Abstract
This report details the development of foveolitis in a patient undergoing antiparasitic treatment and corticosteroid taper for reactivated toxoplasmic retinochoroiditis. OCT findings included ellipsoid zone disruption and scattered hyper-reflective dots within the inner layers of the retina. Foveolitis is a frequent form of presentation of dengue maculopathy. Although the patient resided in Misiones Province, Argentina, amidst a Dengue epidemic, serological tests for IgM and IgG anti-Dengue antibodies were negative. In this manuscript we provide possible explanations on the association of active Ocular Toxoplasmosis and the sudden development of Foveolitis amidst a Dengue epidemic despite negative Dengue serology
Keywords: Ocular Toxoplasmosis; Retinochoroiditis; Foveolitis; Maculopathy; Myodesopsias
Abbreviations: NEA: Northeast region of Argentina; OT: ocular toxoplasmosis; RTR: Reactivation of toxoplasmic retinochoroiditis; ENSO: El niño- Southern Oscillation; RE: right eye; LE: left eye; CF: count fingers; BCVA: best-corrected visual acuity; AC: anterior chamber; OCT: Optical coherence tomography; EZ: ellipsoid zone; ELM: external limiting membrane
Introduction
Misiones Province, located in the extreme Northeast region of Argentina (NEA), together with the neighboring southern Brazilian states of Rio Grande do Sul and Santa Catarina, constitutes a geographic area with a notably high prevalence of ocular toxoplasmosis (OT) [1,2]. Reactivation of toxoplasmic retinochoroiditis (RTR) is the most frequent clinical presentation of OT in this region. The NEA has experienced recurrent epidemic outbreaks of dengue virus infection, occurring approximately every 3 to 4 years over the past decade. In 2023, two key epidemiological factors were observed: (1) the continuous, yearround detection of human dengue virus infections [3], and (2) intense rainfall during October and November, associated with the El Niño- Southern Oscillation (ENSO) phenomenon. Increased rainfall, particularly during ENSO events, has previously been associated with both a higher frequency of OT reactivations [4,5] and an elevated risk of epidemic dengue outbreaks [6,7]. In early 2024, Misiones Province experienced a severe epidemic outbreak of dengue virus infection [8]. Within this epidemiological context, the presentation of a patient with active toxoplasmic retinochoroiditis and concurrent ocular findings suggestive of dengue virus involvement was an anticipated clinical scenario. The present report describes the ocular manifestations associated with dengue virus infection in a patient diagnosed with RTR, residing in Misiones Province, Argentina.
Patient Presentation and History
A 29-year-old male was referred for uveitis consultation on March 5, 2024, presenting with a recent-onset central scotoma in his right eye (RE). This symptom reportedly developed concurrently with the tapering of his systemic corticosteroid therapy (oral meprednisone reduced from 40 mg to 20 mg daily) approximately two weeks after initiating treatment for reactivation of ocular toxoplasmosis (OT). The patient reported that this current episode of OT reactivation began around February 15, 2024, with symptoms of decreased visual acuity and floaters (myodesopsias) in the RE after one day of low-grade fever. His medical history was significant for a prior OT reactivation in the same eye two years previously. He also reported poor central vision in his left eye (LE) since childhood, noted as count fingers acuity, historically due to previous toxoplasmic retinochoroideal scar. His current treatment regimen for the OT reactivation included oral trimethoprim-sulfamethoxazole (160 mg - 800 mg) and oral meprednisone (initially 40 mg/day), supplemented with folinic acid.
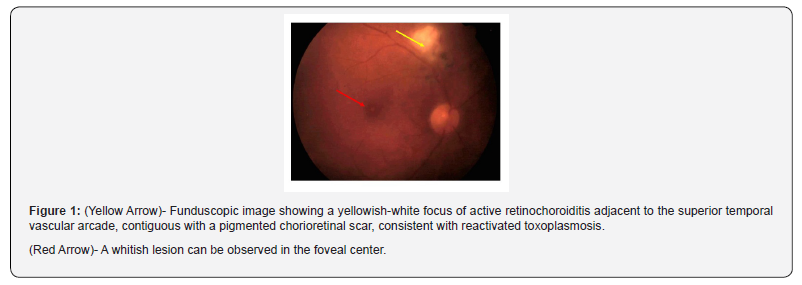
On the ophthalmic examination performed on March 5th, 2024, the best-corrected visual acuity (BCVA) was 0.7 in the RE and count fingers (CF) in the left eye (LE). Slit-lamp examination of the RE revealed 1+ anterior chamber (AC) cells and 1+ flare, along with fine, peripheral keratic precipitates. No significant anterior segment inflammation was noted in the LE. Funduscopic examination of the RE revealed a yellowish-white focus of active retinochoroiditis adjacent to the superior temporal vascular arcade, contiguous with a pigmented chorioretinal scar, consistent with reactivated toxoplasmosis (Figure 1, yellow arrow). Vitreous examination showed moderate vitreous haze with cellularity (vitritis). Critically, a distinct whitish lesion was observed directly in the foveal center (Figure 1, red arrow)..
Ancillary Testing
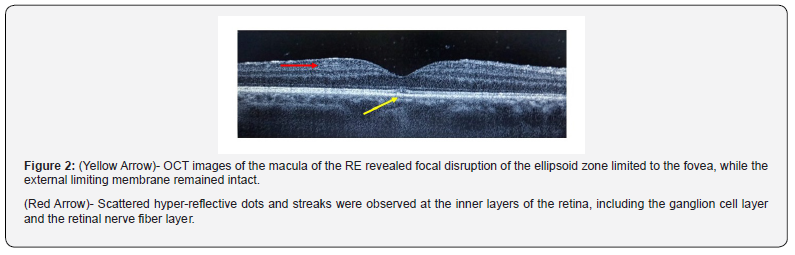
Optical coherence tomography (OCT) centered on the macula of the right RE revealed focal disruption of the ellipsoid zone (EZ) limited to the fovea, while the external limiting membrane (ELM) remained intact throughout the macular region.
This alteration topographically corresponded with the patient’s subjective central scotoma and was interpreted as foveolitis. Scattered hyper-reflective dots and streaks were also observed at the inner layers of the retina, including the ganglion cell layer and the retinal nerve fiber layer (Figure 2). Additionally, hyperreflective dots-consistent with inflammatory cells-were noted in the overlying vitreous, confirming active vitritis. The primary active lesion, adjacent to a pigmented scar, was consistent with reactivation of OT. However, the new-onset foveal lesion and central scotoma, which developed despite appropriate anti-toxoplasma therapy and during corticosteroid tapering, raised diagnostic concern. Given the ongoing dengue virus epidemic in Misiones Province since late December 2023, and the documented potential for dengue-associated maculopathy (including foveolitis), serologic testing for Dengue and Chikungunya virus (IgM and IgG antibodies) was ordered to evaluate a possible viral coinfection or alternative explanation for the foveal involvement. Peripheral blood was drawn and submitted for virus antibody analysis. The patient was instructed to increase the oral meprednisone dose back to 40 mg/day, while continuing oral trimethoprim-sulfamethoxazole (TMP-SMX) with folinic acid supplementation. Laboratory testing confirmed chronic Toxoplasma gondii infection. However, initial serologic results were negative for both anti-Dengue and anti-Chikungunya IgM and IgG antibodies.
Follow Up
On his second ophthalmic examination on March 15, 2024, the patient reported resolution of the central scotoma, with best corrected visual acuity (BCVA) improving to 1.0 in the RE. Despite this improvement, fundoscopic examination still revealed a milder whitish dot in the macular center. The retinochoroidal toxoplasmic lesion appeared flattened with well-demarcated borders. The patient was advised to gradually taper the meprednisone dose but did not attend the scheduled follow-up examination two weeks later.
Discussion
Ocular manifestations of dengue virus infection typically occur after the febrile phase, often coinciding with the recovery period, but may also arise in asymptomatic individuals [9]. Common retinal findings include retinal hemorrhages, dengue maculopathy, perivascular exudative retinal detachments, and retinal vasculitis. OCT has proven essential in characterizing dengue-related macular involvement. Theo et al. [10] described three distinct patterns of dengue maculopathy [10]: A) diffuse macular edema, B) cystoid macular edema, and C) foveolitis [10]. Among these, foveolitis is characterized by disruption of the ellipsoid zone and the external limiting membrane, typically accompanied by a conical elevation of the foveal contour. To date, foveolitis has been predominantly associated with dengue fever [11] and, more recently, with SARSCoV- 2 vaccination [12]. In the present case, the patient denied receiving any recent SARS-CoV-2 vaccination prior to the onset of reactivated toxoplasmic retinochoroiditis (OT). Three possible explanations were considered for the development of foveolitis in this clinical context:
First, the patient may have experienced a concurrent dengue virus infection, undetected serologically due to the effects of high-dose corticosteroid therapy, administered alongside TMPSMX. Corticosteroids are known to impair and delay antibody production [13] through multiple mechanisms: inhibition of B-cell proliferation and differentiation, modulation of gene expression leading to impaired B-cell receptor and Toll-like receptor signaling, induction of apoptosis in specific B-cell populations. Further corticosteroids also induce transient redistribution of lymphocytes, reducing their interaction with antigens. More importantly, corticosteroid use has also been shown to delay the initiation of humoral immune responses, as reported in SARSCoV- 2 infection studies [14]. These immunosuppressive effects could explain the absence of detectable anti-dengue IgM and IgG antibodies during initial testing.
Second, a false-negative serologic result could not be ruled out. To address this possibility, an aliquot of the same serum sample was sent for confirmatory testing at the Instituto Nacional de Enfermedades Virales Humanas “Dr. Julio I. Maiztegui” (Pergamino, Buenos Aires Province, Argentina). Testing conducted by expert virologists at the center also yielded a negative result.
Third, and considered the least likely explanation, foveolitis might represent a rare manifestation of toxoplasmic retinochoroiditis itself, although this association has not been documented in the literature.
To the best of our knowledge, this report represents the first documented case of active OT associated with foveolitis in the context of an epidemic dengue virus outbreak.
Acknowledgement
The authors would like to thank Dr. Maria Alejandra Morales, Head of National Institute of Human Viral Infections “Dr. Julio Maiztegui” (INEVH), for her assistance with the serological diagnosis of the patient samples.
References
- Glasner P D, Silveira C, Kruszon-Moran D, Martins MC, Burnier Jr M, et al. (1992) An unusually high prevalence of ocular toxoplasmosis in southern Brazil. Am J Ophthalmol 114 (2): 136-144.
- Rudzinski M, Khoury M, Couto C, Ajzenberg D (2016) Reactivation of Ocular Toxoplasmosis in Non-Hispanic Persons, Misiones Province, Argentina. Emerg Infect Dis 22 (5): 912-913.
- Ferna ́ndez G, El Ahmed Y, Rueda D, Santoro FM, Mosis S, et al. (2024) Abovirus. National Epidemiological Bulletin No. 737, SEM 52.
- Rudzinski M, Meyer A, Khoury M, Couto C (2013) Is reactivation of toxoplasmic retinochoroiditis associated to increased annual rainfall? Parasite 20: 44.
- Boada-Robayo L, Cruz-Reyes DL, Cifuentes-Gonzalez C, Rojas-Carabali W, Vargas-Largo AP, et al. (2022) Exploring the association between precipitation and population cases of ocular toxoplasmosis in Colombia. PLoS Negl Trop Dis 16 (10): e0010742.
- Hales S, Weinstein P, Woodward A (1996) Dengue fever epidemics in the South Pacific: driven by El Nin˜o Southern Oscillation? 348(9042): 1664-1665.
- Anyamba A, Chretien JP, Britch SC, Soebiyanto RP, Small JL, et al. (2019) Global Disease Outbreaks Associated with the 2015-2016 El Nin˜o Event. Sci Rep 9(1): 1930.
- Ferna´ndez G, Amelotti L, Amato B, Kurten Perez M, Fernandez L, et al. (2024) Boletin Epidemiolo´gico Nacional N° 705, SEM 20.
- Rudzinski M, Echeverria A (2020) Ocular inflammatory manifestations of Dengue Virus infection. Oftalmol Clin Exp 13(3): 113-126.
- Teoh SC, Chee CK, Laude A, Goh KY, Barkham T, et al. (2010) Optical coherence tomography patterns as predictors of visual outcome in dengue-related maculopathy. Retina 30(3): 390-398.
- Agarwal A, Aggarwal K, Dogra M, Kumar A, Akella M, et al. (2019) OCTA Study Group. Dengue- induced inflammatory, ischemic foveolitis and outer maculopathy: a swept-source imaging evaluation. Ophthalmol Retina 3(2): 170-177.
- Khochtali S, Krifa H, Zina S, Khairallah M, Jelliti B, et al. (2022) Multimodal Imaging of Acute Foveolitis following COVID-19 Vaccination. Ocul Immunol Inflamm 30(5): 1214-1217.
- Sarcevic J, Cavelti-Weder C, Berger CT, Trendelenburg M (2020) Case Report-Secondary Antibody Deficiency Due to Endogenous Hypercortisolism. Frontiers in Immunology 11:1435.
- Wang Y, Guo L, Fan G, Han Y, Zhang Q, et al. (2024) Impact of corticosteroids on initiation and half-year durability of humoral response in COVID-19 survivors. Chin Med J Pulm Crit Care Med 2(1): 48-55.






























