The Influence of Electrical Stimulation on the Proliferation and Differentiation of Retinal Progenitor Cells
Ting Liang#, Zhaojun Meng# and Xiangchun Gao*
Department of Ophthalmology, Harbin Medical University the 4th Affiliated Hospital, Harbin, China
Submission: April 15, 2024; Published: May 01, 2024
*Corresponding author: Xiangchun Gao, Department of Ophthalmology, 4th Affiliated Hospital of Harbin Medical University, No. 37, Yiyuan Street, Harbin, Heilongjiang Province, China
#Co-first author
How to cite this article: Ting Liang, Zhaojun Meng and Xiangchun Gao*. Glucagon-like Peptide-1 Receptor Agonist for Diabetic Retinopathy: Friend or Foe?. JOJ Ophthalmol. 2023; 11(1): 555801. DOI: 10.19080/JOJO.2024.11.555801
Abstract
Purpose: This study aimed to evaluate the effects of electrical stimulation on the proliferation and differentiation of retinal progenitor cells (RPCs) and explore the potential mechanism.
Methods: RPCs were got from retinas of postnatal days 0-2 GFP+ C57BL/6 mice. Electrical stimulation (ES) was performed with a biphasic current pulse at the pulse width of 2ms and 20Hz pulse frequency. The intensity of the electrical stimulation was 300μA. RPCs were treated with or without ES one hour per day for consecutive 3 days in a state of cell proliferation and differentiation. Cell morphology was observed under a fluorescence microscope to evaluate RPCs viability. And the proliferation ability of RPCs was evaluated by EdU. Immunocytochemistry and western blot analyses were used to observe expression levels of p-Akt, p-Erk, Akt, Erk, rhodopsin, β3-tubulin and PKC-α.
Results: RPCs in ES group exhibited higher cell densities and viability than the cells in control group without ES (p<0.05). The relative fluorescence intensity in the ES group was nearly two times that of the control group. Immunocytochemistry and western blot analyses showed that expression levels of p-Akt, p-Erk, Akt and Erk in the ES group was significantly higher than that of the control group (p<0.05). And the relative protein expression levels of rhodopsin (photoreceptor marker),β3-tubulin (panneuron marker) and Pkc-a (bipolar cell marker) in RPCs affected by ES were approximately 1.6-fold, 2.1-fold and 2.4-fold of those in control group, respectively.
Conclusion: ES could improve RPCs proliferation via the activation of the Akt and Erk signaling pathways. Moreover, ES markedly induced RPCs differentiation toward retinal neurons. Therefore, ES can be used as a means to modulate the survival, proliferation, and differentiation of RPCs before in vivo implantation.
Keywords: Electrical Stimulation; Retinal Progenitor Cells; AKT; ERK; Green Fluorescence
Abbreviations: ES: Electrical Stimulation; RDDs: Retinal Degenerative Diseases; anti-VEGF: anti-Vascular Endothelial Growth Factor; RPCs: Retinal Progenitor Cells ; ESCs: Embryonic Stem Cells; iPSCs: induced Pluripotent Stem Cells; MSC: Mesenchymal Stromal Cells; GFP+: Green Fluorescence Protein; PAGE: Polyacrylamide Gel Electrophoresis; PVDF: Polyvinylidene Difluoride
Introduction
Retinal degenerative diseases (RDDs) are the main reasons for irreversible blindness world wild [1]. Currently available treatments for RDDs include antioxidants, anti-vascular endothelial growth factor (anti-VEGF) agents, neuroprotective strategies, laser or surgery therapy [2]. However, the clinical efficacy of these treatments is limited. Because RDDs are characterized by progressive degeneration of retinal neurons, stem cell transplantation to replace and/or rescue degenerative cells shows great potential for treating RDD [3,4]. The stem cells investigated for RDDs treatment mainly include retinal progenitor cells (RPCs), embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs) and mesenchymal stromal cells(MSC). Each has its own advantages and disadvantages [2,5]. Among them, RPCs have many attractive advantages such as avoiding ethical issues and relatively low risk of immune rejection and tumorigenisis. Therefore, RPCs are a good source of donor cells for transplantation therapy of retinal degeneration. And many studies have already verified the safety and efficacy of RPCs-based transplantation therapy and got some outcomes of improved visual acuity in clinical trials [2,6,7]. Unfortunately, the efficacy of RPCs transplantation for RDDs therapy is not long-lasting. Even the clinical improvement did not persist after 12 months [7,8].
In fact, the fate of implanted stem cells was affected by many factors such as glial scarring [9,10], chronic inflammation [11,12], and abnormal metabolic microenvironment [13] in degenerated retina. These abnormal situations are often unfavorable for longterm survival, proper differentiation and functional integration into the host neural network. Therefore, the effect of stem cell therapy on RDDs could not be maintained for a long time [2,13]. If it is difficult to change an already existing abnormal retinal structure and microenvironment, further research is urgently required for enhance survival, proliferation and differentiation ability of RPCs to obtain a longer-lasting treatment effect.
Various biochemical and physical approaches have been explored to regulate the stem cell biological activity such as survival, proliferation, differentiation, migration and integration with the host, among which electrical stimulation has been validated as a promising way to affects neural stem cell fate and function [14-16]. As a physical means of affecting neural stem cell fate and function, electrical stimulation has many advantages such as precisely controlling the stimulation time, no chemical residuals left in the system and effective biological effects. So, it is a relatively safe and effective method in regulating stem cell survival, proliferation and differentiation.
Currently, most studies focus on investigating the effect of electrical stimulation on behavior of ESC MSC iPSC or NSC. There are few relative studies on RPCs [17,18]. So, in our study, electrical stimulation was applied to mouse RPCs during culture in vitro. The aim is to investigate the influence of electrical stimulation on RPCs survival, proliferation, differentiation and explore the potential mechanism. These results would provide an experimental basis for better using this physical approach to control the biological activity of RPCs so as to obtain better transplantation results.
Method
Animals
Adult enhanced green fluorescence protein (GFP+) C57BL/6 mice were purchased from Cyagen Biosciences, Suzhou, China. All the procedures followed the ARVO Statement in using animals for ophthalmic and vision research as well as the policies listed in the guide of the care and use of laboratory animals issued by the NIH.
RPC Isolation and Culture
Retinas were isolated from postnatal days 0–2 GFP+ C57BL/6 mice. The obtained retinas were dissociated by mincing, and then digested with 0.1% type I collagenase for 30 min at room temperature with intermittent shaking every 10 minutes. At the end of the digestion, two volumes of DMEM medium with 10% FBS were added to the solution to terminate digestion. The solution was passed through a 100 μm mesh filter, centrifuged at 1000 rpm for 5 min. The liberated mRPCs were resuspended in neurobasal medium containing 20 ng/ mL epidermal growth factor (Promega), 2% B-27 (Invitrogen), 1% N2 (Invitrogen), 100 mg/mL penicillin/streptomycin (Sigma), 2 mM l-glutamine (Sigma), and 200 U/mL nystatin (Sigma) and plated into culture wells. Cells were cultured in an eight-well plate at 37°C with 5% CO2 and the medium changed every 2 days. Primary neuro spheres formed within the first 2 weeks. These proliferating cultures were passaged 1:3 every 7 days. The cell morphologies in the proliferation and differentiation media were observed by a fluorescence microscope (Nikon, Tokyo, Japan), then Image J was used to count cell numbers and analyze fluorescence intensity.
Electrical Stimulation of RPCs
A multichannel neurophysiology workstation (System 3; Tucker-Davis Technologies) was used to apply ES to the cells in DMEM. Eight wells of RPCs were stimulated simultaneously by immersing microelectrodes connected. Four wells received ES, and four wells received sham stimulation in a cell culture incubator. Electrical stimulation was performed with a biphasic current pulse at the pulse width of 2ms and 20Hz pulse frequency. The intensity of the electrical stimulation was 300μA. RPCs were stimulated one hour per day for consecutive 3 days.
Edu Assay
Edu assays were used to evaluate the proliferative capacity of RPCs. The steps of the Edu assays were performed according to recommended procedures. After discarding Edu medium mixture, RPCs were fixed with 4% paraformaldehyde for 30 min and stained with Apollo Dye Solution, and then nucleic acids were stained with Hoechst 33,342 for 30 min. Photographs were taken by fluorescent microscope (Nikon). The cell proliferation ratio was the (Edu add-in cells/Hoechst-stained cells in the field) × 100%. EdU-positive cells and total cells were counted within each field. EdU-positive cells are expressed as a ratio of EdU-positive cells to all cells in a given visual field.
Immunocytochemistry
The immunocytochemistry analyses were carried out to evaluate the differentiation abilities and potential mechanism of the RPCs in 6-well plates prepared. RPCs were fixed for 30 min in 4% paraformaldehyde and then washed 3 times with PBS. After that, these cells were blocked, and permeabilized in PBS containing 10% normal goat serum and 0.5% TritoX-100 for 2 hr. Samples were incubated with the primary antibodies: mouse monoclonal anti-PKC-α (1 : 200, BD Biosciences), β3-tubulin (1: 200, Chemicon), rabbit monoclonal anti-P-Erk, P-Akt (1 : 200, Cell Signaling Technology), and mouse monoclonal anti-rhodopsin (1: 200, Millipore), in blocking buffer at 4°C overnight.The cells were incubated with fluorescence conjugated secondary antibody (Jackson Immuno Research 1: 100) for 2h at room temperature. Lastly, the cell nuclei of the RPCs were stained with DAPI, and images were obtained by a fluorescence microscope (Nikon, Tokyo, Japan).
Westerm Blot Analysis
Total proteins were extracted after three days of proliferation or seven days of differentiation culture conditions. Proteins were separated by SDS-polyacrylamide gel electrophoresis (PAGE) and then transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked by 5% (w/v) skimmed milk buffer (skimmed milk powder/TBST) for 2h at 37°C followed by incubation with primary antibodies of target proteins at 4°C overnight. The primary antibodies used here included the following: mouse monoclonal anti-PKC-a (1: 1000), nestin, rhodopsin (1: 1000; Millipore), as well as rabbit monoclonal anti-vimentin, Akt, P-Akt, Erk and P-Erk (1: 1000; Cell Signaling Technology).Then, the membranes were incubated with appropriate horseradish peroxidase-conjugated secondary antibody (Sigma-Aldrich). The ECL Plus Western Blot detection kit (Tanton) was used to detect the protein expression.
Statistical Analysis
The data were analyzed with GraphPad Prism 5.0 by the Student’s t-test. All data were presented as mean ± SEM, and p < 0.05 was designated as statistically significant.
Results
RPCs Viability
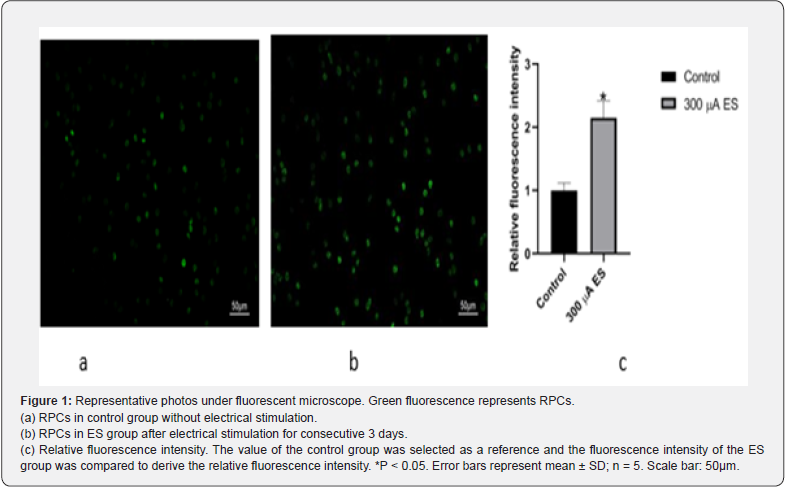
RPCs were stimulated one hour per day for consecutive 3 days. Cell morphology was observed under a fluorescence microscope. Five fields of view were randomly selected for observation under the microscope and counting RPCs number. The value of the control group was selected as a reference and the fluorescence intensity of the ES group was compared to derive the relative fluorescence intensity. As shown by Figure 1 a, b, RPCs in ES group exhibited higher viability than the cells in control group without ES (p<0.05). The relative fluorescence intensity in the ES group was nearly two times that of the control group (Figure 1c).
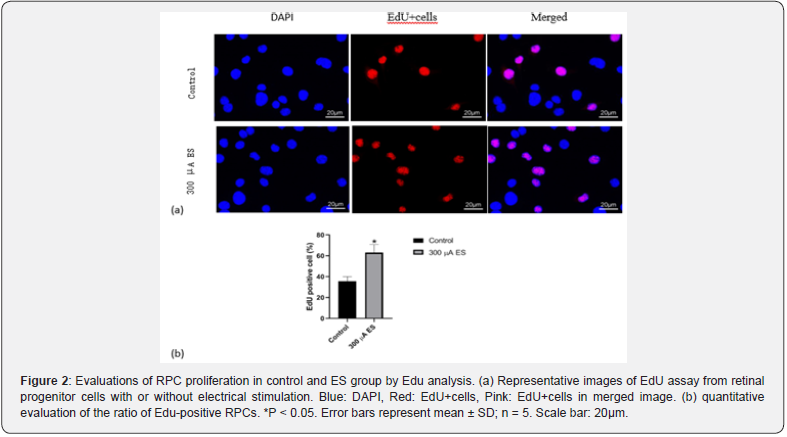
The Effect of Electrical Stimulation on RPCs Proliferation
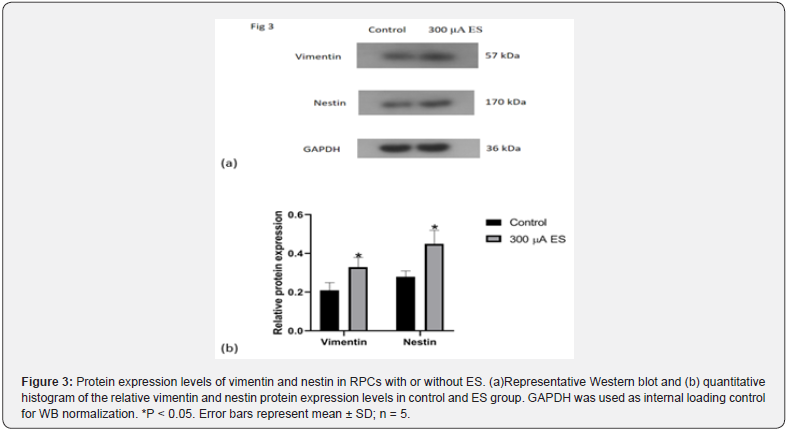
The proliferation ability of RPCs affected by ES was evaluated by EdU, immunocytochemistry and western blot analyses after that RPCs were stimulated under proliferation conditions. It was found that ES group has higher cell densities than control group without ES(p<0.05 Figure 2). Without ES, EdU positive cells account for 36% of the total cells in the field of view. However, under the influence of ES, EdU positive cells in ES group account for 61% of the total cells in the field of view (Figure 2). The protein expression levels of undifferentiated retinal progenitor cell markers, namely, Vimentin and Nestin, were dramatically increased in ES group as compared to those in control group (Figure 3).
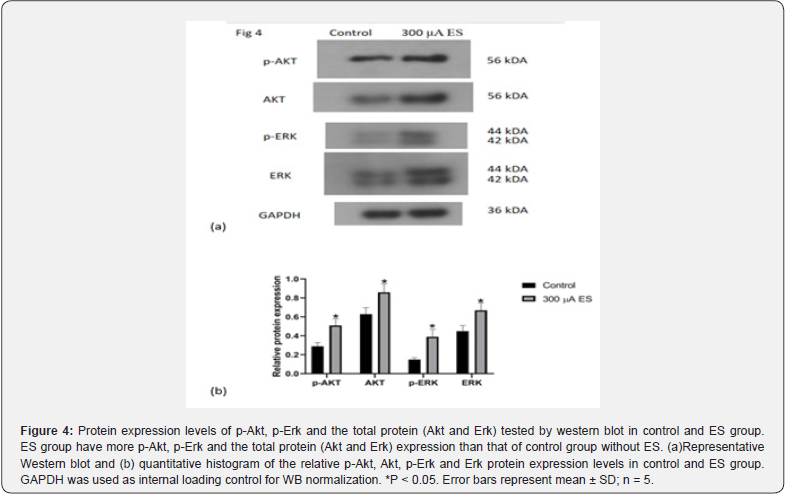
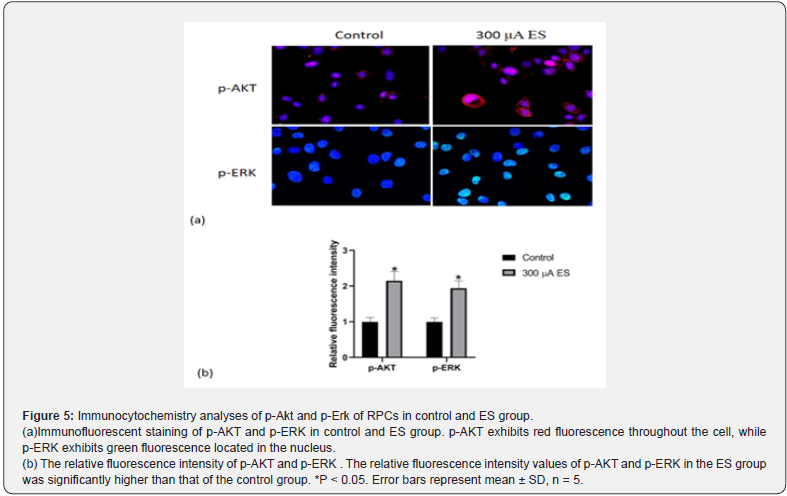
Obviously, ES could promote the proliferation and selfrenewal of RPCs. In order to further understand the molecular mechanism of the pro-proliferation effect of ES on RPCs, the Akt and Erk phosphorylation in RPC proliferation affected by electrical stimulation was further investigated through western blot and immunocytochemistry analysis. The results show that ES group have more Akt and Erk phosphorylation expression than that of control group without ES, (Figure 4), which is in agreement with the results of the immunocytochemistry analysis. Figure 5 shows immunofluorescent staining of p-AKT and p-ERK in control and ES group. p-AKT exhibits red fluorescence throughout the cell, while p-ERK exhibits green fluorescence located in the nucleus. The relative fluorescence intensity values of p-AKT and p-ERK in the ES group was significantly higher than that of the control group (Figure 5, p<0.05). Therefore, we speculate that electrical stimulation may improve RPCs proliferation by the activation of Akt and Erk pathways.
The Effect of Electrical Stimulation on the RPC Differentiation
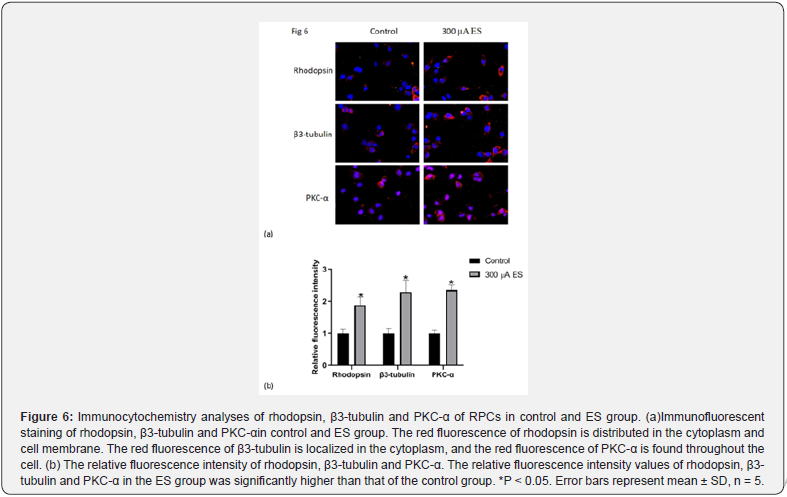
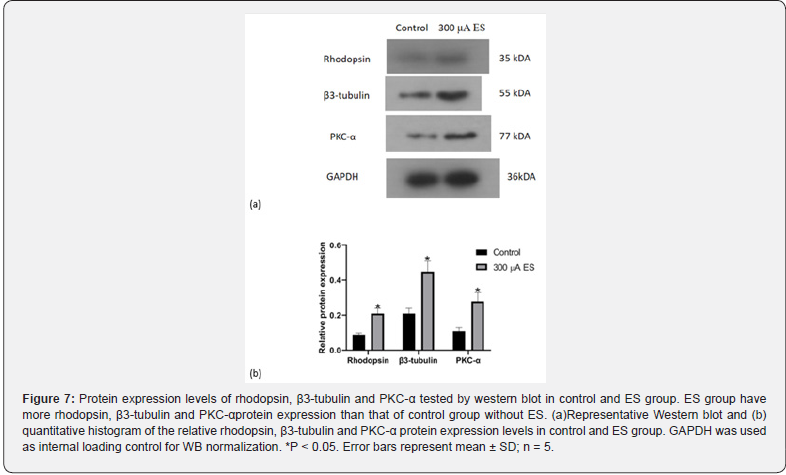
The differentiation ability of the RPCs affected by ES was evaluated by immunocytochemistry and western blot analyze under differentiation conditions (Figure 6,7). The results show that the relative protein expression levels of rhodopsin (photoreceptor marker), β3-tubulin (panneuron marker) and Pkc-a (bipolar cell marker) in RPCs affected by ES were approximately 1.6-fold, 2.1- fold and 2.4-fold of those in control group, respectively (Figure 7, p<0.05). The results of immunofluorescence staining were consistent with the western blotting. As shown in Fig 6a, the red fluorescence of rhodopsin is distributed in the cytoplasm and cell membrane. The red fluorescence of β3-tubulin is localized in the cytoplasm, and the red fluorescence of PKC-α is found throughout the cell. The relative fluorescence intensity of rhodopsin, β3- tubulin and Pkc-a in ES group were approximately 1.8-fold, 2.3- fold and 2.4-fold of those in control group, respectively (Figure 6, p<0.05).These results suggested that RPCs are more likely to be induced to differentiate into retinal neurons after being affected by ES.
Discussion
Currently, ES is used as a physical method to affect the function and fate of stem cell and produced a series of positive biological effects [19]. Various studies related to the effect of ES have been conducted on different stem cells including NSC, MSCs, iPSCs and ESCs [6,7]. However, there are few studies on the effects of ES on RPCs. In addition, the existing research results show that different types of stem cells respond differently to ES [20,21]. Therefore, this article used ES during the culture process of RPCs to observe the effects of ES on the survival, proliferation and differentiation functions of RPCs. At the same time, the underlying mechanism of proliferation improved by ES was also investigated. It is hoped that could improve RPCs limited ability to proliferate and differentiate into specific retinal neurons and promote their application to RDDs treatment.
Our results show that ES significantly improves proliferation, differentiation, and survival of RPCs. And we obtained similar results about higher expression of ßIII-tubulin in ES group compared to those previously observed by Nina et al, although the electrical stimulation parameter used in studies were different [22]. ßIII-tubulin is a protein exsiting throughout retinal neurons, which demonstrates that these cells are likely to differentiate into retinal neurons rather than into glial cells. In addition. Nina et al reported that electrically stimulated mRPCs expressed comparable levels of the photoreceptor marker as compared to non-stimulated control. What is different is that our results significantly increased expression levels of Pkc-a (bipolar cell marker) and rhodopsin (photoreceptor marker) in RPCs treated with ES. The reason for this difference may be that our results were obtained from RPCs treated by ES in a differentiated state. However, the results in Nina et al. study were observed in the cell proliferation process.
Subsequently, we explored the potential mechanism involved in proliferation improved by ES. Many studies showed that Akt and Erk signaling pathways play a crucial role in enhancing neural proliferation, differentiation and function maturation [23-25]. And activation of the pathways is shown by phosphorylation of Akt and ERK (p-Akt and p-ERK). Our results showed that ES group have more Akt and Erk phosphorylation expression. Therefore, we speculated that electrical stimulation may improve RPC proliferation by the activation of Akt and Erk pathways. And on olfactory bulb neural progenitor cells and injured spinal cord, results also showed that electrical stimulation could activate PI3K/Akt and Erk signaling pathway to protect against NSC apoptosis or promote the recovery of electrophysiological function of the injured spinal cord [25,26]. In fact, activation of AKT and ERK signaling pathway was usually closed related to increased Ca2+ influx induced by ES [23,27]. Additional factors may be also involved in the mechanism and still require further research.
The electrical stimulation parameters currently used in each study were different. Because there are many factors that can be changed such as frequency, electrical strength, and duration. In this study, we used a biphasic current pulse with varied stimulation amplitude, fixed frequency and duration. The results show that the relationship between the promotion of cell survival and ES intensity is not a simple linear one. Compared with 100μA and 500μA, 300μA stimulus intensity exerted the most strongest effect on the promotion of RPCs survival. It can be seen that there exists optimal stimulus parameters that affect cell function. It is even possible that high-intensity stimulation may not necessarily have positive effects on cell biological behavior [28]. Therefore, we believe that finding the optimal stimulation parameters is a critical part of RPCs practical applications in future. At the same time, we cannot ignore the determination of stimulus parameters that cause harmful damage to cells.
Our results show that biphasic electrical stimulation appears to be an effective method for influencing cellular survival, proliferation and differentiation of RPCs. In fact, there are still many aspects about the influence of ES on RPCs needed to be explored in the future. Firstly, effects of different electrical stimulation parameters and patterns (electric fields or electromagnetic fields) should be explored in order to optimize the stimulation parameters. Optimal electrical stimulation not only involved ES frequency, duration and intensity, but also included the choice of time points to electrical stimulation during cell development. How long RPCs should be electrical stimulated before, during, or after the implantation would be problems that need to be addressed before clinical application in future. Secondly, in order to better use electrical stimulation to improve the efficiency of RPCs clinical application, an in-depth understanding of the underlying mechanism is required. Thirdly, different cells respond differently to electrical stimulation. So, comparing the effects of ES on different cells also requires our attention.
Conclusion
ES could be used as an effective method for influencing survival, proliferation and differentiation of RPCs. Moreover, ES markedly induced RPCs differentiation toward retinal neurons and improve RPCs proliferation via the activation of the Akt and Erk signaling pathways. In future, there are still many aspects that require further investigation in order to better understand and use this physical method.
Funding
This project is supported by The Fourth Affiliated Hospital of Harbin Medical University Science and Technology innovation talent project Number: HYDSYCXRC202140 and Heilongjiang Natural Science Foundation Project LH2021H038.
References
- Majidi S, Ogilvie JM, Flaveny CA (2021) Retinal degeneration: short-term options and long-term vision for future therapy. Mo Med 118(5): 466-472.
- Yiqi W, Zhimin T, Ping G (2020) Stem/progenitor cell-based transplantation for retinal degeneration: a review of clinical trials. Cell Death Dis 11: 793.
- Mead B, Berry M, Logan A, Scott RA, Leadbeater W, et al. (2015) Stem cell treatment of degenerative eye disease. Stem Cell Res 14(3): 243-257.
- Van Gelder RN, Chiang MF, Dyer MA,Greenwell TN, Leonard AL, et al. (2022) Regenerative and restorative medicine for eye disease. Nat Med 28(6): 1149-1156.
- Xue W, Naihong Y, Ming Z (2022) Retinal Degeneration: Molecular Mechanisms and Therapeutic Strategies. Curr Med Chem 29(40): 6125-6140.
- US jCyte (2020) Safety and effificacy of intravitreal injection of human retinal progenitor cells in adults with retinitis pigmentosa.
- Liu Y, Chen SJ, Li SY, Qu LH, Meng XH, et al. (2017) Long-term safety of human retinal progenitor cell transplantation in retinitis pigmentosa patients. Stem Cell Res Ther 8(1): 209.
- Hirofumi U, Michiko M, Masayo T (2021) Stem cell-based therapies for retinal degenerative diseases: Current challenges in the establishment of new treatment strategies. Dev Growth Differ 63(1): 59-71.
- Katherine WC, Amanda CB, Jane CS, Robin RA, Rachael AP (2014) Migration, integration and maturation of photoreceptor precursors following transplantation in the mouse retina. Stem Cells Dev 23(9): 941-954.
- Young MJ, Ray J, Whiteley SJO, Klassen H, Gagem FH (2000) Neuronal differentiation and morphological integration of hippocampal progenitor cells transplanted to the retina of immature and mature dystrophic rats. Mol Cell Neurosci 16(3): 197-205.
- Balasubramaniam B, Carter D A, Mayer E J, Dick A D (2009) Microglia derived IL-6 suppresses neurosphere generation from adult human retinal cell suspensions. Exp Eye Res 89(5): 757-766.
- Xu Y, Balasubramaniam B, Copland DA, Liu J, Armitage MJ, et al. (2015) Activated adult microglia influence retinal progenitor cell proliferation and differentiation toward recoverin-expressing neuron-like cells in a co-culture model. Graefes Arch Clin Exp Ophthalmol 253(7): 1085-1096.
- Barber A C, Hippert C, Duran Y, West EL, Bainbridge JWB, et al. (2013) Repair of the degenerate retina by photoreceptor transplantation. Proc Natl Acad Sci USA 110(1): 354-359.
- Rong Z, Zhongqing S, Chuping L, Seeram R, Kin C, et al. (2019) Electrical stimulation affects neural stem cell fate and function in vitro. Exp Neurol 319: 112963.
- Hong C, Yan H, Hangqi Y, Yubo F (2021) Electrical stimulation promotes stem cell neural differentiation in tissue engineering. Stem Cells Int 2021: 6697574.
- Shen Y, Shen H, Guo D, Sun X, Sun Y, et al. (2020) Recent developments in regenerative ophthalmology. Sci China Life Sci 63(10): 1450-1490.
- Abby Leigh M, Wei L, Zhixin J, Marcus Hiu KL, Jian J, et al. (2017) Using electrical stimulation to enhance the efficacy of cell transplantation therapies for neurodegenerative retinal diseases: Concepts, Challenges, and Future Perspectives. Cell Transplant 26(6): 949-965.
- Lim JH, McCullen SD, Piedrahita JA, Loboa EG, Olby NJ (2013) Alternating current electric fields of varying frequencies: effects on proliferation and differentiation of porcine neural progenitor cells. Cell Reprogram 15(5): 405-412.
- Wang H, Mit B, Zhihua H, John HB (2020) Effects of electrical stimulation on stem cells. Curr Stem Cell Res Ther 15(5): 441-448.
- Ross AP, Peter AM, Martina Z, John AR, Shu X, et al. (2018) 3D bioprinter applied picosecond pulsed electric fields for targeted manipulation of proliferation and lineage specific gene expression in neural stem cells. J Neural Eng 15(5): 056021.
- Rocha I, Cerqueira G, Varella Penteado F, Córdoba de Torresi S (2021) Electrical stimulation and conductive polymers as a powerful toolbox for tailoring cell behaviour in vitro. Front Med Technol 3: 670274.
- Tandon N, Cimetta E, Taubman A (2013) Biomimetic electrical stimulation platform for neural differentiation of retinal progenitor cells, in 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, pp. 5666-5669.
- Peltier J, O'Neill A, Schaffer DV (2007) PI3K/Akt and CREB regulate adult neural hippocampal progenitor proliferation and differentiation. Dev Neurobiol 67 (10): 1348-1361.
- Dong ZY, Pei Z, Wang YL, Li Z, Amber K, et al. (2019) Ascl1 regulates electric field-induced neuronal differentiation through PI3K/Akt pathway. Neuroscience 404: 141-152.
- Min Cheol J, Chul Hwan J, Jong TP, Seung WC, Seungil R, et al.92018) Effect of electrical stimulation on neural regeneration via the p38-RhoA andERK1/2-Bcl-2 pathways in spinal cord-injured rats. Neural Regen Res 13(2): 340-346.
- M Wang, P Li, M Liu, W Song, Q Wu, Y Fan (2013) Potential protective effect of biphasic electrical stimulation against growth factor-deprived apoptosis on olfactory bulb neural progenitor cells through the brain-derived neurotrophic factor-phosphatidylinositol 3'-kinase/Akt pathway. Exp Biol Med 238(8): 951-959.
- Yang K, Yu SJ, Lee JS, Lee HR, Chang GE, et al. (2017) Electroconductive nanoscale topography for enhanced neuronal differentiation and electrophysiological maturation of human neural stem cells. Nanoscale 9(47): 18737-18752.
- Thrivikraman G, Boda SK, Basu B (2018) Unraveling the mechanistic effects of electric field stimulation towards directing stem cell fate and function: a tissue engineering perspective. Biomaterials 150: 60-86.






























