Beyond Atmospheric Oxygen and Pulmonary Alveoli, where does the Oxygen we have Inside the Human Body come from? the Unexpected Intrinsic Property of Melanin to Dissociate the Water Molecule
Arturo Solís Herrera*, María del Carmen Arias Esparza and Paola Eugenia Solís Arias
Human Photosynthesis© Research Center, Aguascalientes 20000, México
Submission: May 20, 2022;Published: February 14, 2023
*Corresponding author: Arturo Solís Herrera, Human Photosynthesis© Research Center, Aguascalientes 20000, México
How to cite this article: Arturo Solís Herrera, María del Carmen Arias Esparza and Paola Eugenia Solís Arias. Beyond Atmospheric Oxygen and Pulmonary Alveoli, where does the Oxygen we have Inside the Human Body come from? the Unexpected Intrinsic Property of Melanin to Dissociate the Water Molecule. JOJ Ophthalmol. 2023; 9(4): 555768. DOI: 10.19080/JOJO.2023.09.555768
Abstract
The teaching of the medical career is based on dogmas, as in religions. It is a dogma very rooted the wrong belief that our body obtains the oxygen it contains from the air we breathe. Plants, which do not absorb atmospheric oxygen, expel it but they obtain it from the dissociation of the water molecule through chlorophyll. It is another false belief that the oxygen that plants expel, they do it for the benefit of the animal kingdom, because supposedly said oxygen is absorbed through the pulmonary alveoli into the bloodstream, to distribute it to the cells of the organism, which use it to generate energy by combining it with glucose, something like controlled combustion. However, 95% of biochemical reactions that try to concatenate the combination of this atmospheric oxygen with glucose and the energy our body requires are theoretical. It is not possible to consistently explain that the human body consumes and recycles about 150 kg of ATP daily. Each ATP molecule is supposed to be reconstituted about 1000 times every 24 hours. Besides that, each reconstitution requires energy expenditure. On the other hand, since 1893, Haldane demonstrate that blood oxygen concentrations could not be explained by diffusion, and to date textbooks continue to insist that diffusion is the mechanism by which atmospheric Oxygen passes through lung tissues by a theoretical mechanism and reaches the bloodstream. Apparently, these notable discrepancies between the theory and the biology of the human body had been set aside because we had no better way to explain it. Our discovery of the unsuspecting ability of the human body to use the energy of the sun directly through the dissociation of water, such as chlorophyll in plants; opens a new era in Biology and Medicine, which will allow us to correct collective mistakes in the care of the sick.
Keywords: Oxygen; Hydrogen; Water; Dissociation; Energy; Sunlight
Background:
Since the Greeks the idea had spread that substances were combustible by virtue of the “matter of fire” that was present in them. The great English chemist, Robert Boyle (1627-1691), had shown in 1672, that combustion could not be carried out in a vacuum. An initial explanation was the Phlogiston, from the Greek phlogiston (phlos = flame), is “what goes” during combustion, the equivalent of our current hydrogen. This theory was quickly accepted, but contradictions began to emerge that did not solve the riddle of the need for air for combustion. The main difficulty was that the metals gain weight after combustion or calcination. How to reconcile weight gain with the release of something during combustion?
The first serious blows against the phlogiston theory are formulated in 1772 mainly by Lavoisier. who demonstrates that the increase in the weight of the calcined metal is equal to the weight of the absorbed air and concludes that only a part of the air can combine with the metals or serve for respiration. Lavoisier calls this component of the atmosphere “pure” air, which he will later call “imminently breathable air”, later “oxygynous principle”, and then oxygen, from the Greek roots “oxys,” meaning “sharp” in reference to the sour taste of acids and the assumption by Lavoisier that it was an essential element for all acids, and “-genes,” which means “begetter”.
The Swedish pharmacist Carl Wilhelm Scheele (1742-1786) experimentally demonstrated that the atmosphere was composed of corrupt or stale air and pure air or fire air. Oxygen is discovered in 1774 but is not characterized. Oxygen is colorless, tasteless, and has no odor. Lavoisier proposed the role of oxygen in oxidizing metals and in respiration, supposing that it was taken up by the body during inhalation to allow slow combustion of organic substrates and that carbon dioxide was exhaled as a by-product [1].
The naming of oxygen as “begetter” proved correct, considering the essential role it plays in sustaining life on our planet. In terms of mass, it is the third most abundant element in the universe, following hydrogen and helium. It constitutes 50% of the earth’s crust and comprises a third of the mass of humans, finding its way into essential molecules such as proteins, carbohydrates, and fats in addition to non-cellular structural elements such as bones and teeth. But its role in aerobic respiration sparks the greatest interest for oxygen among pulmonary physicians and respiratory therapists.
So far, Oxygen serves (theoretically) as the final electron acceptor in catabolic reactions that convert biochemical energy from nutrients into adenosine triphosphate, which fuels the human body. Because of its highly reactive nature, oxygen serves as an effective electron accepter, or oxidizing agent. This reactive nature forms oxides when oxygen meets almost all other elements explains why it can exist in a free gaseous form only through ongoing generation by photosynthesis. Consequently, oxygen was a relatively recent addition to our atmosphere, beginning to accumulate only 2.5 billion years ago after plant life with sufficient photosynthetic capacity populated the planet [2].
The human body’s energy metabolism is so tangled that ATP is said to be an energy hauler, but not an energy source. That is, ATP contains no energy, but must be imbued. Which is supposedly achieved through oxidative phosphorylation, which implies the presence and availability of oxygen. However, oxygen from the atmosphere cannot penetrate through the lungs and reach the bloodstream, nor can oxygen travel from the blood to the tissues, as since 1960 it has been reported that molecular oxygen cannot cross the blood-brain barrier [3]. A growing number of studies have addressed the blood-brain barrier characteristics [4] and it is now well documented that most hydrophilic molecules and ions pass through this membrane extremely slowly.
The observed brain tissue values are substantially below venous PO2 values. Average cell, venous, and arteriolar tensions are ≈ 20 mm Hg [5]. Values derived from tissue spectrophotometry indicate that brain tissue PO2 values may be considerably lower than this. In isolated mitochondria, cytochrome a3 is completely oxidized above PO2 values of 1.0 mm Hg [6]. Reflectance spectrophotometry reveals a significant amount of reduced cytochrome a3 in the brain of the anesthetized animal, [7] suggesting that even lower PO2 values may be present in the tissue.
Neither value is explainable with the classical Krogh [8] model, which envisages no resistance to the passage of oxygen from blood capillary to tissue; tissue values substantially lower than venous would be explained if there were a microvascular resistance to the transfer of oxygen. Interestingly, experiments with oxygen 18, to study the flow of oxygen into the brain, were not done via pulmonary, but oxygenated blood with oxygen 18 was injected through the carotid.
Introduction
Diffusion is defined as the tendency of molecules and atoms to occupy all the space around them. Displacing of the highest concentration to the lower concentration zone. Therefore, if the composition of the gases in the blood depended on such a simple mechanism, the composition of the gases in the bloodstream would be a reflection of the composition of the atmosphere, i.e., 78 % Nitrogen, 21 % oxygen, 0.039% CO2, etc. However, the concentration of N2 in blood at 37 °C (body temperature) and atmospheric pressure (partial pressure of N2 = 0.80 atm) is 0.00056 mol/L or 5.6 x 10-4 mole/L. Nitrogen has a molar mass of 14.0067 g/mol. The molar mass of nitrogen gas is 28 g/L. The volume ratio is equal to molar ratio, thereby, Nitrogen in dry air has a concentration of 0.78084 mol/molair and 78.084 vol %. The molar mass of Nitrogen in air is 21.872266 g/molair.
Oxygen has a molar mass of 15.9994 g/mol. The molar mass of oxygen gas is 32 g/mol. The molar mass of oxygen in air is 6.701942 g/molair. Carbon dioxide has a volume ratio = molar ratio compared to dry air of 0.000412 mol / mol air and 0.412 vol%. CO2 has a molar mass of 44.010 g/mol or kg/kmol. The molar mass of Carbon dioxide in air is 0.018132 g/molair. A deep-sea diver breathes compressed air with the partial pressure of N2 equal to 4.8 atm.
However, the normal SpO2 in blood is more than 90 %, thereby, nitrogen in the blood does not correspond to the composition of nitrogen in the atmospheric air. If diffusion is a passive mechanism by which molecules move, thereby, we would have to think that something has prevented nitrogen from spreading from the atmosphere into the bloodstream. About oxygen, there seems to be no doubt, as it apparently spread rapidly reaching blood concentrations even higher than those of the atmosphere. Thus, if oxygen apparently passes easily through diffusion the pulmonary tissues into the blood, why does nitrogen not seem to do it?
The physical and chemical properties of both nitrogen and oxygen are similar; it is even a single proton difference in the atomic number of both. Carbon has 6 protons, Nitrogen 7, and oxygen 8 protons. Both carbon and nitrogen can be made to react with oxygen, but otherwise they are pretty inert. Supposedly oxygen is the only one we can use for energy production combining it in a very controlled way with glucose. But if glucose were an energy source, diabetics would fly. Glucose is the universal precursor to organic matter, but it is not able to provide the energy that its own metabolism requires.
The ancient idea that glucose has the simultaneous function of being the universal precursor while producing the energy that its own metabolism requires, seems to originate from Lavoisier’s work in Paris and Priestley in London almost 250 years ago. To date, however, of the nearly seven thousand chemical reactions in this regard, there is consensus in about 200 alone, the rest are controversial [9]. What was to be expected since it is not possible to extract energy where there is no energy. That is why the contradictions are numerous when we try to explain cellular metabolism from glucose as an energy source.
Briefly analyzing the sequence, we have that chlorophyll dissociates the molecule from water, so photonic energy, on which all living beings depend, is transformed into susceptible chemical energy from being used by living beings. Supposedly the energy released during the dissociation of the water molecule is stored in the covalent bonds of glucose, which is a contradiction as the energy cannot be stored.
The next step also poorly understood as the previous ones; is that mitochondria, through respiration, is able to recover the supposed energy stored in glucose by combining it with oxygen. But to do this, oxygen needs to be obtained from the atmosphere through breathing. It should be noted that the oxygen that plants throw into the atmosphere, they do not obtain it from the air around them, but from the water they contain.
Humans have many similarities to plants, and this could be one more, referring to the oxygen that our body has, we get it from the water inside the organism. And our discovery of melanin’s intrinsic property of dissociating the molecule from water, such as chlorophyll in plants, points in that sense.
The above contradictions about the energetic metabolism of glucose have not been resolved after almost 250 years. And to top it all off, the physical and chemical properties of oxygen, as well as those of water, make it highly unlikely that oxygen can pass through delicate lung tissues through diffusion and reach the bloodstream. It is not possible since molecular oxygen is a polar as oil, and water is a highly polar liquid. They simply do not combine, at most they are mixed by shaking them, but soon they are separated again.
The Physical and Chemical Properties of Oxygen
The physical and chemical properties of oxygen make the amount that can pass through lung tissues extremely limited. For instance, the diffusion rate of oxygen in water is 3.3 X 105 times lower than in air. Which is paradoxical if we stop to think that the amount of oxygen required by a 70 kg body to combine it with glucose (combustion) is in the order of liters per minute. Then one would expect the rate of oxygen diffusion in the water to increase 300,000 times, not decrease. It is shocking that the following sentence was published in 1897 by John Haldane and J Lorrain Smith: “From our observations, by the carbonic oxide method, on the oxygen tension of the blood as it leaves the lungs in man, we were led to the conclusion that the exchange of oxygen which takes place between the atmosphere and the blood cannot be adequately explained by diffusion alone.” [10].
To date it remains forgotten, but since then experimentally it was found that oxygen concentrations in the blood could not be explained merely by the mechanism of diffused atmospheric air to the bloodstream. Perhaps there was no better option, but to date, even textbooks described repeatedly that oxygen from the atmosphere passes into the bloodstream through the barriers that arise in its path by diffusion. The alveoli are adapted to make gas exchange in lungs happen easily and efficiently, referring CO2, and not oxygen. Some features of the alveoli that allow this are
a) they give the lungs a big surface area,
b) the have moist, thin walls. Recall that oxygen has velocity of diffusion in water 300000 times slower in water than in air. CO2 is just 1000 times slower in water than in the air.
c) Alveoli has a lot of tiny blood vessels or capillaries.
Supposedly, the gases move by diffusion from where they have a high concentration to where they have a low concentration. Oxygen diffuses from the air in the alveoli into the blood. Carbon dioxide diffuses from the blood into the air in the alveoli. The term diffusion is used indiscriminately to try to explain oxygen and CO2 saturation in both the blood and alveolar air, although experimental tests report since 1897 that diffusion does not explain the exchange of gases between the blood and lung.
The use of the term diffusion in textbooks seems to be justified by stating that the alveolar wall is equivalent to the thickness of a single cell, but any cell in the body contains on average 70% water. Furthermore, if we add that the surface of the alveoli is constantly moisturized, then we have the presence of an aqueous film. Thereby, we have several water barriers that repels oxygen instead of absorbing it. If we add to this that the stroma or supporting tissue also has a significant amount of water, followed by the cells with a minimum of 70% water that make up the wall of the capillary vessels, and in the end we have or bloodstream formed for the most part water, then we have to conclude that lung tissues repel oxygen without absorbing it.
It is as if nature counters our sacred concept that the lung introduces oxygen into the body. But experimental data confirm that the difficulties in oxygen entering the body are enormous, because everything seems to be against it for that to happen. Being objective, neither nitrogen nor oxygen can pass through lung tissues to enter the bloodstream, as the physical and chemical properties of both gases and tissues are opposite each other. Oxygen moves 300,000 times slower in water than in the air, and CO2 moves only 1000 times slower.
As expected, the rate of CO2 diffusion in water increases with increasing temperatures. A significant decrease in the rate of CO2 diffusion was found with increasing salinity [11]. There is an uncertainty associated with estimating the molecular diffusion coefficient of a gas in seawater from values obtained in pure water, reflecting the poor understanding of the effect of salinity on diffusion [12]. Carbon dioxide in air has a diffusion coefficient of 16 mm2/s, and in water its diffusion coefficient is 0.0016 mm2/s.
It can then be thought that CO2 is thousands of times more likely to travel from the bloodstream into the atmospheric air contained in the alveoli than the oxygen to penetrate the blood from the alveoli. And if we add the presence of an enzyme whose presence is in all living beings, and whose action is to accelerate the diffusion of CO2 in water by transforming it into carbonic acid which spontaneously transforms into bicarbonate which is soluble in blood plasma, and when it reaches the lungs, the same enzyme catalyzes the opposite reaction so that the bicarbonate is transformed back into gas , then we have to increase the solubility of CO2 in water, inside the organism, is increased not in thousands but in millions of times, because carbonic anhydrase has one of the highest known pKas: between 1 and 3 million molecules per second.
Just as important is the management of CO2 for nature, which is how our body gets rid of the oxygen that the dissociation of the water molecule produces, because by breaking down it we obtain hydrogen and oxygen at the same time. Although from an energy point of view the really valuable gas is hydrogen because it is the main energy hauler in the whole universe, therefore, once it is generated, our body makes the most of it, because it does not waste a single drop. And the oxygen that is generated is actually a necessary evil, for it has always been present in the equation. But it needs to be eliminated as soon as possible given its high toxicity, and to achieve it, our body combines it with carbon skeletons, from glucose; forming carbon dioxide.
A significant portion of CO2 that we exhale always results of the need to expel the toxic oxygen that is generated during water dissociation; and the remaining portion comes from the metabolism of the body’s nutrients. Ancient idea that living organisms derive their energy solely from food/fuel substrates, through either anaerobic or aerobic respiration, now is broken down in thousands of pieces. Anaerobic respiration does not involve the utilization of oxygen, like in beginning of times; however, it is an inefficient process of energy generation in the form of adenosine- 5’-triphosphate (ATP), which is mainly true because neither glucose nor ATP are sources of energy. The main purpose of anaerobic respiration is the rapid formation of biomass, so it is present in the embryo, in muscle cells, and in cancer cells.
Cellular respiration involves enzyme-catalyzed oxidation of fuel substrates, however fuel substrates do not exist, It is an old idea that these theoretical fuel substrates are combined primarily by oxygen, to yield the energy required for biologic processes; but if glucose were source of energy, diabetics should fly. Aerobic respiration takes place in two regions of the cell, glycolysis occurring in the cytoplasm, and Krebs’ cycle and electron-transport chain (ETS) in the mitochondria.
It is exhaustively describing how Glucose, the primary substrate for cellular aerobic respiration, is converted into pyruvate in the glycolytic phase, which is shunted into the Krebs’s cycle for oxidative phosphorylation that is undertaken by the ETS in the mitochondrial inner membrane. Oxygen undergoes a four-electron reduction in the ETS, thus generating ATP, the ultimate biologic currency of energy. However, these descriptions have significant differences in the different biochemistry books and are silent in terms of the energy source required by each and every transformation of the molecule, culminating in the misconception that ATP is the universal energy exchange currency, when it does not have a clear, even remote idea of what the mechanism by which ATP releases energy is, at most a conformational variation is adducted.
The whole process of cellular aerobic respiration yields 28 ATP molecules as opposed to two ATP molecules that are generated during anaerobic respiration. Thus, it is evident that the efficiency of the respiratory substrate (e.g., glucose) utilization to generate the maximal number of ATP molecules is best in the cells during aerobic respiration, in which the involvement of oxygen is essential. However, both mitochondria and ATP have functions related to temperature control, which is also strictly regulated in living beings, in addition to when ATP is hydrolyzed to ADP, energy is absorbed, and when ADP is transformed into ATP, energy is released. About the “indispensable” oxygen, we would have to reconsider its true function, for it is actually a necessary evil.
A part of the central nervous system, the medulla oblongata, controls the process of respiration and thereby the release of CO2 to the lungs. It can alter the process of respiration based on neurosensory feedback. The blood concentration of oxygen that comes from water dissociation and not from the atmosphere, carbon dioxide which is how the organism gets rid of the oxygen from the dissociation of the water that takes place in each and every cell in the body, and for this our body simply sticks a carbon skeleton to the oxygen molecule, forming carbon dioxide, and whose rate of diffusion in the water is 290,000 faster than that of molecular oxygen , allowing faster removal of toxic oxygen; because oxygen from the atmosphere cannot penetrate the blood through the lungs, it also cannot leave the bloodstream unless it is expelled in the form of CO2 and the pH (acid–base equilibrium) also are important in the regulation of respiration.
Overall, the essential basis for respiration at the cellular level is the availability of hydrogen, and not from oxygen. Hemoglobin is not merely an oxygen carrying-protein. Furthermore: Hemoglobin can dissociate the water molecule, irreversible, like chlorophyll in plants. But chlorophyll, given the anatomy of the leaf, can directly expel toxic oxygen into the atmosphere, limiting the damage that such a reactive molecule can cause to the biochemical processes of life, and instead hemoglobin, despite its great resemblance to chlorophyll, cannot expel lethal oxygen into the atmosphere, given the histological and anatomical circumstances of different tissues; so it needs to be transformed into a molecule whose diffusion rate is substantively higher than that of oxygen alone, so it simply adds a carbon atom, probably from glucose; which significantly increases the rate of diffusion of it, plus the presence of the extraordinary carbonic anhydrase enzyme.
By the way, when the body’s cells, through melanin, dissociate the water molecule; it obtains hydrogen and oxygen at the same time, but the energy that is released by breaking down the water molecule is captured and carried by hydrogen, the main energy hauler in the entire universe. Therefore oxygen, which is a poisonous gas, even lethal; neither the body nor any living, captures it from the atmosphere, but is generated from the water that the organism itself contains.
Therefore, it has been an equivocal conclusion that the molecular entry of oxygen into an actively respiring cell is metabolically driven, instead is expelled of the cell as soon as possible, for instance in the form of CO2. The supply of oxygen to the actively respiring cell cannot be interrupted by blockade of the blood supply such as ischemia because oxygen does not come from atmosphere. Instead, ischemia causes insufficient expelling of CO2 from tissues (cell), thus theoretically leading to hypoxia or anoxia when in fact it is tissue damage caused by the increasing concentration of CO2 or hypercapnia. In short, tissue ischemia leads to a detriment of their ability to rapidly evacuate CO2 that occurs constantly in cells and is transported to the lungs through blood plasma mostly in the form of bicarbonate.
Lipids of the cell membranes, including those of the plasma membrane, play a critical role avoiding oxygen entry into the lung cells and blood stream. It is almost impossible for oxygen to pass through the thin alveolar walls of the lungs. Atmospheric gases pass into the lung but do so by preserving the composition of atmospheric air; that is how far we agree; but neither oxygen nor atmospheric nitrogen is able to pass through the delicate walls of the cells that make up the alveolar walls, mainly because of the high water content of the cells.
From the behavior of gases in pure water, it is difficult to predict the behavior of gases in a complex aqueous medium such as that of cells, but in general they are considered to further hinder the diffusion of both oxygen and nitrogen to the bloodstream. For instance, an increase in the cholesterol concentration of the RBCs has been shown to decrease oxygen transport across the cells, presumably because of a decrease in the membrane fluidity and stiffening of the lipid bilayer [13].
Some of the cellular oxygen acts as an important substrate for several oxygenation reactions catalyzed by oxygenase (monooxygenases and dioxygenases) of nonmitochondrial sources [14], anyway, energy is needed to enzymes action which is provided by molecular hydrogen. The carotid body, situated in the carotid artery, apparently is the primary sensor of oxygen concentration in the mammals [15]. Glomus cells of the carotid body are depolarized in response to hypoxia and initiate a sequence of events. However, there is not a clear explanation of its mechanism of action, only theories. In our opinion, oxygen is only an indirect indicator of hydrogen levels since both gaseous elements are obtained at the same time by dissociation of the water molecule. It is congruent that when hydrogen levels drop, glomus cells cannot maintain proper polarization, and therefore depolarize.
The High Levels of Oxygen in the Eyeball come from the Dissociation of the Water Molecule
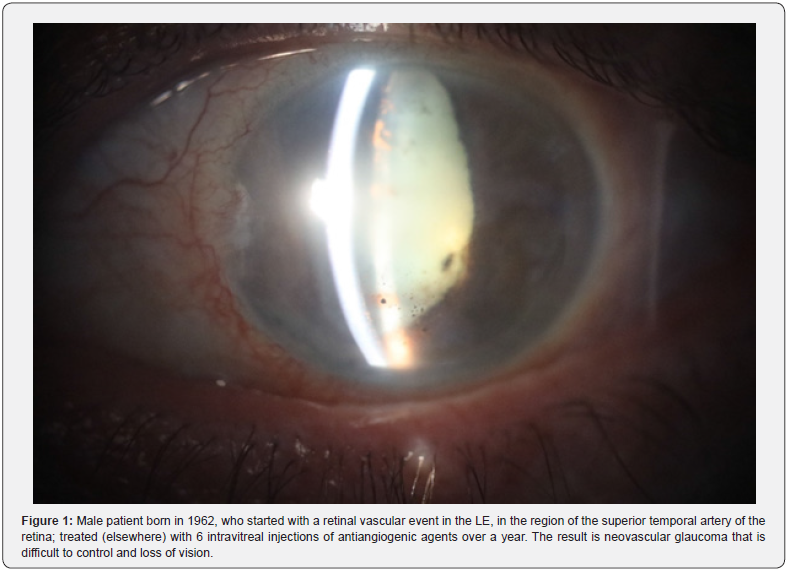
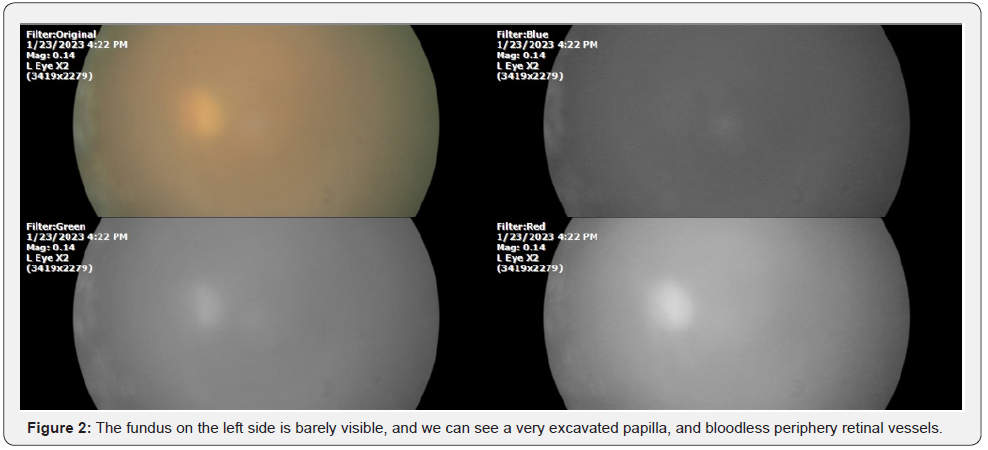
Until now it had been believed that the oxygen present in the blood vessels and eye tissues came from the atmosphere. And the interpretation of tissue alterations in ocular diseases, for example of the retina, were based on said dogma. But being a basic misconception, treatments developed on that basis give very poor results. Let’s take an example: a retinal vascular event, predominantly venous; If we treat it with intra-vitreous antiangiogenics, the results are as follows (Figure 1, 2):
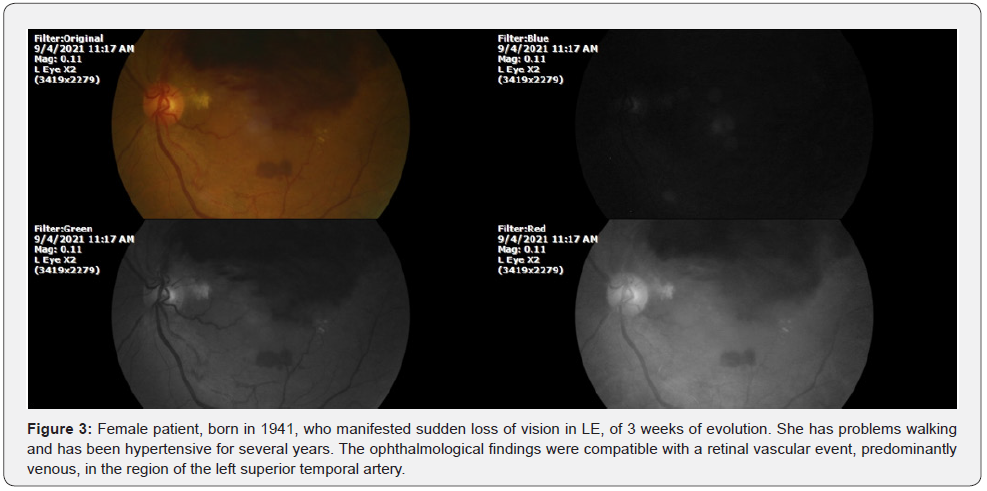
Intravitreal injections, based on selective immunogenic therapy, have not given the expected results. Local and systemic complications are very frequent, as demonstrated by this case. We present a similar case below, but the treatment was based on our discovery of the unsuspected ability of eukaryotic cells to take up or extract oxygen from the water they contain [16] (Figure 3).
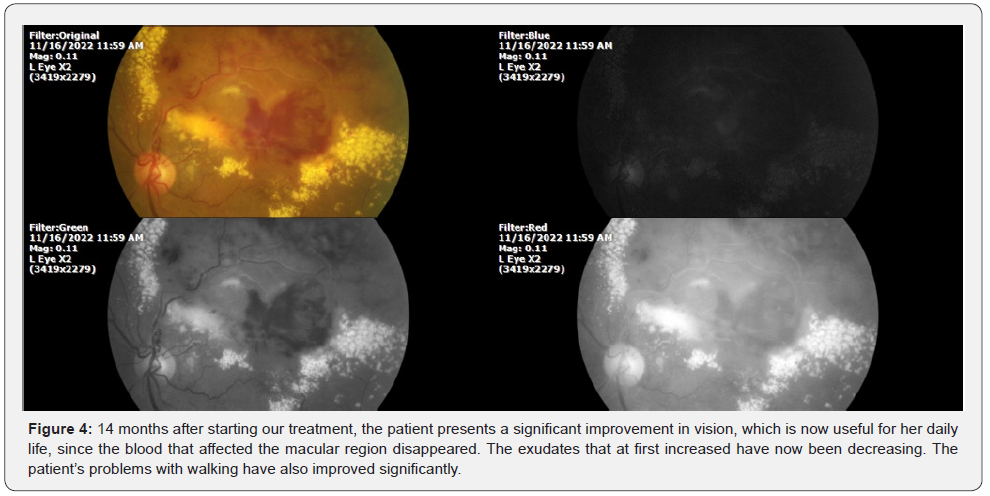
The therapeutic approach we used was explained to the patient and family members, and once they agreed and the informed consent was signed, we began treatment with QIAPI 1(TM), a pharmacological agent implemented in our facilities that restores the organism’s ability to take in or extract oxygen from the water it contains, like plants. The following clinical photograph was taken 14 months after the start of treatment (Figure 4):
Comment
According to the prevailing dogma, the oxygen required by the eye tissues was believed to come from the atmosphere, however the oxygen present in the atmosphere is relatively little, and on the other hand, the levels of oxygen inside the human eye are almost five times more than in the atmosphere. Such a difference cannot be explained by the anatomy of the lung, which is designed to expel almost 900 g of CO2 every 24 hours, but not to absorb the little oxygen that the atmosphere contains, concentrate it 5 times and then introduce it into the blood.
The origin of oxygen inside our body is a controversy that dates to the end of the 19th century and the beginning of the 20th century [17]. It seems ironic that the answer to this age-old controversy was resolved by an observational, descriptive study; about the vessels of the optic nerve and its possible correlation with the three main causes of blindness in the world (Macular Degeneration due to age, diabetic retinopathy, and glaucoma.) The fact that the oxygen we have in the body comes from the dissociation of water, as in plants, radically modifies our concepts about the etiopathogenesis and treatment of vascular events, not only in the retina but also in other parts of the human body, such as the brain, lung, muscle, intestine, etc.
If we restore the ability of the organism and the affected tissues to take in or extract the oxygen from the water that each cell contains, then the tissue tends to recover, without the need to dissolve or extract the spilled blood either with medicines or heroic measures, since the tissue with adequate levels of oxygen handles spilled blood adequately, with significant recovery of both anatomy and function.
Conclusion
THE concept that arose in Europe in the 18th century, about our body taking the oxygen it requires to function from the atmosphere, despite how widespread it is, has not been able to be explained, much less demonstrated. The theory in vogue, enunciated by Krogh at the beginning of the 20th century, is hypothetical, and it is such a complex mathematical model that it cannot even be tested experimentally [18]. Results like the one shown here in the case of retinal vascular events allow us to foresee a new era in problems that, to date, do not have a satisfactory solution. It is necessary to rethink cell biology as a whole, modifying the error that has been dragging on for centuries, but which can allow us to make significant progress in the fight against diseases that afflict the population and that could not be resolved because they were misunderstood.
Acknowledgements
This work was supported by an unrestricted grant from Human Photosynthesis™ Study Center. Aguascalientes 20000, México.
References
- Heffner, John E (2013) The Story of Oxygen. Respiratory Care January 58(1): 18-31.
- Emsley J (2001) Oxygen: nature’s building blocks: an A-Z guide to the elements. Oxford: Oxford University, pp. 297-304.
- Kassisia Ibrahim G, Goresky Karl A, Rose Colin P, Schwab Andreas J, Simard André, et al. (1995) Tracer Oxygen Distribution is Barrier-Limited int eh Cerebral Microcirculation. Circulation Research 77: 1201-1211.
- Paulson R, Barry D, Hortz M (1980) The blood-brain barrier in mammalians with special reference to insulin effects on glucose transport. Acta Neurol Scand 78: 147-156.
- Lubbers, DW (1981) Tissue Oxygen supply and critical oxygen pressure. In: Kovach ACR. Dora, E. Kosslar, M. Silver, IA (Eds.) Oxygen transport in tissues. Advances in physiological Science. Budapest, Hungary: Pergamon Press, UK, pp. 25: 3-11.
- Chance R, Oshima N, Sugama T, Maversky A (1973) A basic principle of tissue oxygen determination from mitochondrial signals. In: Richer HI, Brulov DE (Eds.) Oxygen Transport to Tissue, Plenum publishing Corp, New York, NY, pp. 277-292.
- Jöbsis EE, Plantadosi CA, Sylvia AL (1983) Cyanide-induced Cytochrome a, a3 Oxidation-reduction responses in rat brain in vivo. J. Clin. Invest 72(4): 1224- 1233.
- Krogh, A (1919) The number and distribution of capillaries in muscle with calculations of the avian pressure head necessary for supplying the tissue. J App Physiol 52(6): 409-415.
- Stobbe, Miranda D (2012) The road to knowledge: from biology to databases and back again. University of Amsterdam. UvA-DARE (Digital Academic Repository), Amsterdam, Netherlands.
- Haldane John, Smith J, Lorrain (1987) The absorption of oxygen by the lungs. 1897, from Pathological Laboratory, Queen´s College, Belfast, Northern Ireland.
- Zarghami Shahin, Boudaki Fathi, Al-Wahabi Yahya (2017) Diffusion of carbon dioxide in formation water as a result of CO2 enhanced oil recovery and CO2 J Petrol Explor Prod Technol 7: 161-168.
- King DB, De Bryun WJ, Zheng M, Saltzman ES (1995) Uncertainties in the molecular diffusion coefficient of gases in water for use in the estimation of air -sea exchange. Air-Water Gas Transf 33: 13 -23.
- Buchwald Henry, O´Dea Thomas J, Menchaca Hector, Rohde Thomas (2000) Effects of plasma cholesterol on red blood cell oxygen transport. Clin Exp Pharma Physiol 27(12): 951-955.
- Groves JT (2003) The bioinorganic chemistry of iron in oxygenases and supramolecular assemblies. Proc Natl Acad Sci USA 100(7): 3569-3574.
- Prabhakar NR (2000) Oxygen sensing by the carotid body chemoreceptors. J Appl Physiol 88(6): 2287-2295.
- Herrera AS, Del CA Esparza M, Md Ashraf G, Zamyatnin AA, Aliev G (2015) Beyond mitochondria, what would be the energy source of the cell? Cent Nerv Syst Agents Med Chem 15(1): 32-41.
- Gjedde A (2010) Diffusive insights: on the disagreement of Christian Bohr and August Krogh at the Centennial of the Seven Little Devils. Adv Physiol Educ 34(4): 174-185.
- Titus A, Sanghavi D (2022) Extracorporeal Carbon Dioxide Removal. 2022 Nov 7. In: Stat Pearls. Treasure Island (FL): Stat Pearls Publishing, USA.






























