stewart R, McGwin G, Alonso J (2009) Negative Pressure Wound Therapy After Severe Open Fractures: A Prospective Randomized Study. J Orthop Trauma 23(8): 552-557.
- Achten J, Parsons N, Bruce J, Petrou S, Tutton E, et al. (2015) Protocol for a randomised controlled trial of standard wound management versus negative pressure wound therapy in the treatment of adult patients with an open fracture of the lower limb: UK Wound management of Open Lower Limb Fractures (UK WOLFF). BMJ Open 5(9): e009087.
- Kong R, Shields D, Bailey O, Gupta S, Mahendra A (2017) Negative Pressure Wound Therapy for Closed Surgical Wounds in Musculoskeletal Oncology Patients - A Case-Control Trial. Open Orthop J 11(Suppl-3, M5): 502-507.
- Ploeg A, Lardenoye J, Vrancken Peeters M, Breslau P (2005) Contemporary Series of Morbidity and Mortality after Lower Limb Amputation. Eur J Vasc Endovasc Surg 29(6): 633-637.
- Philbek Jr T, Whittington K, Millsap M, Briones R, Wight D, et al. (2017) The clinical and cost effectiveness of externally applied negative pressure wound therapy in the treatment of wounds in home healthcare Medicare patients. Ostomy Wound Manage 44(11): 41-50.
- Jones D, Neves Filho W, Guimarães J, Castro D, Ferracini A (2016) The use of negative pressure wound therapy in the treatment of infected wounds. Case studies. Rev Bras Ortop 51(6): 646-651.
- Arti H, Khorami M, Ebrahimi Nejad V (1969) Comparison of Negative Pressure Wound Therapy (NPWT) and Conventional Wound Dressings in the Open Fracture Wounds. Pak J Med Sci 32(1): 65-69.






























