Evaluation of Mitomycin C Effect on Corneal Endothelial Cell Density After Photorefractive Keratectomy
Kianoush Shahraki, Hossein Moghtader, Paria Ghasemi Boroumand and Kourosh Shahraki*
1Department of Ophthalmology, Alzahra Eye Hospital, Zahedan University of Medical Sciences, Zahedan, Iran
2ENT, Head and Neck Research Center and Department, Iran University of Medical Science, Tehran, Iran
3Assistant Professor of Ophthalmology, Oculofacial plastic fellowship, Department of Ophthalmology, Zahedan University of Medical Sciences, Iran
Submission: August 29, 2023; Published: September 05, 2023
*Corresponding author: Kourosh Shahraki, Assistant Professor of Ophthalmology, Oculofacial plastic fellowship, Department of Ophthalmology Health Promotion Research Center, Zahedan University of Medical Sciences, Zahedan, Iran
How to cite this article: Kianoush Shahraki, Hossein Moghtader, Paria Ghasemi Boroumand and Kourosh Shahraki. Evaluation of Mitomycin C Effect on Corneal Endothelial Cell Density After Photorefractive Keratectomy. JOJ Ophthalmol. 2023; 10(2): 555783. DOI: 10.19080/JOJO.2023.10.555783
Abstract
Background: Photorefractive keratectomy (PRK) is a commonly performed surgical procedure for the correction of refractive errors, such as myopia. Mitomycin C (MMC) has been used as an adjunctive agent to modulate the healing response and reduce haze and fibrosis after PRK. However, the effect of MMC on corneal endothelial cell density, a crucial factor in maintaining corneal transparency and function, remains a subject of debate. This study aimed to evaluate the effect of intraoperative application of MMC 0.02% during PRK on corneal endothelial cell density.
Methods: In this case series study conducted at Alzahra Eye Hospital of Zahedan University of Medical Sciences in 2021, a total of 40 eyes from 20 patients with myopia (spherical equivalent refractive error ranging from ‑3.0 to ‑8.0 Diopter) underwent PRK with intraoperative application of mitomycin C 0.02% for a duration of 40 seconds. Preoperative and postoperative assessments of central corneal endothelial cell density were performed using confocal microscopy with Confoscane III (Nidek Japan), with the latter assessment conducted 3 months after the surgery.
Results: The mean preoperative central corneal thickness was determined to be 533.875±23.6 microns, while the mean ablation depth was measured at 81.435±18.737 microns. Additionally, the mean preoperative spherical equivalent was found to be ‑4.65±1.2 D. The analysis revealed that the mean endothelial cell density experienced an insignificant reduction from 2,825±305 cells/mm2 preoperatively to 2,815±330 cells/mm2 postoperatively (P = 0.539).
Conclusion: Based on our findings, the application of MMC 0.02% for 40 seconds during PRK in patients with moderate myopia did not yield a significant impact on central corneal endothelial cell density after a 3-month follow-up period.
Conclusion: Choroidal hemangiomas are rare benign tumors. They can be problematic in their management because they can mimic choroidal melanomas.
Keywords: Endothelial Cell; Cornea; Mitomycin C; PRK
Abbreviations: PRK: Photorefractive keratectomy; MMC: Mitomycin C
Introduction
Photorefractive keratectomy (PRK) is a widely performed surgical procedure for the correction of refractive errors, such as myopia [1]. While PRK is effective in improving visual acuity, it can also lead to complications such as corneal haze and scarring, which can impact visual outcomes. Mitomycin C (MMC), an antifibrotic agent, has been used as a pharmacologic adjunct to modulate the healing response and reduce fibrosis after PRK, potentially improving surgical outcomes. However, the effect of MMC on corneal endothelial cell density, a critical factor in maintaining corneal transparency and function, remains a topic of debate. [1,2] The corneal endothelium plays a vital role in maintaining corneal clarity by regulating corneal hydration and transparency [3]. Damage or loss of corneal endothelial cells can lead to corneal edema and compromised vision.3 Therefore, understanding the effect of MMC on corneal endothelial cell density is crucial for evaluating the safety and long-term outcomes of PRK procedures.
MMC is a potent anti-tumor Alkylating medication that was first isolated from Streptomyces Caespitosus in 1956 [1,4]. Its mechanism of action involves disrupting DNA replication, inhibiting protein synthesis, and blocking mitosis. While it affects all cells, Mitomycin C exerts a particularly strong influence on highly proliferative cells [4,5]. In the field of Ophthalmology, Mitomycin C finds wide application in the treatment of pterygium1, primary and recurrent malignancies [6], squamous cell lesions of the ocular surface [7], and Trabeculectomy. Its safety and efficacy have been established through extensive laboratory animal studies, leading to its recommendation for reducing post-surgical opacity in refractive surgeries [1,2]. Photorefractive Keratectomy (PRK) is a commonly performed refractive surgery that has been proven effective and safe in correcting refractive errors [1]. By utilizing an argon fluoride excimer laser with a wavelength of 193 nm, PRK reshapes the anterior surface of the cornea, thereby reducing refractive errors. During the procedure, the epithelium is removed, and the stromal tissue is sculpted to alter the corneal refractive power. As a consequence, changes in the number and morphology of cells occur, leading to variable levels of extracellular matrix damage and the formation of Myofibroblasts [8]. This can result in reduced corneal transparency, manifested as sub-epithelial opacity, which may be clinically clear in some patients [9]. Numerous studies have consistently demonstrated that the occurrence of corneal opacity following photorefractive keratectomy can be significantly reduced with the use of Mitomycin C [1,9,10]. Mitomycin C serves as an effective pharmacological intervention to modulate the wound healing response, reduce fibrosis, and improve the outcomes of keratorefractive surgery [9,10].
Prophylactic application of a 0.02% Mitomycin C solution has been shown to reduce corneal opacity and enhance visual acuity following corneal refractive surgery [1,10]. However, concerns have been raised regarding the notable toxic effects and potential side effects of Mitomycin C [10]. It is capable of damaging all layers of the cornea, including the epithelial cells, stromal cells, and endothelial cells [1]. The optimal concentration and duration of Mitomycin C application that minimizes post-surgical opacity while causing minimal impact on corneal cells have yet to be determined. Various durations ranging from 13 seconds to 2 minutes and concentrations ranging from 0.01% to 0.4% have been utilized. Several studies have examined the effect of Mitomycin C on corneal endothelial cells in the context of refractive corneal surgery. Both clinical [11] and laboratory [12] investigations have explored the toxic effects of Mitomycin C on corneal endothelial cells. However, most clinical studies have failed to demonstrate significant changes in the number and morphology of endothelial cells within 3 to 18 months following Mitomycin C application [10,11]. This study aimed to contribute to the existing body of knowledge by evaluating the effect of MMC on corneal endothelial cell density in patients with moderate myopia.
Materials and Method
The present study encompasses all patients who sought treatment at Alzahra Ophthalmology Clinic for PRK, with a range of approximate myopia values between -3D and -8D. The Exclusion criteria were:
• Cases where refractive surgery is contraindicated, such as sever dry eye syndrome, corneal dystrophy, keratoconus, corneal ulcers, cataracts, glaucoma, and active retinal disease.
• Individuals who would be left with less than 400 microns of corneal stroma after the surgery.
• Patients who did not return for further examinations.
The eligible patients provided their informed consent and underwent a comprehensive ophthalmic examination. The number of corneal endothelial cells was quantified using Confoscan III (Nidek Co), and the resulting data were recorded in the device. The clinical information of the patients, including the assessment of their uncorrected visual acuity and best corrected visual acuity with glasses (UCVA, BCVA) using the Snellen chart (auto chart projector CP 670; Nidek Co., Ltd, Gamagori, Japan), manifest and cycloplegic refractive error, anterior and posterior segment examination using slit lamp biomicroscopy (Photo-Slit Lamp BX 900; Haag-Streit, Koeniz, Switzerland) , intra ocular pressure assessment with the Goldmann applanation tonometer (AT900, Haag-Streit, Koeniz, Switzerland), and Pentacam evaluation (Pentacam 70700: Oculus, Wetzlar Germany), were documented in dedicated forms. At the conclusion of the followup period (at the end of the third month), the corneal endothelial cells were counted once again using Confoscan III. For each phase of cell counting, manual images were captured from the central segment of the patient’s cornea. The Navis software, installed on the device, was utilized to manually count the number of cells in a 0.5 mm2 area.
The surgeries were performed by one experienced surgeon (K.S) using the Nidek vision excimer laser system (Nidek Co., Ltd., Gamagori, Japan). Corneal anesthesia was achieved using 0.5% Tetracaine hydrochloride, while 10% betadine was applied as an antiseptic to the skin surrounding the eye and eyelid for a duration of 3 minutes. A saline solution of 20 cc was used for rinsing. The central 8 mm epithelium of the cornea was removed for 20 seconds using 96% alcohol, followed by photoablation using the appropriate software. Subsequently, a 2 mg vial of Mitomycin C was diluted with 10 cc of distilled water. A sponge soaked in 0.02 Mitomycin C was placed on the corneal stroma for 40 seconds after laser removal. The corneal stroma was then rinsed with BBS solution and replaced with a contact lens (B&L). The following post-surgical drops were administered: 0.5% Chloramphenicol every 6 hours for one week, and 0.1% betamethasone every 6 hours until full epithelialization of the cornea. Once the corneal epithelium had fully recovered (between the 3rd and 5th days), the contact lens was removed, and follow-up examinations were conducted on the 1st, 3rd, and 7th days, as well as the 1st and 3rd months post-surgery. As previously mentioned, the corneal endothelial cells were counted once again using Confoscan III at the end of the third month. (Figure 1,2) Finally, all the collected information, both pre- and post-operation, was subjected to statistical analysis.
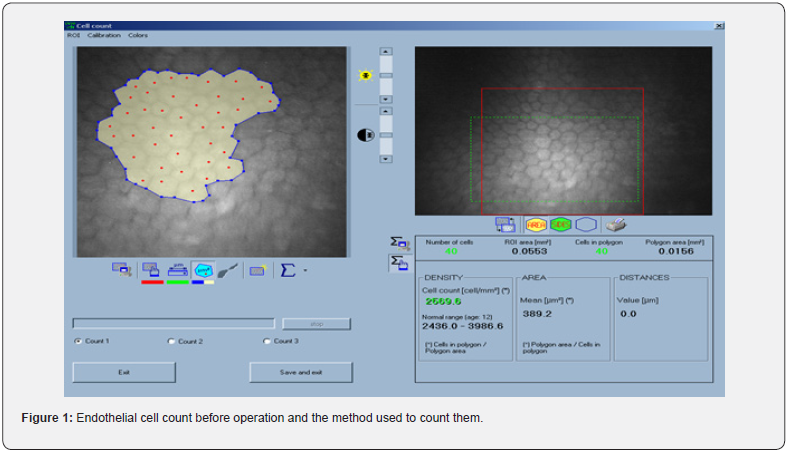
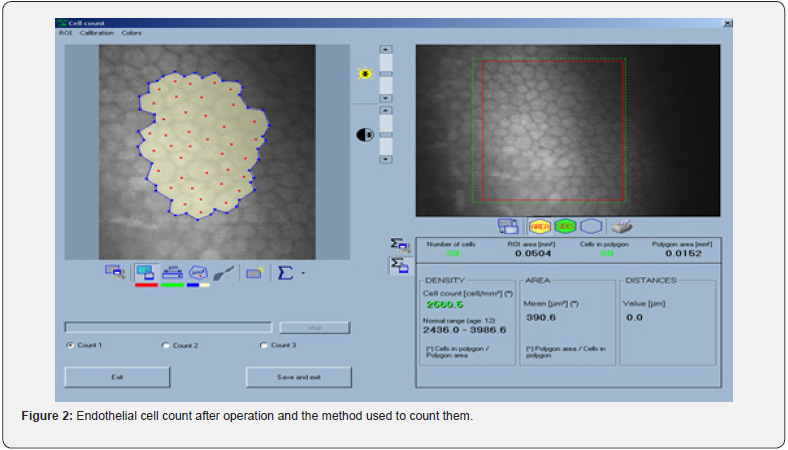
Results
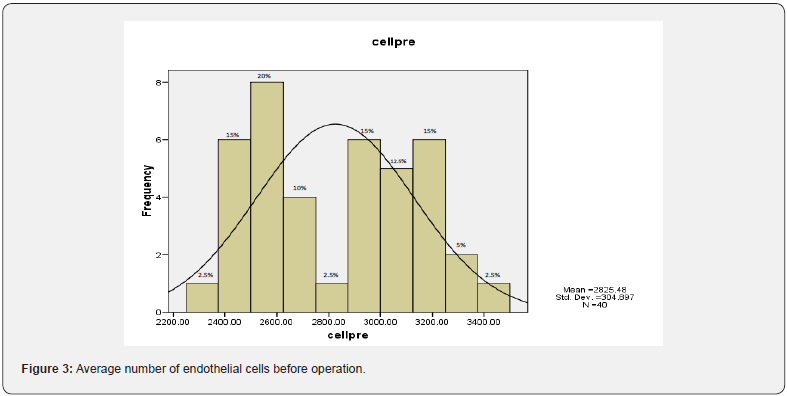
A total of 20 participants (40 eyes) from the patients seeking treatment at the Alzahra Eye Hospital for PRK with a myopia range of -3D to -8D, who met the exclusion/inclusion criteria and maintained their conditions until the conclusion of the research, graciously participated in this study. Among them, 4 males (20%) and 16 females (80%) were meticulously examined. The average thickness of the central region of the patients’ cornea prior to the surgical procedure was determined to be 533.875 ± 23.600 microns, with the minimum and maximum thickness recorded as 491 and 582 microns, respectively. Furthermore, the average depth of corneal thickness extraction was found to be 81.435 ± 18.737 microns, with the least and greatest thickness values measured at 49.60 and 120.30 microns, respectively. The mean spherical equivalent of the patients’ condition prior to the operation was calculated as -4.650 ± 1.2 diopters, with the lowest and highest recorded values being -3.00 and -7.125 diopters, respectively. Additionally, the average count of endothelial cells prior to the operation was established as 2825 ± 305, with the minimum and maximum cell counts observed at 2359 and 3415 cells, respectively (Figure 3).

Subsequently, three months after the operation, the average count of endothelial cells was determined to be 2815 ± 330, with the lowest and highest recorded counts standing at 2257 and 3619 cells, respectively (Figure 4). It is noteworthy that the average count of endothelial cells before the operation was 2825 ± 305, with the lowest and highest counts observed at 2359 and 3415, respectively. Similarly, the average count of endothelial cells three months after the operation was 2815 ± 330, with the lowest and highest counts recorded at 2257 and 3619, respectively. Importantly, no statistically significant difference was found between the count of endothelial cells before and after the operation (P = 0.539) (Table 1).
Discussion
The prophylactic application of 0.02% Mitomycin C has been shown to reduce corneal opacity and improve visual power after corneal refractive surgery [9]. However, concerns have been raised about the toxic effects and side effects of Mitomycin C. Several other studies have reported no measurable changes in the number and shape of endothelial cells after using 0.02% Mitomycin C for 12 seconds to 2 minutes, which supports the findings of our study. In a retrospective study by Lee et al, which included 1011 eyes, it was found that the number of corneal endothelial cells did not decrease after topical application of 0.02% Mitomycin C solution for 30 seconds to 2 minutes following PRK [13]. Similarly, a study by Goldsberry et al found no changes in the number and shape of endothelial cells after one year in 16 eyes that received 0.02% Mitomycin C after PRK [14].
On the other hand, Diakonis et al studied 15 eyes that received 0.02% Mitomycin C for 15 seconds after PRK and compared them to the other eye that underwent epi-LASIK. They observed a reduction in the number of endothelial cells at 1 and 3 months after the operation, but this reduction was not statistically significant [15]. Nassiri et al conducted a prospective study on 162 eyes and concluded that using 0.02% Mitomycin C would lead to a significant reduction in the number of endothelial cells within 6 months, which was correlated with the duration of contact 11. According to the results of a prospective study by Morales et al on 18 eyes, a contact time of 30 seconds with 0.02% Mitomycin C caused a certain reduction in the number of endothelial cells at 1 and 3 months [16]. The clinical significance of these two studies and laboratory studies [12] that demonstrate the toxic effects of Mitomycin on endothelial cells is still unclear because no cases of corneal endothelial failure following surface withdrawal have been reported since its first use in corneal refractive surgeries in 1991. The reduction in the number of keratocytes after the use of Mitomycin depends on the density and duration of usage. A similar correlation can be assumed for the effect of Mitomycin on endothelial cells. The depth of withdrawal is also an important factor. As the depth increases, less stroma remains, and higher densities of the medicine can penetrate and accumulate in the anterior chamber [14]. The use of 0.02% Mitomycin C for 12 seconds to 2 minutes after superficial withdrawal was not toxic to endothelial cells due to the shallow depth of withdrawal [15,17].
According to this study, using 0.02% Mitomycin C for 40 seconds during PRK had no adverse effect on the number of endothelial cells in patients with average myopia during the 3-month follow-up period. The main advantage of this study is the inclusion of patients with average myopia, who represent the majority of cases in refractive surgery. Additionally, the duration of Mitomycin usage in this study was nearly the maximum time used in surgery. Therefore, the results of this study can be applied to most cases of refractive surgeries involving Mitomycin C. We also utilized Confoscan III to count endothelial cells, which has not been used in previous studies. A 3-month follow-up period is not sufficient to determine the long-term safety of 0.02% Mitomycin C for endothelial cells, and further long-term follow-up is necessary to draw a definitive conclusion. In this study, the application of MMC 0.02% for 40 seconds during PRK in patients with moderate myopia did not significantly affect central corneal endothelial cell density after a 3-month follow-up period. The slight reduction in cell density observed in the postoperative period was not statistically significant. These findings suggest that MMC, when used within the specified parameters, may not have a detrimental effect on corneal endothelial cells in the short term.
The lack of a significant reduction in corneal endothelial cell density observed in this study may be attributed to the low concentration of MMC used (0.02%) and the short duration of application (40 seconds). Previous studies that reported a significant reduction in cell density often used higher concentrations and longer durations of MMC application. It is possible that the lower concentration and shorter duration in this study were within a safe range that did not cause significant damage to corneal endothelial cells.
Conclusion
In conclusion, the application of MMC 0.02% for 40 seconds during PRK in patients with moderate myopia did not significantly affect central corneal endothelial cell density after a 3-month follow-up period. These findings suggest that the use of MMC within the specified parameters may be safe in terms of corneal endothelial cell health. However, further studies with larger sample sizes and longer follow-up periods are warranted to validate these results and provide more comprehensive insights into the long-term effects of MMC on corneal endothelial cells after PRK. Understanding the impact of MMC on corneal endothelial cell density is crucial for optimizing surgical outcomes and ensuring the long-term safety of PRK procedures.
Acknowledgement
This paper is dedicated to the memory of our dear co-worker Dr. Mohammad Naeim Aminifard, who sacrifices his life for his country and people.
References
- Shahraki K, Saeidi S, Boroumand PG, Shahraki K. Effects of mitomycin-C on tear film function following photorefractive keratectomy for mild-to-moderate myopia. Medical hypothesis, discovery & innovation in optometry. 2021;2(4):150-5.
- Jabbarvand M, Khodaparast M, Moravvej Z, et al. Vector analysis of moderate to high myopic astigmatism after small-incision lenticule extraction (SMILE): 12-month follow-up. Eur J Ophthalmol. 2022;32(6):3312-3320. doi:10.1177/11206721221080821
- Shahraki K, Fard MNA, Shahri F, et al. Effects of intracameral cefuroxime on corneal endothelial cell counts and its morphology after cataract surgery. Interv Med Appl Sci. 2017;9(2):100-104. doi:10.1556/1646.9.2017.2.13
- Bahar I, Kaiserman I, Lange AP, et al. The effect of mitomycin C on corneal endothelium in pterygium surgery. Am J Ophthalmol. 2009;147(3):447-452.e1. doi:10.1016/j.ajo.2008.08.035
- Kim TI, Pak JH, Lee SY, Tchah H. Mitomycin C-induced reduction of keratocytes and fibroblasts after photorefractive keratectomy. Invest Ophthalmol Vis Sci. 2004;45(9):2978-2984. doi:10.1167/iovs.04-0070
- Bagheri A, Shahraki K, Yazdani S. Trichloroacetic acid 10% injection for treatment of conjunctival inclusion cysts. Orbit. 2020 Apr;39(2):107-111. doi: 10.1080/01676830.2019.1631358. Epub 2019 Jul 8. PMID: 31282238.
- Shahraki K, Shahraki K, Ghasemi Boroumand P, Sheervalilou R. Promotor methylation in ocular surface squamous neoplasia development: epigenetics implications in molecular diagnosis. Expert Rev Mol Diagn. 2023 Aug 7:1-17. doi: 10.1080/14737159.2023.2240238. Epub ahead of print. PMID: 37493058.
- Shahraki K, Hosseini SR, Amini Fard A, et al. Effects of Topical 1% Sodium Hyaluronate and Hydroxypropyl Methylcellulose in Treatment of Corneal Epithelial Defects. Med Hypothesis Discov Innov Ophthalmol. 2016;5(4):136-144.
- Wilson SE. Biology of keratorefractive surgery- PRK, PTK, LASIK, SMILE, inlays and other refractive procedures. Exp Eye Res. 2020;198:108136. doi:10.1016/j.exer.2020.108136
- Giral JB, Bloch F, Sot M, et al. Efficacy and safety of single-step transepithelial photorefractive keratectomy with the all-surface laser ablation SCHWIND platform without mitomycin-C for high myopia: A retrospective study of 69 eyes. PLoS One. 2021;16(12):e0259993. Published 2021 Dec 7. doi:10.1371/journal.pone.0259993
- Nassiri N, Farahangiz S, Rahnavardi M, Rahmani L, Nassiri N. Corneal endothelial cell injury induced by mitomycin-C in photorefractive keratectomy: nonrandomized controlled trial. J Cataract Refract Surg. 2008;34(6):902-908. doi:10.1016/j.jcrs.2008.03.007
- Khodaparast M, Shahraki K, Jabbarvand M, Shahraki K, Rafat M, Moravvej Z. Sutureless Femtosecond Laser-Assisted Anterior Lamellar Keratoplasty Using a Bioengineered Cornea as a Viable Alternative to Human Donor Transplantation for Superficial Corneal Opacities. Cornea. 2020;39(9):1184-1189. doi:10.1097/ICO.0000000000002394
- Lee DH, Chung HS, Jeon YC, Boo SD, Yoon YD, Kim JG. Photorefractive keratectomy with intraoperative mitomycin-C application. J Cataract Refract Surg. 2005;31(12):2293-2298. doi:10.1016/j.jcrs.2005.05.027
- Goldsberry DH, Epstein RJ, Majmudar PA, et al. Effect of mitomycin C on the corneal endothelium when used for corneal subepithelial haze prophylaxis following photorefractive keratectomy. J Refract Surg. 2007;23(7):724-727. doi:10.3928/1081-597X-20070901-14
- Diakonis VF, Pallikaris A, Kymionis GD, Markomanolakis MM. Alterations in endothelial cell density after photorefractive keratectomy with adjuvant mitomycin. Am J Ophthalmol. 2007;144(1):99-103. doi:10.1016/j.ajo.2007.03.039
- Morales AJ, Zadok D, Mora-Retana R, Martínez-Gama E, Robledo NE, Chayet AS. Intraoperative mitomycin and corneal endothelium after photorefractive keratectomy. Am J Ophthalmol. 2006;142(3):400-404. doi:10.1016/j.ajo.2006.04.029
- Eftekhari MH, Aghaei H, Kangari H, et al. Abobotulinum toxin A for periorbital facial rejuvenation: impact on ocular refractive parameters. Clin Exp Optom. 2021;104(1):115-118. doi:10.1111/cxo.13117






























